Association of Comorbid Diabetes With Clinical Outcomes and Healthcare Utilization in Colorectal Cancer Survivors
Objectives: To compare clinical outcomes and healthcare utilization in colorectal cancer (CRC) survivors with and without diabetes.
Sample & Setting: CRC survivors (N = 3,287) were identified from a statewide electronic health record database using International Classification of Diseases (ICD) codes. Data were extracted on adults aged 21 years or older with an initial diagnosis of stage II or III CRC with diabetes present before CRC diagnosis or no diagnosis of diabetes (control).
Methods & Variables: ICD codes were used to extract diabetes diagnosis and clinical outcome variables. Healthcare utilization was determined by encounter type. Data were analyzed using descriptive statistics, multivariable logistic, and Cox regression.
Results: CRC survivors with diabetes were more likely to develop anemia and infection than CRC survivors without diabetes. In addition, CRC survivors with diabetes were more likely to utilize emergency resources sooner than CRC survivors without diabetes.
Implications for Nursing: Oncology nurses can facilitate the early identification of high-risk survivor groups, reducing negative clinical outcomes and unnecessarily high healthcare resource utilization in CRC survivors with diabetes.
Jump to a section
Colorectal cancer is the third most common cancer diagnosed in men and women in the United States (Giovannucci et al., 2010), and there are about 1.5 million individuals living with colorectal cancer in the United States as of 2021 (American Cancer Society, 2021). Because individuals with colorectal cancer are living longer as a result of advances in screening and treatment, the potential for living with comorbid medical conditions has increased. Type 2 diabetes is a common comorbid condition among individuals with colorectal cancer (Peeters et al., 2015; Tsilidis & Ioannidis, 2015), with a reported 20% of individuals with colorectal cancer having a type 2 diabetes diagnosis (De Bruijn et al., 2013) compared to about 10% of the general population (Centers for Disease Control and Prevention, 2020). The higher prevalence of diabetes among individuals with colorectal cancer may, in turn, contribute to poorer clinical outcomes and increased utilization of healthcare resources.
Individuals with colorectal cancer and diabetes have poorer survival rates (American Cancer Society, 2021; Prieto et al., 2017; Storey et al., 2017; Storey & Von Ah, 2012), increased mortality (Prieto et al., 2017; Tao et al., 2020), and poorer quality of life (Vissers et al., 2013, 2014) than colorectal cancer survivors without diabetes. In general, individuals with colorectal cancer receiving chemotherapy are primarily managed in the outpatient setting. However, some survivors experience poorer clinical outcomes, such as bone marrow suppression (neutropenia, anemia) (Busti et al., 2018; Weycker et al., 2015), infection (Hong et al., 2014), diarrhea (Bultman, 2017; Dávila et al., 2018; González et al., 2017; Meyerhardt et al., 2003; Piper & Saad, 2017), and dehydration (El-Sharkawy et al., 2015), all of which may require the utilization of additional healthcare resources, such as emergency departments and/or inpatient hospitalizations (Brooks et al., 2015; Foltran et al., 2014). Comorbid diabetes in individuals with colorectal cancer may accelerate the onset of these clinical outcomes, increasing the utilization of healthcare resources.
Despite the high prevalence of diabetes among individuals with colorectal cancer, there is a paucity of research on differences in clinical outcomes and utilization of healthcare resources between individuals with colorectal cancer with and without diabetes. However, research suggests that demographic and clinical characteristics may play a role in clinical outcomes and healthcare resource utilization. For example, older age is a risk factor for diabetes (Bigelow & Freeland, 2017; Katz et al., 2013), colorectal cancer (Brown et al., 2020; de Kort et al., 2017), and poor clinical outcomes (Chang et al., 2019). With age, there is a gradual deterioration of the immune system (immunosenescence), which increases vulnerability to myelosuppression and infection (Aiello et al., 2019). Body mass index (BMI) also is a risk factor for diabetes and colorectal cancer (American Cancer Society, 2021; González et al., 2017; Peeters et al., 2015). Research has noted that individuals with a higher BMI have poorer clinical outcomes (Kalb et al., 2019) and utilize more healthcare resources (Nørtoft et al., 2018). Comorbidities also are associated with poorer clinical outcomes and increased demand for healthcare resources (McPhail, 2016; Safarti et al., 2016; Vegda et al., 2009). Gender may also play a role in complications associated with diabetes, with women having an increased likelihood of complications (Huebschmann et al., 2019; Peters & Woodward, 2018). Research also indicates that women utilize more healthcare resources than men (Wang et al., 2019). There is compelling evidence that race and ethnicity (Black and Hispanic) is a risk factor for poorer clinical outcomes and higher utilization of healthcare resources because of delays in seeking treatment (Fiscella & Sanders, 2016; Tawk et al., 2016; Walker et al., 2016). Smoking may also affect clinical outcomes; it has been shown to exacerbate diabetes-related complications (Campagna et al., 2019). A common troublesome symptom among people with diabetes and colorectal cancer is diarrhea (McQuade et al., 2014), which contributes to dehydration, poor clinical outcomes, and increased healthcare utilization. Lastly, cancer stage is important because individuals with diabetes often have more advanced-stage cancers at diagnosis than those without diabetes (Lipscombe et al., 2015). Advanced-stage cancer at diagnosis can lead to poorer clinical outcomes and greater healthcare resource utilization (Yabroff et al., 2013). Although there is ample research showing that these characteristics contribute to poorer outcomes among individuals with diabetes or colorectal cancer, research on the combined effect of having diabetes and colorectal cancer is limited. Therefore, the purpose of this study was to use data from electronic health records to compare individuals with colorectal cancer with and without diabetes in terms of (a) clinical outcomes (anemia, neutropenia, and infection) and (b) utilization of healthcare resources (emergency department services, outpatient services, and inpatient services) while controlling for age, BMI, comorbidities, gender, race, smoking status, diarrhea, dehydration, and cancer stage. The findings from the current study can be used to identify high-risk survivor groups, mitigate negative clinical outcomes by developing targeted interventions, and reduce unnecessarily high healthcare resource utilization.
The conceptual framework for this study was derived from Hammer et al’s. (2019) conceptual model of hyperglycemia in patients with cancer, which describes the physiological linkages between cancer, hyperglycemia (a common characteristic of diabetes), and clinical outcomes. Specifically, this model depicts the intracellular processes initiated by hyperglycemia, which contributes to myelosuppression and, subsequently, an increased risk for infection, organ dysfunction, and death. The deleterious impact of diabetes has been demonstrated in other populations of patients with cancer (Ma et al., 2019; Seymour et al., 2015; Storey et al., 2017, 2019; Storey & Von Ah, 2012). However, this has not been fully examined among individuals with colorectal cancer. For this study, the current authors modified Hammer et al.’s (2019) model to not only include clinical outcomes of myelosuppression and infection but also healthcare resource utilization (emergency department services, inpatient services, and outpatient services) because it is often associated with poor clinical outcomes (Epstein et al., 2020; Erichsen et al., 2013). To better evaluate the contributions of diabetes to clinical outcomes and healthcare resource utilization, the current study focused on individuals with colorectal cancer with and without diabetes, while controlling for known covariates identified in the literature (see Figure 1). 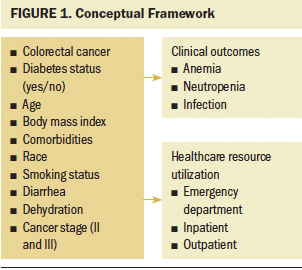
Methods
Design
This is a retrospective cohort study of individuals with colorectal cancer with and without diabetes treated in a large hospital system in the midwestern United States between 2007 and 2017. Clinical data were extracted from a statewide healthcare network, and the state cancer registry was used to determine cancer diagnosis.
Eligibility
Eligible patients were selected from the cohort if they met the following inclusion criteria for the study: (a) having a nonmetastatic diagnosis of colorectal cancer (stage II or III) per International Classification of Diseases (ICD) code; (b) receiving chemotherapy either alone or in combination with other adjuvant therapy (surgery and/or radiation therapy); (c) having a diagnosis of type 2 diabetes prior to the diagnosis of cancer (i.e., colorectal cancer with diabetes) or no diagnosis of diabetes; and (d) being an adult aged 21 years or older (colorectal cancer is rare in children). Exclusion criteria were (a) having another diagnosis of cancer, except basal or squamous skin cancer; (b) having metastatic colorectal cancer; and (c) receiving subsequent chemotherapy after the initial chemotherapy for their cancer diagnosis.
Procedures
A total of 19,032 patients with colorectal cancer were identified based on ICD codes (Centers for Medicare and Medicaid Services, 2020a, 2020b). ICD codes for type 2 diabetes were used to determine those individuals with colorectal cancer and diabetes, along with reviews of medication lists for antidiabetes medications. After applying the inclusion and exclusion criteria, 3,287 individuals with colorectal cancer were included in the study cohort. Demographic and clinical information was extracted from electronic health records to describe the sample. Clinical outcomes, including anemia and neutropenia, were identified based on the ICD codes. Healthcare utilization was extracted from the electronic health record based on documented encounters (visits) to emergency department services, inpatient services, and outpatient services. Accuracy of the data set was verified in two ways. First, the data extraction was repeated, and the number of records and variables was reviewed and compared with the original data set. In addition, a second data analyst verified the data code used to conduct the extraction and create the data set. This study was approved by the Indiana University Institutional Review Board.
Statistical Analysis
Descriptive statistics were used to analyze the demographic and clinical characteristics of the sample. The primary independent variable was diabetes status, categorized as a dichotomous variable (yes or no). All analyses regarding the effect of diabetes on clinical outcomes and healthcare utilization were adjusted for potential confounding covariates, including age, BMI, comorbidity, gender, race, smoking status, diarrhea, dehydration, and cancer stage (II or III). Multivariable logistic regression was performed to evaluate the relationship between diabetes and clinical outcomes (e.g., anemia, neutropenia, infection). Cox regression analysis was used to predict factors associated with time to utilization of emergency department services, inpatient services, or outpatient services. All analyses were performed using SAS, version 9.4.
Results
Patient Characteristics
Table 1 shows the characteristics of the colorectal cancer cohort (N = 3,287). The mean age was 59.86 years. Most were male (53%) and White (92%). Only 4% reported being a former smoker, whereas 6% reported being a current smoker. A little less than one-third had stage II cancer, and more than two-thirds had stage III cancer. Approximately 20% of patients also had type 2 diabetes. In this sample, individuals with colorectal cancer and diabetes were older, had a higher BMI, and had more comorbidities than individuals with colorectal cancer without diabetes. 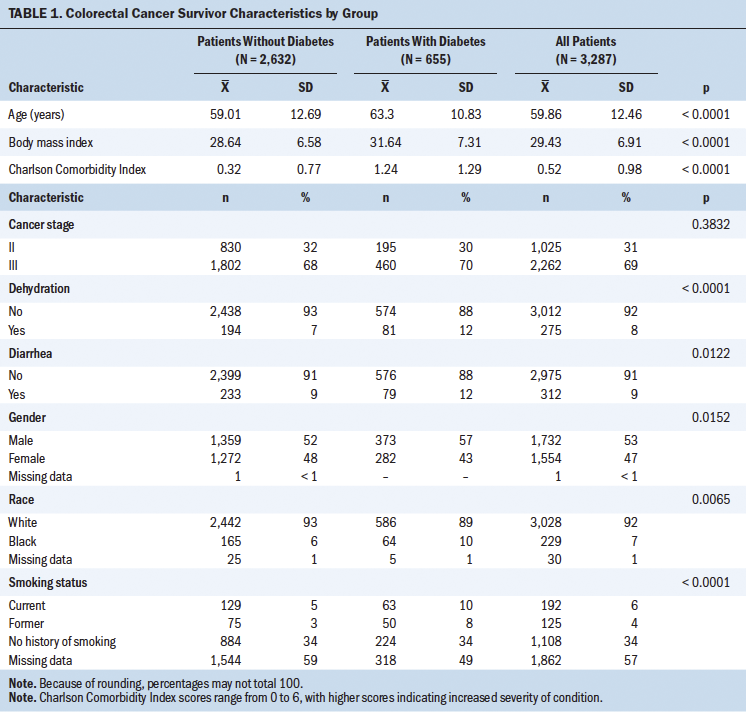
Clinical Outcomes
Anemia: Table 2 shows the results of multivariable logistic regression analyses. A diagnosis of diabetes increased the odds of anemia in individuals with colorectal cancer by 1.45 (95% confidence interval [CI] [1.04, 2.01]), when controlling for age, gender, race, smoking, diarrhea, and dehydration. Smoking status also was a predictor of anemia. Being a former smoker (versus a nonsmoker) was associated with higher odds (odds ratio [OR] = 1.78; 95% CI [1.10, 2.88]) of anemia. In addition, the presence of diarrhea (OR = 2.39; 95% CI [1.59, 3.59]) and dehydration (OR = 3.91; 95% CI [2.62, 5.85]) predicted anemia. 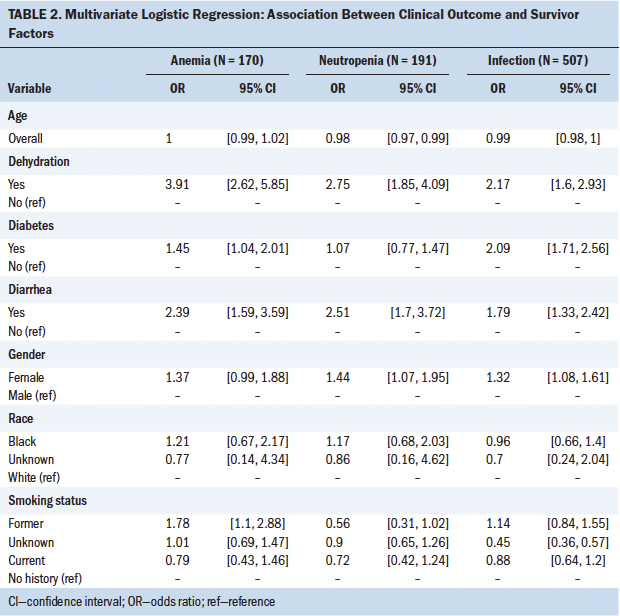
Neutropenia: In this sample of individuals with colorectal cancer, diabetes was not associated with an increased risk of neutropenia. However, being a woman with colorectal cancer increased the odds for neutropenia (OR = 1.44; 95% CI [1.07, 1.95]). Having diarrhea (OR = 2.51; 95% CI [1.7, 3.72]) and dehydration (OR = 2.75; 95% CI [1.85, 4.09]) also were associated with higher odds of having neutropenia.
Infection: Individuals with colorectal cancer and diabetes were more likely to develop an infection than individuals with colorectal cancer without diabetes (OR = 2.09; 95% CI [1.71, 2.56]). Women with colorectal cancer were more likely to develop an infection (OR = 1.32; 95% CI [1.08, 1.61]) than their male counterparts. Having diarrhea (OR = 1.79; 95% CI [1.33, 2.42]) and dehydration (OR = 2.17; 95% CI [1.6, 2.93]) also increased the odds of an infection.
Healthcare Utilization
Table 3 shows the results of the Cox regression model. Individuals with colorectal cancer and diabetes were more likely to utilize emergency department resources (hazard ratio [HR] = 1.11; 95% CI [1.01, 1.21]) compared to individuals with colorectal cancer without diabetes. Diarrhea also was associated with shorter time to emergency department utilization (HR = 1.62, 95% CI [1.4, 1.88]) and higher inpatient visit utilization (HR = 1.27; 95% CI [1.14, 1.41]). Lastly, dehydration (HR = 1.47; 95% CI [1.26, 1.71]) was associated with shorter time to emergency department utilization. 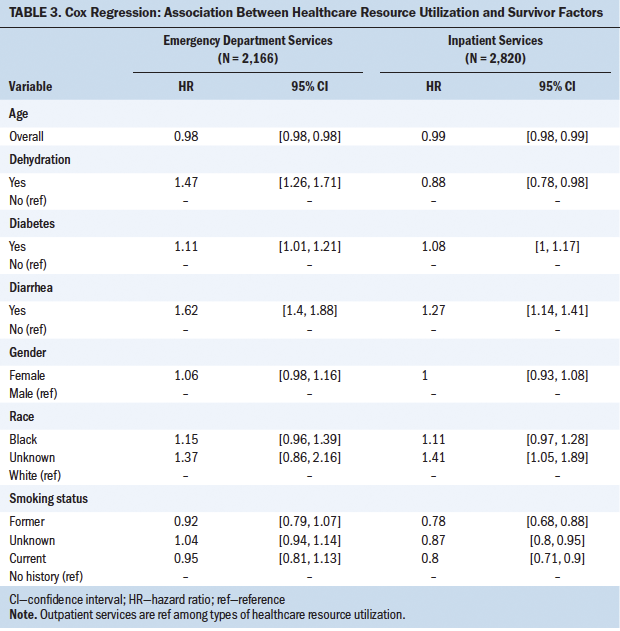
Discussion
This study comprehensively examines clinical outcomes and healthcare utilization of individuals with colorectal cancer with and without diabetes. Using a large data set, a cohort of individuals with colorectal cancer was identified; about 20% of individuals within the cohort had diabetes, which is comparable to other studies showing the prevalence of diabetes among individuals with colorectal cancer to be as high as 24% (Prieto et al., 2017; Vissers et al., 2013, 2014). Importantly, the current study found that individuals with colorectal cancer and diabetes were more likely to have anemia and infection and utilize emergency department resources.
Clinical Outcomes
Results indicate that individuals with colorectal cancer and diabetes were more likely to have anemia than those without diabetes. Anemia is common in individuals with colorectal cancer (Khanbhai et al., 2014; Ludwig et al., 2004), with about 40% experiencing anemia as a result of cancer or its treatment (Busti et al., 2018). Colorectal cancer and diabetes are both inflammatory disease processes (Andrews & Arredondo, 2012), which may lead to epigenetic changes that alter the structure and function of bone marrow (Gallagher et al., 2015; Keating & El-Osta, 2013; Maiuri & O’Hagan, 2016; Parcesepe et al., 2016; Shurin, 2012; Tsilidis & Ioannidis, 2015). Therefore, it is plausible that the combination of these co-occurring inflammatory conditions (colorectal cancer and diabetes) may cause more profound bone marrow suppression, resulting in an increased risk of anemia among individuals with colorectal cancer and diabetes. More research is needed to understand how comorbid diabetes affects the bone marrow of individuals with colorectal cancer.
The current authors also found that a history of smoking was associated with anemia. Smoking stimulates inflammation (Levitzky et al., 2008) and has adverse effects on the hematologic system (Malenica et al., 2017). The current study found anemia to be greater among former smokers compared to current smokers. In this sample, it was not possible to determine the length of time since smoking cessation had occurred. However, the long-term effects of smoking on red blood cells may persist well beyond cessation (Pedersen et al., 2019), remaining a contributing factor for anemia.
Diarrhea and dehydration also were associated with anemia in this sample. Cancer therapies can cause epigenetic changes in the gastrointestinal tract and to the structure and functions of the colon, resulting in diarrhea (Bultman, 2017; Dávila et al., 2018; González et al., 2017; Meyerhardt et al., 2003; Piper & Saad, 2017). Changes in the lining of the gastrointestinal tract can cause a disturbance in iron metabolism (Qujeg et al., 2011), which may be a contributing factor to the risk of anemia. In addition, it is possible that severe diarrhea can lead to intermittent blood loss and exacerbation of anemia. Erythrocytes (red blood cells) require cellular hydration to function and survive (Gallagher, 2017). Dehydration can cause perturbations in the intracellular water of erythrocytes, further exacerbating anemia (Gallagher, 2017). The relationship of diarrhea and dehydration with anemia bears further investigation.
In the current study, an association was not found between diabetes and neutropenia. Neutropenia is associated with cancer and cancer therapies, and it is estimated to occur in about 11% of individuals with colorectal cancer (Weycker et al., 2015). In patients with diabetes, a hyperglycemic state decreases the mobilization, chemotaxis, and phagocytic activity of neutrophils (Peleg et al., 2007; Vardakas et al., 2007). The role of diabetes in neutropenia in individuals with colorectal cancer has not been fully explored. Only one systematic review (Alenzi & Kelley, 2017) has examined the impact of diabetes and hyperglycemia on neutropenia in other solid tumor (breast, lung, and ovarian) populations. Alenzi and Kelley (2017) noted that cancer survivors with hyperglycemia and diabetes had increased odds of chemotherapy-induced neutropenia. It is possible that the current authors did not find an association between diabetes and neutropenia in individuals with colorectal cancer because those with diabetes were well controlled or, more likely, there were individuals with colorectal cancer with prediabetes in the control, which may have influenced findings. In addition, neutropenia was identified using ICD codes rather than laboratory values, which may have precluded some individuals with colorectal cancer and neutropenia. More research is needed to examine the impact of hyperglycemia and diabetes on neutropenia among individuals with colorectal cancer.
The current authors found that being female and having diarrhea and dehydration increased the odds of experiencing neutropenia. The increased risk of neutropenia among women with colorectal cancer may be related to the fact that right-sided colon cancer, which is more common among women, has been associated with increased leukopenia, including neutropenia (Chansky et al., 2005; Katz et al., 2013; Kim et al., 2015, 2018). In addition, right-sided colorectal cancer is associated with increased diarrhea (Kim et al., 2018), which can result in dehydration. Studies assessing the impact of gender differences in clinical outcomes among individuals with colorectal cancer are important for understanding the biological and sociocultural risks and responses to colorectal cancer treatment.
The current authors found that individuals with colorectal cancer and diabetes were more likely to develop an infection. Other researchers also have found that individuals with colorectal cancer and diabetes had a higher risk for infection-related adverse events, per the Common Terminology Criteria for Adverse Events (Hong et al., 2014). The hyperglycemic environment caused by diabetes contributes to increased virulence of some pathogenic organisms, increasing susceptibility to infection (Casqueiro et al., 2012).
In the current study, women with colorectal cancer were more likely than their male counterparts to have infections. Because women are at a higher risk for leukopenia and neutropenia (Chansky et al., 2005; Katz et al., 2013; Kim et al., 2015, 2018), the resultant compromised immune status may place them at increased risk for developing infections. Prospective studies examining the role of gender in individuals with colorectal cancer are warranted.
The current authors found that diarrhea and dehydration were associated with infection. Colorectal cancer and its treatment can cause diarrhea. Changes in the intestinal milieu co-occurring with immunosuppression can increase the vulnerability of colorectal cancer survivors to life-threatening infections (Bossi et al., 2018). Dehydration is often associated with diarrhea in individuals with colorectal cancer, particularly during cancer treatment. Dehydration also is associated with infection, including urinary tract infection (El-Sharkawy et al., 2015).
Healthcare Utilization
The current study demonstrated that individuals with colorectal cancer and diabetes utilized healthcare resources more than individuals with colorectal cancer without diabetes. Specifically, the authors found that individuals with colorectal cancer and diabetes utilized emergency resources more than individuals with colorectal cancer without diabetes. In a study of breast and colorectal cancer survivors by Yao et al. (2015), those with diabetes received poorer diabetes management after their cancer diagnosis than before their cancer diagnosis. Healthcare providers are focused on curative intent and may not focus on the management of diabetes (Yao et al., 2015). In addition, individuals with cancer and diabetes often prioritize self-management of treatment side effects over self-management of diabetes (Hershey et al., 2012). It is plausible that poor diabetes management among individuals with colorectal cancer could increase toxicity, side effects, or acute symptoms, leading to greater utilization of emergency department services. Additional studies on the challenges of self-management among individuals with colorectal cancer and diabetes are needed to develop interventions specific to this high-risk subgroup of cancer survivors.
Although the current authors could not directly determine if emergency visits of individuals with colorectal cancer with diabetes were related to either colorectal cancer or diabetes, findings from this study showed that individuals with co-occurring conditions were more likely to utilize these resources. Independently, both cancer and diabetes place a substantial financial burden on society (Agency for Healthcare Research and Quality, n.d.; American Diabetes Association, n.d.). However, research describing the impact of diabetes in addition to colorectal cancer on the utilization and subsequent costs of healthcare services is lacking. More research is needed to determine the cost of healthcare utilization and the personal financial burden for individuals with colorectal cancer and diabetes.
Lastly, data from the electronic health records of individuals with colorectal cancer were used to expand on the original conceptual model of hyperglycemia in a patient with cancer (Hammer et al., 2019). Information was added related to the impact of diabetes on myelosuppression (anemia), and the model was expanded to include other clinical outcomes. Findings of the current study support this revised model depicting the deleterious role of diabetes on infection, myelosuppression (anemia), and healthcare utilization, all of which are important to identify, given that cancer survivors are living longer with comorbid conditions that can greatly affect resources.
Strengths and Limitations
This study filled a gap in knowledge about clinical outcomes and healthcare utilization among individuals with colorectal cancer and diabetes. It included a large sample size with a focus on a specific common cancer (colorectal cancer) and comorbid diabetes. In addition, it used data from electronic health records to examine the influence of comorbid diabetes on clinical outcomes and healthcare resource utilization. Big data from electronic health records provide a rich source of information for biomedical research (Duan et al., 2014), allowing researchers to capture real-world data that otherwise would be costly and difficult to acquire in a longitudinal trial. This study provides a foundation for the development of prospective studies on clinical outcomes and healthcare utilization of individuals with colorectal cancer with type 2 diabetes.
These findings should be considered in light of several limitations. Although the current study provides preliminary data that are useful for guiding the development of future prospective studies, the descriptive, retrospective study design precludes the ability to establish causality. The goal of this study was to compare clinical outcomes and healthcare utilization among individuals with colorectal cancer with and without diabetes. Therefore, the authors did not examine the role of other comorbidities on these outcomes, which may have affected the findings. More research is needed to understand the influence of comorbidities on these and other outcomes of cancer survivors. Lastly, it is possible that clinicians may have incorrectly assigned or omitted some health data and ICD codes in the electronic health record. However, to mitigate this risk, the diagnosis of type 2 diabetes was verified by a review of medication lists. Given that the prevalence of diabetes is increasing (Rowley et al., 2017), prospective studies exploring the differences in clinical outcomes and healthcare resource utilization among other cancer survivor groups with diabetes are important.
Implications for Nursing
The current study supports prior studies that showed poorer clinical outcomes for individuals with colorectal cancer and diabetes while also adding important information about healthcare utilization. Oncology nurses’ understanding of the contribution of comorbid diabetes to poor outcomes in individuals with colorectal cancer can inform the development of proactive assessment, intervention, monitoring, and treatment strategies, improving clinical outcomes and reducing the utilization of healthcare resources. The current study identified important outcomes for future interventional research. Such research should apply these findings to develop evidence-based interventions that target clinical outcomes and healthcare resource utilization among high-risk survivor groups. Lastly, future research should look specifically at hyperglycemia and outcomes, as suggested in the model. 
Conclusion
Individuals with colorectal cancer and diabetes experienced more anemia, infection, and emergency department visits than survivors without diabetes. The best approach for the clinical management of comorbid diabetes in individuals with colorectal cancer remains unclear. Most clinical practice guidelines focus on a single disease, limiting guidance for clinicians in the treatment of cancer survivors with diabetes (Deshields et al., 2013; Lee et al., 2017; Storey et al., 2017), while also possibly placing this subset of individuals with colorectal cancer at risk for poorer clinical outcomes and higher utilization of healthcare resources. Because cancer survivors are living longer, it is imperative to understand the ramifications that comorbid conditions have on the survivorship experience. More studies are needed to explore the impact of comorbid conditions on clinical outcomes and healthcare utilization in individuals with colorectal cancer.
About the Author(s)
Susan Storey, PhD, RN, AOCNS®, is an assistant professor in the School of Nursing, and Zuoyi Zhang, PhD, is an assistant research professor in the School of Medicine, both at Indiana University; Xiao Luo, PhD, is an assistant professor in the Purdue School of Engineering and Technology at Indiana University—Purdue University Indianapolis; Diane Von Ah, PhD, RN, FAAN, is a professor in the School of Nursing at Indiana University; Megan Metzger, MS, is a project manager at the Regenstrief Institute; Jie Zhang, PhD, is an assistant professor in the School of Medicine, and Amrutha Ravali Jakka, PharmD, is a research assistant in the School of Nursing, both at Indiana University; and Kun Huang, PhD, MS, FAIMBE, is a professor in the Department of Medicine, the Indiana University School of Medicine Precision Health Initiative Chair for Genomic Data Science, the director of data science and informatics in the Precision Health Initiative, the assistant dean for data science in the School of Medicine, and the associate director of data science at the Simon Comprehensive Cancer Center, all at Indiana University, and an investigator at the Regenstrief Institute, all in Indianapolis, IN. The study was funded, in part, by a research investment fund from the Indiana University School of Nursing (principal investigator [PI]: Storey), an RE01 grant from the Oncology Nursing Foundation (PI: Storey), and a pilot grant from Indiana University Simon Cancer Center’s Cancer Prevention and Control program (PI: Storey). Storey, Z. Zhang, Von Ah, Jakka, and Huang contributed to the conceptualization and design. Storey, Z. Zhang, Luo, Jakka, and Huang completed the data collection. Storey, Z. Zhang, and Von Ah provided statistical support. Storey, Z. Zhang, J. Zhang, and Huang provided the analysis. All authors contributed to the manuscript preparation. Storey can be reached at sustorey@iu.edu, with copy to ONFEditor@ons.org. (Submitted June 2020. Accepted October 10, 2020.)




