The Relationship Between Taste and Smell Alterations and Quality of Life Among Women With Breast Cancer Receiving Chemotherapy
Objectives: To understand taste and smell alterations (TSAs) and their relationship with quality of life among women with breast cancer receiving chemotherapy.
Sample & Setting: A descriptive correlational study was conducted with 121 women with breast cancer receiving chemotherapy, and data were collected through an online survey.
Methods & Variables: The Taste and Smell Survey was used to measure TSAs, and the Functional Assessment of Cancer Therapy–Breast was used to measure quality of life.
Results: More than half of the participants experienced severe TSAs. The longest-lasting taste after the start of chemotherapy was a bitter taste, and participants experienced it more strongly after starting chemotherapy than before. The more severe the TSAs, the lower the overall quality of life, particularly physical well-being and functional well-being.
Implications for Nursing: Nurses should be vigilant about whether people with cancer receiving chemotherapy are experiencing TSAs. It is recommended to educate women in this population about avoiding bitter foods and foods with strong smells. Providing information about TSAs before treatment can help people with cancer cope.
Jump to a section
Breast cancer is the fifth most common cancer type among all cancers in Korea and the most frequently diagnosed among Korean women (National Cancer Information Center, 2021). Because the ability to diagnose breast cancer early and the development of treatment technology, the survival rate of people with breast cancer has greatly improved, and the five-year relative survival rate has reached 93% (National Cancer Information Center, 2021). This high survival rate may imply that the length of treatment time is increasing; therefore, additional support and research focused on quality of life rather than survival after treatment may be needed.
Chemotherapy is widely used as the main treatment for breast cancer; this treatment affects not only cancer cells but also noncancerous cells throughout the body (Yoshimoto et al., 2020). Chemotherapy affects the oral cavity, so individuals with cancer receiving chemotherapy experience oral complications like stomatitis, candidiasis, and taste and smell alterations (TSAs) (Ponticelli et al., 2017). TSAs are among the most common problems experienced by people with cancer undergoing chemotherapy (Donald, 2022). Although the precise mechanisms underlying TSAs are not yet clearly elucidated, the most widely accepted view is that they occur because of cytotoxic damage to taste and olfactory cells, which divide quickly (Sevryugin et al., 2021). Taste alterations were reported in 56%–76% of people with cancer undergoing chemotherapy, and smell alterations appeared in 16%–49% of this population (de Haan et al., 2021).
TSAs can lead to decreased appetite, lack of energy, reduced nutrition, and weight loss (Yoshimoto et al., 2020), and they can weaken immunity and slow adaptation to treatment (Kiss et al., 2021). People with cancer are sometimes reluctant to eat food because of TSAs, which may also interfere with social activities related to eating and drinking, disrupt activities of daily living, and eventually lead to decreased quality of life (de Haan et al., 2021). TSAs have effects beyond simple sensory changes, negatively affecting everyday life and causing physical and mental stress (Belqaid et al., 2018). Therefore, it is crucial to understand TSAs related to chemotherapy in women with breast cancer and their effects on quality of life.
Studies have identified factors related to TSAs and the effects of TSAs on quality of life in women with breast cancer undergoing chemotherapy (de Vries et al., 2018; Tugbamenekli & Yildirim, 2022). According to previous studies, there were differences in TSAs based on age (Amézaga et al., 2018), elapsed time after chemotherapy (Belqaid et al., 2014), and chemotherapy agent type (Campagna et al., 2018). For example, individuals receiving taxane-based agents experience more TSAs than those treated with non–taxane-based agents (Amézaga et al., 2018; Campagna et al., 2018). In addition, the taste type most affected by TSAs related to chemotherapy differed from country to country. The salty taste was most affected in Italy (Campagna et al., 2018), and the sour taste was most affected in patients in a study conducted in Sweden (McGreevy et al., 2014). The response to taste and smell differs depending on where an individual is from and the characteristics of the food and living environment (Trachootham et al., 2017). However, few studies regarding TSAs among women with breast cancer in Asia were found. It is necessary to identify taste and smell changes in Korean women with breast cancer to alleviate their TSAs and help with difficulties caused by TSAs.
Accordingly, this study had the following aims: (a) to evaluate the severity of TSAs and the quality of life of Korean women with breast cancer undergoing chemotherapy and (b) to identify the relationship between TSAs and quality of life in Korean women with breast cancer undergoing chemotherapy.
Methods
Design, Setting, and Sample
This study used a cross-sectional design and was conducted online to examine TSAs in women with breast cancer undergoing chemotherapy. Recruitment documents were posted in online communities for women with breast cancer between October and November 2019. After reading these documents, potential participants were given a link to review the research participation guide and explanation. If they were still interested, they were given the option to participate in the study.
The inclusion criteria for this study were as follows: (a) being aged older than 18 years, (b) being diagnosed with breast cancer, and (c) undergoing chemotherapy. The following exclusion criteria were used: (a) being diagnosed with a neurologic disease, (b) being diagnosed with rhinosinusitis, or (c) being diagnosed with any other cancer except breast cancer. Rhinosinusitis can affect olfactory function; one study reported that 30% of patients with rhinosinusitis experienced olfactory disorders (Alt et al., 2014).
The sample size was calculated using G*Power, version 3.1.9. With a significance level of 0.05, a power of 0.9, and an effect size of 0.3 for correlation analysis, a sample of 109 participants was required. To allow for a noncompletion rate of 10%, 130 eligible participants were targeted.
Measurement
TSAs: TSAs were obtained using the Korean translation of the Taste and Smell Survey (TSS) (Heald et al., 1998). The TSS was originally developed for patients with HIV, but it has been widely used in studies of people with cancer (Belqaid et al., 2014; Hutton et al., 2007; McGreevy et al., 2014). This instrument has never been introduced in Korea. After getting approval from the original author, two bilingual people translated and reverse translated the TSS, and the researchers modified it to suit Korean cultural understanding. For example, the original questionnaire was made in the United States and used lemon, vinegar, and crab apples as examples of sour food. However, crab apple is a fruit that is difficult to find in Korea, so it was excluded from the final questionnaire. Next, five experts measured the content validity index; these experts consisted of one professor of nursing and four experts with more than 10 years of nursing experience with patients with cancer. Content validity results showed an item content validity index of 1 and a scale-level content validity index of 0.97.
The TSS consists of 16 questions, divided in the following way: 9 questions about taste alteration, 5 questions about smell alteration, and 2 open-ended questions about personal experiences or feelings and the effect of TSAs on quality of life. The total score ranges from 0 to 16 points. The higher the total score, the greater the severity of TSAs. A score of 0–4 means that the TSAs are insignificant to mild, a score of 5–9 means moderate, and a score of 10–16 means severe.
Quality of life: The Functional Assessment of Cancer Therapy–Breast (FACT-B), which contains questions based on the FACT–General (Cella et al., 1993), was used to measure quality of life (Brady et al., 1997). The Korean version of the FACT-B was used after it was licensed through the developer’s website (www.facit.org).
The FACT-B measures the following five domains of quality of life: physical well-being (seven questions), social/family well-being (seven questions), emotional well-being (six questions), functional well-being (seven questions), and breast cancer–specific areas (nine questions). The FACT-B is a 37-item questionnaire using a five-point Likert-type scale ranging from 0 (not at all) to 4 (very much). Some questions are reverse scored. Higher total scores indicate greater quality of life.
Ethical Consideration
The institutional review board at Ewha Womans University approved the study protocol. All participants received thorough information through an online form and provided consent to participate in the study. Participants were notified that they could withdraw from the study at any time without any consequences, and the collected data were managed anonymously.
Statistical Analysis
Descriptive statistics were used to describe the study sample. Demographic and clinical characteristics, the prevalence of TSAs, and quality of life were presented using means and SDs or numbers and percentages. Statistical analyses were performed using independent t tests, one-way analysis of variance, or Scheffe’s test to assess the relationships between TSAs and demographic or clinical variables. Correlation analyses were used to assess the relationship between TSAs and quality of life. All tests were two-tailed, and the significance value was set at less than 0.05. The analyses were performed using IBM SPSS Statistics, version 26.0.
Results
Study Participants’ Characteristics
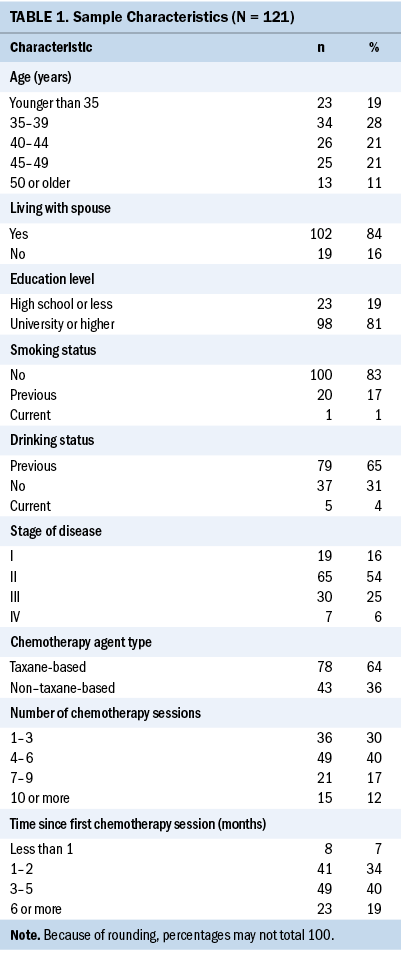
Table 1 presents the sociodemographic and clinical profiles of the study participants (N = 121). More than one-fourth (28%) of participants were aged between 35 and 39 years. More than 80% of participants were living with a spouse, more than half of the participants (54%) had stage II breast cancer, and 64% of the participants were receiving taxane-based chemotherapy.
Prevalence and Characteristics of Self-Reported TSAs
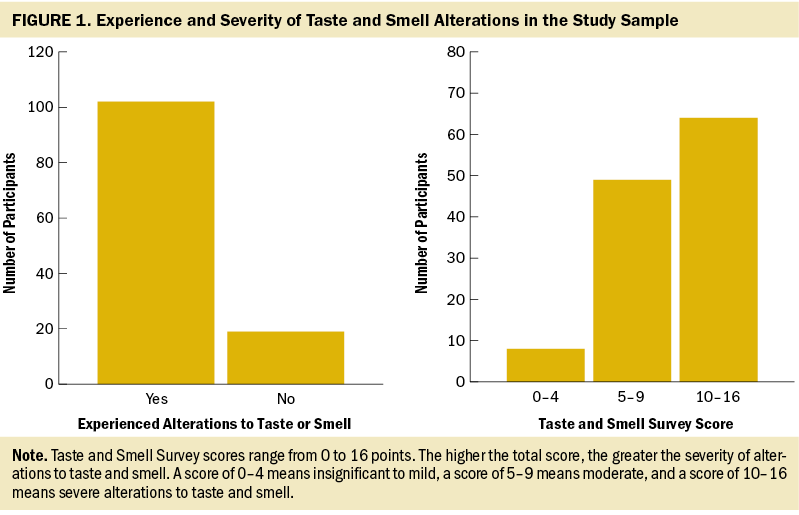
In the current study, 84% (n = 102) of participants experienced TSAs associated with chemotherapy. Of these participants, 51 experienced taste and smell changes. The mean score on the TSS was 13.2 (SD = 2.44) out of a maximum score of 16. Regarding the severity of TSAs, 53% (n = 64) of the participants reported experiencing severe TSAs, 40% (n = 49) reported moderate TSAs, and 7% (n = 8) reported insignificant to mild TSAs (see Figure 1).
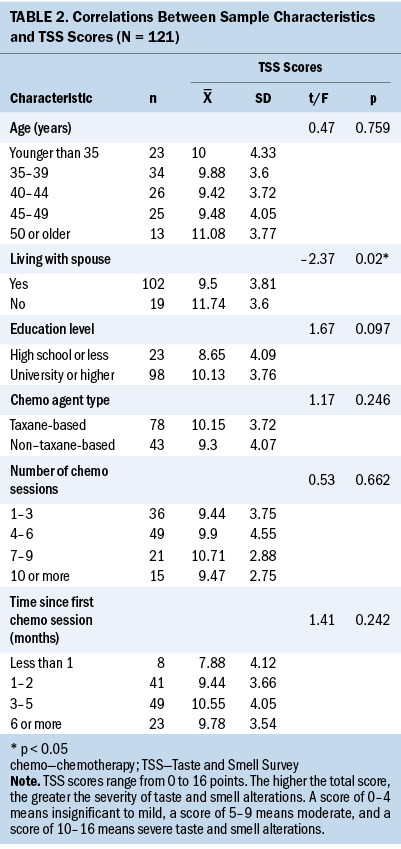
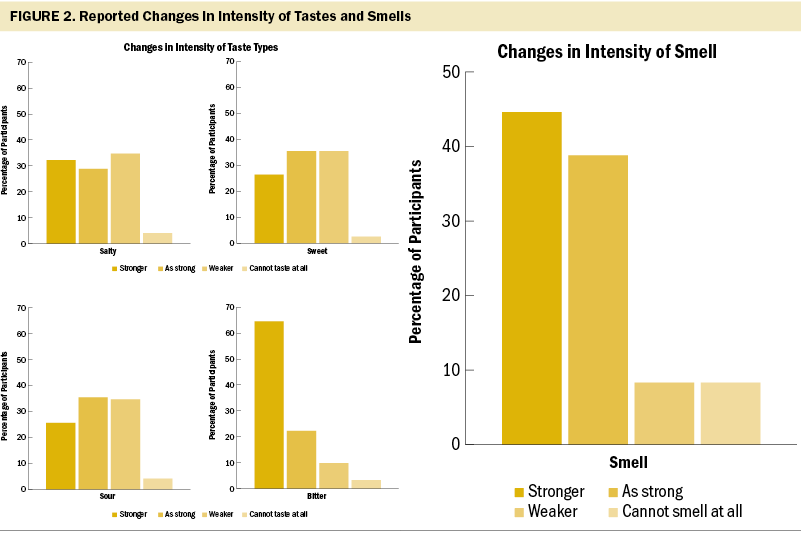
Women living with a spouse had significantly lower scores on the TSS compared to women not living with a spouse (see Table 2). Table 3 presents the descriptive statistics for each item on the TSS. For questions regarding changes in taste, 97 participants (80%) experienced a change in taste. More than half (59%) of the participants reported that they had a long-lasting bad taste in their mouth, and 68% answered that the consistent taste was bitter. Participants felt less intense salty (n = 42, 35%), sweet (n = 43, 36%), and sour (n = 42, 35%) tastes than before they started chemotherapy. On the contrary, 78 participants (64%) felt the bitter taste was stronger than before they started chemotherapy (see Figure 2). In addition, 79 participants (65%) responded “yes” to the question of whether specific drugs interfered with their sense of taste. Of these participants, 29 provided answers that included doxorubicin and cyclophosphamide, which is a non–taxane-based chemotherapy agent, to an open-ended question about the specific drugs that interfered with their sense of taste.
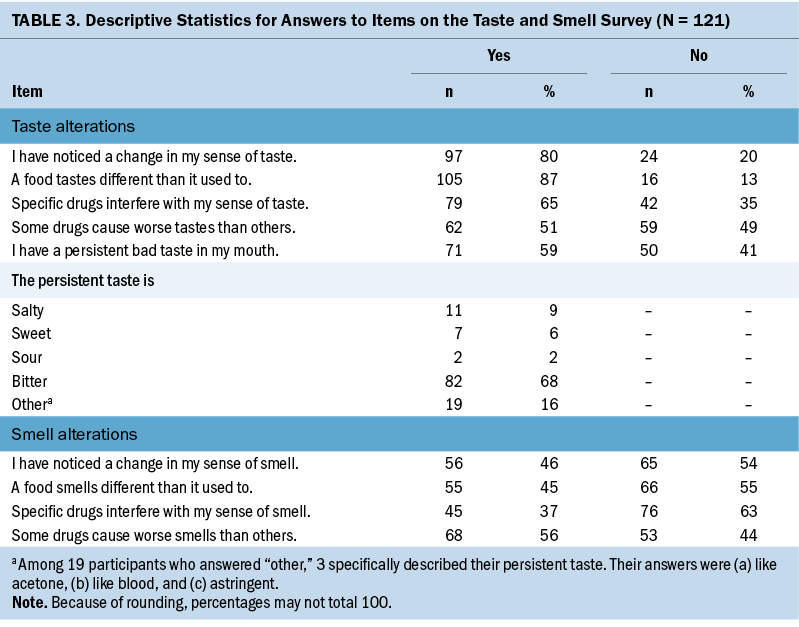
In response to questions about changes to their sense of smell, 56 participants (46%) experienced a change. More than half of the participants (56%) said that some drugs caused things to smell worse than other drugs. The most frequent answer to this question was doxorubicin and cyclophosphamide, as mentioned about drugs interfering with taste. Fewer than half of the participants (45%) said that smells were stronger than before chemotherapy began.
Correlation Between TSAs and Quality of Life
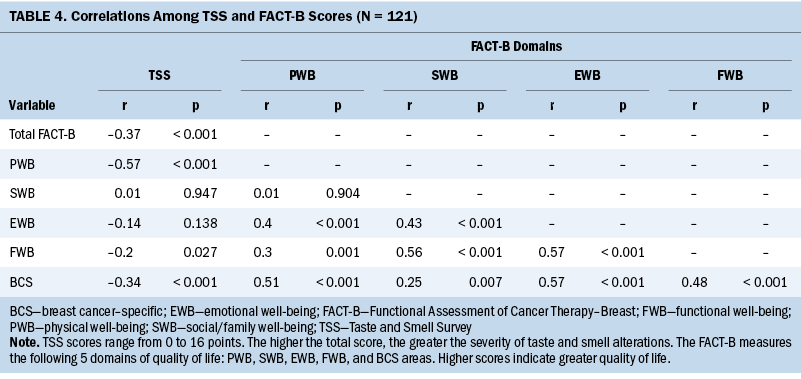
Scores on the TSS were found to have a negative correlation with scores on the FACT-B (r = –0.37, p < 0.001), which means the more severe the TSAs, the lower the quality of life. There was a significant negative correlation between TSAs and the quality-of-life domains of physical well-being (r = –0.37, p < 0.001), functional well-being (r = –0.20, p = 0.027), and breast cancer–related areas (r = –0.34, p < 0.001) (see Table 4), indicating that the more severe the TSAs, the lower the quality of life related to physical well-being, functional well-being, and breast cancer characteristics. Some of the answers from the open-ended questions asking whether TSAs affected participants’ quality of life support these results, particularly regarding physical, social, and emotional well-being. Participants were concerned that treatment and recovery would be delayed because they could not eat. Some participants’ responses included the following statements: “As I lose appetite and can’t eat well, my treatment plan is delayed,” and “I am a slow recoverer because I do not eat well during treatment.” In addition, participants struggled with unpleasant feelings about eating with others and had difficulties during social situations because of TSAs, stating the following: “There is no pleasure to eat, and it is painful to lose taste,” and “When I meet friends to eat out, I can only eat nonstimulating food.”
Discussion
This study was conducted to explore TSAs and their relationship with quality of life among women with breast cancer undergoing chemotherapy in Korea. Compared to previous studies reporting that 59%–65% of women with breast cancer receiving chemotherapy experienced taste alterations (de Vries et al., 2018; Kaizu et al., 2021), the current study showed that 84% of participants experienced taste alterations. In addition, 68% of participants experienced a bitter taste continuously and felt the bitter taste more strongly after they began receiving chemotherapy. These results differ from those of previous studies conducted in European countries in which the most affected taste type was salty (Campagna et al., 2018; Pedersini et al., 2022). Considering that the response to taste varies according to food culture and individuals’ environments (Trachootham et al., 2017), the results of the current study may have been affected by Korean food ingredients or food culture. Therefore, the influences and meaningful cultural factors surrounding TSAs among women with breast cancer receiving chemotherapy should be examined. In addition, when measuring the presence and severity of TSAs, this study did not ask about the timing or triggers of TSAs, including food types. When TSAs occur, such as after food consumption, and whether TSAs are related to participants’ dietary preferences or other possible factors should also be explored. Accordingly, guidelines, educational materials, or educational interventions to minimize or cope with TSAs based on a variety of food cultures need to be developed and distributed.
Because the participants reported that bitterness was the strongest and longest-lasting taste they experienced, it may be helpful to educate patients with breast cancer receiving chemotherapy to avoid bitter foods or foods with strong smells to reduce the severity of TSAs. It may be helpful to develop educational materials about countermeasures based on recommendations from the National Cancer Institute (2021) to alleviate TSAs associated with chemotherapy. In addition, educating women with breast cancer about TSAs and how to cope with them before starting chemotherapy is recommended so they can respond proactively.
The results of the current study found that women living with a spouse report less severe TSAs than women living without a spouse. This may be supported by the fact that spouses are often the primary sources of emotional and functional support for people with cancer (Hasson-Ohayon et al., 2014). Previous studies found that married people were less likely to experience TSAs than unmarried people (Rawal et al., 2016), and that the more supportive care they received, the less likely they were to experience TSAs (Wagland et al., 2016). Therefore, it is possible that women with breast cancer living with their spouses may have had support or other influences that alleviated TSAs. However, living with a spouse does not guarantee that women with breast cancer will receive support from their spouses. Consequently, additional research on spousal support and TSAs may be required.
This study found no significant relationship between chemotherapy agent types and TSAs. Based on the results of previous studies (Amézaga et al., 2018; Campagna et al., 2018), the participants who received taxane-based chemotherapy agents were expected to experience more TSAs. However, that was not the case for the current study. Previous studies that identified TSAs included individuals with a range of cancer types, such as breast, colorectal, and lung (Amézaga et al., 2018; de Haan et al., 2021). However, the current study contained only women with breast cancer, and because of the wide variety of chemotherapy regimens used to treat patients with breast cancer, even those who receive taxane-based chemotherapy may also receive additional treatments like anthracycline and carboplatin at the same time, which may have affected the study’s findings (Amézaga et al., 2018). In addition, even if the same taxane-based agent was administered over the course of a chemotherapy regimen, patients may have experienced different changes to taste and smell at different times depending on the length of time elapsed since the patient had received chemotherapy (IJpma et al., 2017). These complex factors may have influenced the results of the current study. Therefore, additional studies with more individuals undergoing taxane-based chemotherapy are necessary to confirm the association between taxane-based chemotherapy and TSAs in consideration of specific chemotherapy regimens and time elapsed after chemotherapy.
In alignment with previous studies (Campagna et al., 2018; Pang et al., 2019), the current study found that more severe TSAs were associated with lower quality of life. A study by Yoshimoto et al. (2020) reported that people with breast cancer receiving chemotherapy had lower energy intake because of TSAs, which led to malnutrition and worsened cachexia. Another study reported that TSAs made it difficult for women with breast cancer to conduct daily activities and work (Wagland et al., 2016). The current study also reported that lower physical and functional well-being scores on the FACT-B were associated with more severe TSAs. However, additional studies identifying and analyzing which aspects of physical and functional well-being are affected most during active treatment need to be conducted to assist women in recovery from breast cancer.
In the current study, there were no significant correlations between TSAs and social/family well-being or emotional well-being. Previous studies also showed inconsistent results (de Vries et al., 2018; Ponticelli et al., 2017). It may be challenging to accurately compare the results of these studies and the current study because they used different instruments to measure quality of life. In addition, the study by Ponticelli et al. (2017) consisted of individuals with various cancer types in addition to breast cancer. Research is lacking regarding the relationship between chemotherapy-related problems like TSAs and quality of life in the social and emotional domains (Wagland et al., 2016). In a hospital, health professionals may prioritize the physical aspects related to chemotherapy-induced problems and consider the social and emotional domains less important. There may be a perception that emotional and social domains are not a priority for people with cancer (Wagland et al., 2016). However, as can be seen from the results of the open-ended questions, the participants experienced difficulties with social and emotional well-being because of TSAs. Although there were no statistically significant correlations between TSAs and the social/family well-being or emotional well-being quality-of-life domains of the FACT-B, these findings still have clinical significance because participants expressed difficulty in these areas. Therefore, research about the relationship between TSAs and social and emotional quality of life in women with breast cancer receiving chemotherapy should be conducted. This will enable clinicians to improve social and emotional quality of life in women with breast cancer who experience TSAs.
Limitations
The strength of this study is that it explored TSAs related to chemotherapy in Korean women with breast cancer. The results provided a foundation for developing nursing interventions suitable for Korean women with breast cancer undergoing chemotherapy. However, there are several limitations of this study. First, the participants were recruited using convenience sampling, so it is difficult to generalize the results of this study to all women with breast cancer undergoing chemotherapy. Second, the TSS, the tool used to measure the severity of TSAs in this study, has been used many times to measure TSAs outside of Korea, but this study was the first time it was used in Korea. Although it was used after translation, reverse translation, and content validity verification, because the psychometric properties of this tool have not been examined, it is required to be validated. In addition, when evaluating whether it is appropriate for Korean women with breast cancer, the validity must be verified from multiple angles, including specialists in the physiology of TSA as well as experts in nursing and oncology. Third, the association between TSAs and quality of life during chemotherapy could be attributed to other factors that this study did not account for.
Implications for Nursing
Based on the results, some suggestions can be made for oncology nurses and caregivers. Strategies like educating women with breast cancer undergoing chemotherapy to eat fewer bitter foods and foods that do not smell as strongly may help to reduce TSAs. In addition, before starting chemotherapy, efforts should be made to provide individuals with cancer with knowledge about TSAs, including onset and recovery times, for emotional support in preparation for treatment. The risk factors for TSAs should be assessed before starting treatment, and educational materials, including coping measures, need to be developed to use in daily life during the treatment process.

Conclusion
This study is the first to explore TSAs and their relationship with quality of life among women with breast cancer receiving chemotherapy in Korea. More than 80% of the participants experienced TSAs, and many experienced an exceptionally strong bitter taste. Also, results revealed that severe TSAs were closely related to low quality of life during chemotherapy. Regarding quality of life in the domains of social and emotional well-being, additional research should be conducted to understand TSA-related difficulties experienced by women with breast cancer. Based on the results of this study, it is crucial to develop a supportive care plan to manage TSAs in women with breast cancer receiving chemotherapy. Finally, the study results may provide insights about caring for Korean immigrant women or Asian women living in different countries.
About the Authors
Hae Jeong An, MSN, RN, is a PhD candidate and Sook Jung Kang, PhD, FNP-C, is a professor, both in the College of Nursing at Ewha Womans University in Seoul, Korea. No financial relationships to disclose. Hae Jeong An completed the data collection. Both authors contributed to the conceptualization and design, provided statistical support and the analysis, and contributed to the manuscript preparation. Sook Jung Kang can be reached at sookjungkang@ewha.ac.kr, with copy to ONFEditor@ons.org. (Submitted November 2022. Accepted March 16, 2023.)
References
Alt, J.A., Mace, J.C., Buniel, M.C.F., Soler, Z.M., & Smith, T.L. (2014). Predictors of olfactory dysfunction in rhinosinusitis using the Brief Smell Identification Test. Laryngoscope, 124(7), E259–E266. https://doi.org/10.1002/lary.24587
Amézaga, J., Alfaro, B., Ríos, Y., Larraioz, A., Ugartemendia, G., Urruticoechea, A., & Tueros, I. (2018). Assessing taste and smell alterations in cancer patients undergoing chemotherapy according to treatment. Supportive Care in Cancer, 26(12), 4077–4086. https://doi.org/10.1007/s00520-018-4277-z
Belqaid, K., Orrevall, Y., McGreevy, J., Månsson-Brahme, E., Wismer, W., Tishelman, C., & Bernhardson, B.-M. (2014). Self-reported taste and smell alterations in patients under investigation for lung cancer. Acta Oncologica, 53(10), 1405–1412. https://doi.org/10.3109/0284186x.2014.895035
Belqaid, K., Tishelman, C., Orrevall, Y., Månsson-Brahme, E., & Bernhardson, B.-M. (2018). Dealing with taste and smell alterations—A qualitative interview study of people treated for lung cancer. PLOS ONE, 13(1), e0191117. https://doi.org/10.1371/journal.pone.0191117
Brady, M.J., Cella, D.F., Mo, F., Bonomi, A.E., Tulsky, D.S., Lloyd, S.R., . . . Shiomoto, G. (1997). Reliability and validity of the Functional Assessment of Cancer Therapy–Breast quality-of-life instrument. Journal of Clinical Oncology, 15(3), 974–986. https://doi.org/10.1200/JCO.1997.15.3.974
Campagna, S., Gonella, S., Sperlinga, R., Giuliano, P.L., Marchese, R., Pedersini, R., . . . Dimonte, V. (2018). Prevalence, severity, and self-reported characteristics of taste alterations in patients receiving chemotherapy. Oncology Nursing Forum, 45(3), 342–353. https://doi.org/10.1188/18.ONF.342-353
Cella, D.F., Tulsky, D.S., Gray, G., Sarafian, B., Linn, E., Bonomi, A., . . . Brannon, J. (1993). The Functional Assessment of Cancer Therapy scale: Development and validation of the general measure. Journal of Clinical Oncology, 11(3), 570–579. https://doi.org/10.1200/JCO.1993.11.3.570
de Haan, J.J., Renken, R.J., Moshage, Y., Kluifhooft, D.A., Corbier, C., Daly, L.E., . . . Reyners, A.K.L. (2021). Self-reported taste and smell alterations and the liking of oral nutritional supplements with sensory-adapted flavors in cancer patients receiving systemic antitumor treatment. Supportive Care in Cancer, 29(10), 5691–5699. https://doi.org/10.1007/s00520-021-06049-4
de Vries, Y.C., Boesveldt, S., Kelfkens, C.S., Posthuma, E.E., van den Berg, M.M.G.A., de Kruif, J.T.C.M., . . . Winkels, R.M. (2018). Taste and smell perception and quality of life during and after systemic therapy for breast cancer. Breast Cancer Research and Treatment, 170(1), 27–34. https://doi.org/10.1007/s10549-018-4720-3
Donald, M. (2022). A matter of taste: Alteration in patients with cancer. British Journal of Nursing, 31(13), S10–S14. https://doi.org/10.12968/bjon.2022.31.13.S10
Hasson-Ohayon, I., Goldzweig, G., Dorfman, C., & Uziely, B. (2014). Hope and social support utilisation among different age groups of women with breast cancer and their spouses. Psychology and Health, 29(11), 1303–1319. https://doi.org/10.1080/08870446.2014.929686
Heald, A.E., Pieper, C.F., & Schiffman, S.S. (1998). Taste and smell complaints in HIV-infected patients. AIDS, 12(13), 1667–1674. https://doi.org/10.1097/00002030-199813000-00015
Hutton, J.L., Baracos, V.E., & Wismer, W.V. (2007). Chemosensory dysfunction is a primary factor in the evolution of declining nutritional status and quality of life in patients with advanced cancer. Journal of Pain and Symptom Management, 33(2), 156–165. https://doi.org/10.1016/j.jpainsymman.2006.07.017
IJpma, I., Renken, R.J., Gietema, J.A., Slart, R.H.J.A., Mensink, M.G.J., Lefrandt, J.D., . . . Reyners, A.K.L. (2017). Changes in taste and smell function, dietary intake, food preference, and body composition in testicular cancer patients treated with cisplatin-based chemotherapy. Clinical Nutrition, 36(6), 1642–1648. https://doi.org/10.1016/j.clnu.2016.10.013
Kaizu, M., Komatsu, H., Yamauchi, H., Yamauchi, T., Sumitani, M., & Doorenbos, A.Z. (2021). Characteristics of taste alterations in people receiving taxane-based chemotherapy and their association with appetite, weight, and quality of life. Supportive Care in Cancer, 29(9), 5103–5114. https://doi.org/10.1007/s00520-021-06066-3
Kiss, N., Symons, K., Hewitt, J., Davis, H., Ting, C., Lee, A., . . . Tan, S.-Y. (2021). Taste function in adults undergoing cancer radiotherapy or chemotherapy, and implications for nutrition management: A systematic review. Journal of the Academy of Nutrition and Dietetics, 121(2), 278–304. https://doi.org/10.1016/j.jand.2020.08.014
McGreevy, J., Orrevall, Y., Belqaid, K., Wismer, W., Tishelman, C., & Bernhardson, B.-M. (2014). Characteristics of taste and smell alterations reported by patients after starting treatment for lung cancer. Supportive Care in Cancer, 22(10), 2635–2644. https://doi.org/10.1007/s00520-014-2215-2
National Cancer Information Center. (2021). Cancer facts and figures. https://www.cancer.go.kr/lay1/S1T639C641/contents.do
National Cancer Institute. (2021). Mouth and throat problems: Cancer treatment side effects. U.S. Department of Health and Human Services, National Institutes of Health. https://www.cancer.gov/about-cancer/treatment/side-effects/mouth-throat
Pang, D., Qian, L., Chen, Z.J., Lu, Q., Heitkemper, M.M., Yang, P., & Jin, S. (2019). Psychometric properties of the Chinese version of the chemotherapy-induced taste alteration scale. European Journal of Oncology Nursing, 42, 7–13. https://doi.org/10.1016/j.ejon.2019.07.007
Pedersini, R., Zamparini, M., Bosio, S., di Mauro, P., Turla, A., Monteverdi, S., . . . Berruti, A. (2022). Taste alterations during neo/adjuvant chemotherapy and subsequent follow-up in breast cancer patients: A prospective single-center clinical study. Supportive Care in Cancer, 30(8), 6955–6961. https://doi.org/10.1007/s00520-022-07091-6
Ponticelli, E., Clari, M., Frigerio, S., De Clemente, A., Bergese, I., Scavino, E., . . . Sacerdote, C. (2017). Dysgeusia and health-related quality of life of cancer patients receiving chemotherapy: A cross-sectional study. European Journal of Cancer Care, 26(2), e12633. https://doi.org/10.1111/ecc.12633
Rawal, S., Hoffman, H.J., Bainbridge, K.E., Huedo-Medina, T.B., & Duffy, V.B. (2016). Prevalence and risk factors of self-reported smell and taste alterations: Results from the 2011–2012 US National Health and Nutrition Examination Survey (NHANES). Chemical Senses, 41(1), 69–76. https://doi.org/10.1093/chemse/bjv057
Sevryugin, O., Kasvis, P., Vigano, M., & Vigano, A. (2021). Taste and smell disturbances in cancer patients: A scoping review of available treatments. Supportive Care in Cancer, 29(1), 49–66. https://doi.org/10.1007/s00520-020-05609-4
Trachootham, D., Satoh-Kuriwada, S., Lam-Ubol, A., Promkam, C., Chotechuang, N., Sasano, T., & Shoji, N. (2017). Differences in taste perception and spicy preference: A Thai–Japanese cross-cultural study. Chemical Senses, 43(1), 65–74. https://doi.org/10.1093/chemse/bjx071
Tugbamenekli, T., & Yildirim, Y. (2022). Complementary and integrative medicines used by cancer patients to cope with chemotherapy-related taste alterations. Journal of Holistic Nursing and Midwifery, 32(1), 69–77. https://doi.org/10.32598/jhnm.32.1.2217
Wagland, R., Richardson, A., Ewings, S., Armes, J., Lennan, E., Hankins, M., & Griffiths, P. (2016). Prevalence of cancer chemotherapy-related problems, their relation to health-related quality of life and associated supportive care: A cross-sectional survey. Supportive Care in Cancer, 24(12), 4901–4911. https://doi.org/10.1007/s00520-016-3346-4
Yoshimoto, N., Inagaki, M., Sekiguchi, Y., Tomishima, Y., & Masuko, K. (2020). Chemotherapy alters subjective senses of taste and smell but not dietary patterns in Japanese lung cancer patients. Supportive Care in Cancer, 28(4), 1667–1674. https://doi.org/10.1007/s00520-019-04958-z




