Nutritional Screening: Development and Implementation of a Protocol in Patients With Head and Neck Cancer
Background: Half of all patients with head and neck cancer are malnourished at the time of diagnosis. Although nutritional status can be predictive of patient-related outcomes, nutritional screening protocols have not been widely accepted as standard of care.
Objectives: This article aims to develop and pilot a nutritional screening protocol in an outpatient head and neck clinic using the abridged version of the Patient Generated Subjective Global Assessment (abPG-SGA) so that at-risk patients are identified and can maintain or minimize weight loss and body mass index (BMI).
Methods: At initial and subsequent visits, study participants completed the abPG-SGA, documenting percentage of weight loss and BMI from baseline.
Findings: 317 patients completed the abPG-SGA, with 119 scoring 6 or more, prompting a dietitian referral. The nutritional screening protocol accurately identified at-risk patients and resulted in less weight loss and BMI change.
Jump to a section
About 50%–80% of patients with head and neck cancer (HNC) are malnourished at the time of diagnosis (Gorenc et al., 2015; Müller-Richter et al., 2017). Malnutrition has a multifactorial pathogenesis in this population because risky behaviors and unhealthy lifestyle choices contribute to poor dietary consumption. The anatomic site of the tumor and associated treatments can further compound nutritional deficits because of mechanical obstruction, dysphagia, odynophagia, anorexia, and fatigue (Paccagnella et al., 2010). Patients with HNC can expect to have a 20% decrease in total body weight during the treatment and post-treatment phases (Müller-Richter et al., 2017). Weight loss of greater than 5%–10% within a six-month time frame and/or a body mass index (BMI) of less than 20 kg/m2 can be indicative of malnutrition, placing the patient at a higher risk for poor outcomes (Müller-Richter et al., 2017).
Malnutrition has been established as a prognostic indicator for cancer-related morbidity and mortality, adversely affecting clinical, functional, and economic outcomes (Lim et al., 2012). Prevost et al. (2014) suggest that nutritional status can be predictive of patient outcomes and correlated with a patient’s ability to tolerate curative treatment. There are clear correlations between malnutrition and increased length of stay, diminished therapeutic response to treatment, increased complication rates, and elevated healthcare costs (Álvaro Sanz et al., 2019). Severe malnutrition has been associated with a decrease in quality-of-life indicators and overall lower survival rates when compared to well-nourished patients (Álvaro Sanz et al., 2019; Datema et al., 2011; Paccagnella et al., 2010).
A positive correlation exists between nutritional status and improved quality of life, recognizing the importance of frequent nutritional assessment in the management of patients with HNC (Lis et al., 2012). Early and frequent nutritional assessment and intervention are correlated with improved treatment tolerance, decreased interruptions in treatment, and decreased hospitalizations (Paccagnella et al., 2010). To improve patient-centered outcomes, guidelines recommend malnutrition screening and treatment at the time of diagnosis, with assessment continuing until cancer treatment ends (Alshadwi et al., 2013).
Factors and tools to identify malnutrition in patients with HNC include weight loss alone or in combination with the Patient-Generated Subjective Global Assessment (PG-SGA), weight index, BMI, and/or serum albumin (Lis et al., 2012). The PG-SGA is considered the gold standard in nutritional assessment and identification of malnutrition in patients with cancer. However, it is lengthy, requiring extensive time investment from patients and healthcare providers, and it is often met with challenges. Gabrielson et al. (2013) validated an abridged version of the PG-SGA (abPG-SGA) in patients receiving chemotherapy in an outpatient clinic. The abPG-SGA demonstrated a 94% sensitivity and 78% specificity, confirming its validity for identifying malnutrition in this setting (Gabrielson et al., 2013). The abPG-SGA forgoes the physical examination associated with the PG-SGA, instead requiring patients only to complete questions associated with weight, nutrition, symptoms, and activities of daily living, which improves the likelihood of successful implementation (Gabrielson et al., 2013).
Despite National Comprehensive Cancer Network guideline recommendations for serial monitoring of nutritional status for all patients diagnosed with HNC and subsequent evaluation and treatment by a dietitian for at-risk patients, there is not a standardized protocol for nutritional screening (Gill et al., 2018). In addition, from a global perspective, a nutritional screening protocol has not been widely accepted as standard of care for patients with HNC (Álvaro Sanz et al., 2019). The goal of this quality improvement project was to pilot a nutritional screening protocol in an outpatient head and neck clinic, using the abPG-SGA to identify at-risk patients and initiate interventions to maintain weight or minimize weight loss in an effort to optimize clinical and quality-of-life outcomes.
Methods
Objective measures included percentage of weight loss since the initial appointment because longitudinal weight surveillance is the standard of care (Lis et al., 2012) and is the most sensitive outcome marker (Prevost et al., 2014). BMI was also collected because it is a useful measure for determining long-term nutritional status (Alshadwi et al., 2013). Adherence to the nutritional screening protocol was tracked using the number of patients scheduled with the provider minus patients who declined to participate or those who were excluded as compared to the number of completed screening forms. The percentage of patients scoring 6 or more on the abPG-SGA was also tracked and compared to the number of referrals made to the dietitian to determine adherence to the nutritional screening protocol.
The Iowa Model of Evidence-Based Practice to Promote Quality Care was used as the theoretical framework for implementing, maintaining, and evaluating an evidence-based nutritional screening protocol to improve patient outcomes (i.e., weight and BMI). Data were collected via retrospective chart audit to ascertain preimplementation weight loss and BMI change.
This project was implemented in an outpatient head and neck clinic, the Wellin Head and Neck Tumor Center at the Medical University of South Carolina in Charleston. Adult patients aged 18 years or older with a diagnosis of a malignant HNC were offered participation. The project was reviewed and approved by the institutional review board of the Medical University of South Carolina and received exempt status.
Instrument
The abPG-SGA is a five-item self-report survey (see Figure 1). Responses to the survey are assigned a value based on the answer provided, with a possible total score of 47. Scores of 2–5 warrant education, oral supplementation as prescribed by the healthcare provider, and close monitoring. Scores of 6 or more suggest patients who are at risk for or have malnutrition and warrant a referral to a dietitian for further assessment and intervention. Typical interventions include nutritional counseling, oral supplementation with vitamins and/or oral protein supplementation, modification of diets because of mechanical or swallowing limitations, and enteral nutrition. Interventions were at the discretion of the dietitian and not recorded for the quality improvement project. 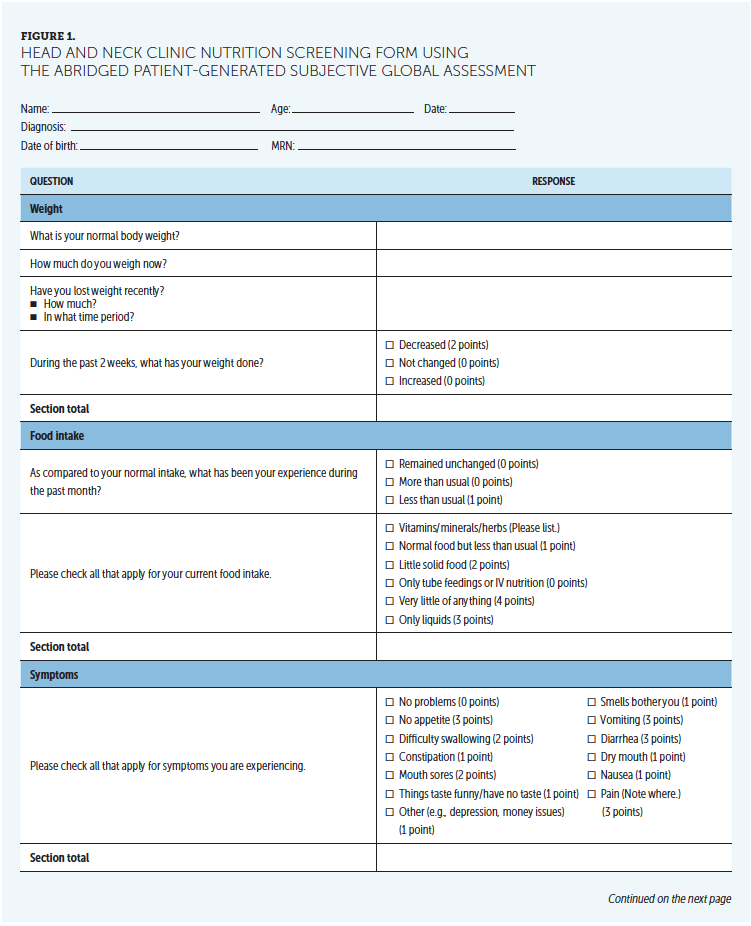
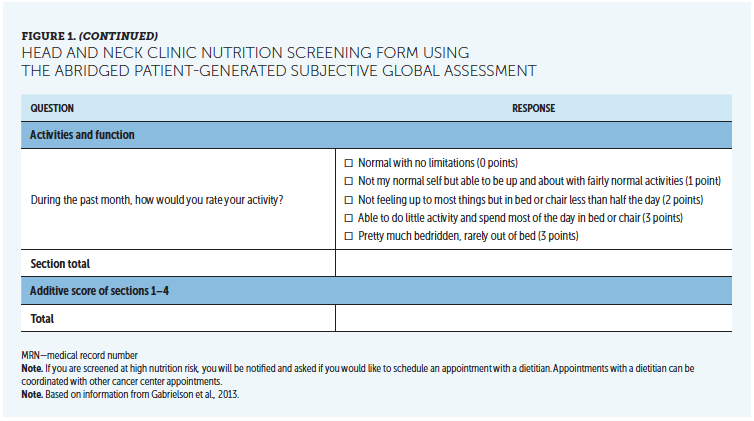
Implementation
Participants were asked to complete the abPG-SGA at the initial visit and all subsequent visits. The abPG-SGA was distributed during the check-in process, allowing participants to complete the survey as they waited for their scheduled appointment. On average, it took participants less than five minutes to complete the survey.
The clinic nurse or the certified medical assistant scored the abPG-SGA during triage or after the participant was placed in a room. The scoring of the tool took less than two minutes and did not interrupt normal clinic flow. All patients received generalized verbal education about the importance of nutrition.
After the abPG-SGA was reviewed by the advanced practice provider (APP), the nutritional protocol was consulted, and appropriate interventions were offered to the patient based on provider discretion. For the purpose of this quality improvement project, prescribing methods for oral supplementation and specific interventions prescribed were not tracked. Patients scoring 6 or higher were given information regarding the role of the dietitian in managing malnutrition. The APP then initiated a referral to the dietitian for further assessment, nutritional counseling, or patient-centered interventions. Referrals to interprofessional team members is a normal task completed during checkout; therefore, referrals to the dietitian did not interfere with normal workflow.
All participants were offered an opportunity to complete the abPG-SGA at subsequent appointments for the duration of the quality improvement project. At any time during the implementation phase, if the patient scored 6 or more, the APP initiated a referral if not previously completed. Patients were scheduled for routine appointments with the dietitian until deemed fit by the dietitian or until the patient declined the need for further treatment.
Data Analysis
For each participant, age, gender, race, diagnosis, stage, weight, BMI, abPG-SGA score, and referral to the dietitian were collected for a six-month period from January through June 2019. All pre- and postintervention data were deidentified, collected via chart review, and entered into a password-protected Microsoft® Excel spreadsheet for further analysis. Descriptive statistics were used to analyze all demographic variables and outcome measures.
Results
Data were collected from 317 patient encounters. Participants ranged in age from 18–92 years, with a mean age of 65 years (see Table 1). The majority of participants were men (n = 221). Participants were diagnosed with cancers of the oral cavity, oropharynx, pharynx, hypopharynx, larynx, parotid, sinus, skin, ear, skull base, and unknown primary. Participants had varying stages of disease and were in different phases of treatment, ranging from newly diagnosed to no evidence of disease with routine surveillance. Two participants died during the course of the project. Participants with a benign diagnosis were excluded from the project. Participants who did not receive a follow-up visit during the quality improvement project were also excluded from the final analysis. 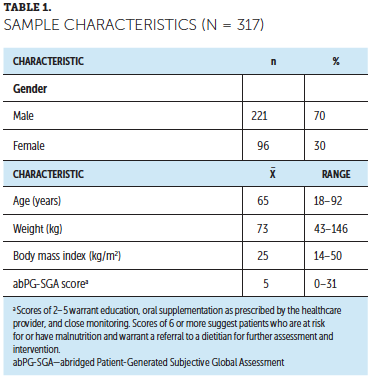
During the six-month period, 584 patient encounters were scheduled with a head and neck surgeon. The adherence rate for initiating the nutritional screening protocol was 72%, with 420 of the 584 scheduled patients completing the abPG-SGA. Ninety-six percent (405 of 420) of participants screened were accurately identified to be at risk for malnutrition using the abPG-SGA based on a BMI of less than 20 kg/m2. Ultimately, 25% (103 of 420) of the patient encounters were excluded from the project because of a benign diagnosis or lack of follow-up during the six-month time frame. Thirty-eight percent (119 of 317) of participants screened were at nutritional risk, scoring 6 or more on the abPG-SGA. These participants qualified for a consultation with the dietitian for further nutritional assessment and monitored for change in weight and BMI.
The adherence rate for referring participants for nutritional consultation was 69% (82 of 119). Thirty-three percent (39 of 119) of at-risk patients declined the consultation, 31% (37 of 119) were not referred for various reasons, and 36% (43 of 119) received consultations before or after completing the abPG-SGA and were referred to the dietitian. Twenty of 43 patients received an outpatient consultation with the dietitian, and 19 patients received an inpatient consultation.
The mean weight for the preimplementation cohort was 79 kg as compared to 73 kg in the postimplementation cohort. The mean BMI was 26 kg/m2 and 25 kg/m2, respectively. However, the mean weight loss since the initial appointment was –4.7 kg in the preimplementation cohort versus a 1.2 kg weight gain in the postimplementation cohort. The BMI change since the initial appointment was –1.3 kg/m2 in the preimplementation cohort versus –0.8 kg/m2 in the postimplementation cohort.
At-risk participants (scoring 6 or more) who refused a nutritional consultation experienced, on average, twice as much weight loss (–1.2 kg in the consultation cohort versus –2.1 kg in the cohort who refused the consultation) than at-risk patients who consented to a nutritional consultation. BMI remained relatively unchanged (–0.5 kg/m2 in the consultation cohort versus –0.7 kg/m2 in the cohort who refused the consultation).
Discussion
This multiple-phase framework incorporated feedback loops that allowed for continual modifications of the evidence-based nutritional screening protocol to ensure successful implementation across the interprofessional team (Melnyk & Fineout-Overholt, 2015).
It is important to consider the multifactorial implications of weight loss for patients diagnosed with HNC and the effects of incorporating a nutritional screening protocol into everyday practice. The findings from this project highlight the importance of recognizing nutrition as an integral component in the management of patients with HNC (Talwar et al., 2016). This study’s results further support findings from prior research that early and frequent nutritional interventions can potentially minimize weight loss and improve outcomes. The literature suggests that the appropriate diagnosis and treatment of malnutrition has been positively correlated with improved tolerance to cancer treatment and improved quality of life (Paccagnella et al., 2010).
These findings also contribute to the continued validation of the abPG-SGA, a self-reported nutritional screening tool, and its ability to accurately identify patients who are at risk for malnutrition (Gabrielson et al., 2013; Shahvazi et al., 2017). In addition, the results further support the plausibility of instituting a standardized evidence-based nutritional screening protocol in a fast-paced outpatient HNC clinic. The nutritional protocol was seamlessly integrated into the workflow with minimal time requirements and little to no financial investment, which contributed to leadership and staff buy-in. These findings continue to build a strong case for making routine nutritional screening the standard of care for patients with cancer (Reber et al., 2019).
In addition, outcomes from this project highlight the potential of nutritional interventions to influence weight and BMI. The introduction of a nutritional protocol can instigate educational opportunities to discuss nutrition in this vulnerable population. This education could be a contributing factor to the difference in weight loss between the pre- and postimplementation cohorts regardless of risk or consultation with a dietitian. The two-fold decrease in weight loss for patients receiving nutritional intervention further heightens the importance of following through with appropriate nutritional interventions and the need for frequent assessment. Ultimately, the introduction of nutrition as an element of treatment appears to warrant a change in practice. Future research is needed to ascertain the influence of each intervention.
Limitations
The appropriation of time was not fully considered for this quality improvement project. Longitudinal weight loss results could be exponentially improved with an increased time frame to account for patients entering the project at different phases of care or the long intervals between provider visits and data collection for weight and BMI. Inconsistent adherence to the screening protocol and initiation of the nutritional intervention were also limitations.
Implications for Nursing
A nurse-driven nutritional screening protocol using a validated, patient-reported screening tool, such as the abPG-SGA, can result in early identification of malnourished or at-risk patients. If used consistently, it can lead to an increase in nurse-driven interventions and identification of at-risk patients with cancer, resulting in improved outcomes. A core oncology nursing competency is the integration of nutritional screening and the appropriate management of malnutrition (Krishnasamy et al., 2017; van Veen et al., 2017). In addition, nurse leaders can consider introducing a nutritional screening protocol for all patients diagnosed with cancers that disproportionately affect nutrition. 
Conclusion
The implementation of a nutritional screening protocol has the potential to improve patient-related health outcomes in the HNC population. Establishing a nutritional screening protocol can predicate early nutritional intervention, providing nurses with the ongoing ability to address malnutrition.
About the Author(s)
Ashley Laursen, DNP, BS, RN, is a medical science liaison at Ipsen Biopharmaceuticals in Charleston, SC. The author takes full responsibility for this content and did not receive honoraria or disclose any relevant financial relationships. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Laursen can be reached at ashleylaursen@gmail.com, with copy to CJONEditor@ons.org. (Submitted August 2019. Accepted February 21, 2020.)
References
Alshadwi, A., Nadershah, M., Carlson, E.R., Young, L.S., Burke, P.A., & Daley, B.J. (2013). Nutritional considerations for head and neck cancer patients: A review of the literature. Journal of Oral and Maxillofacial Surgery, 71(11), 1853–1860. https://doi.org/10.1016/j.joms.2013.04.028
Álvaro Sanz, E., Garrido Siles, M., Rey Fernández, L., Villatoro Roldán, R., Rueda Domínguez, A., & Abilés, J. (2019). Nutritional risk and malnutrition rates at diagnosis of cancer in patients treated in outpatient settings: Early intervention protocol. Nutrition, 57, 148–153. https://doi.org/10.1016/j.nut.2018.05.021
Datema, F.R., Ferrier, M.B., & Baatenburg de Jong, R.J. (2011). Impact of severe malnutrition on short-term mortality and overall survival in head and neck cancer. Oral Oncology, 47(9), 910–914. https://doi.org/10.1016/j.oraloncology.2011.06.510
Gabrielson, D.K., Scaffidi, D., Leung, E., Stoyanoff, L., Robinson, J., Nisenbaum, R., . . . Darling, P.B. (2013). Use of an abridged scored Patient-Generated Subjective Global Assessment (abPG-SGA) as a nutritional screening tool for cancer patients in an outpatient setting. Nutrition and Cancer, 65(2), 234–239. https://doi.org/10.1080/01635581.2013.7555
Gill, A., Givi, B.B., & Moore, M.G. (2018). AHNS series—Do you know your guidelines? Assessment and management of malnutrition in patients with head and neck cancer: Review of the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Head and Neck, 41(3), 577–583. https://doi.org/10.1002/hed.24866
Gorenc, M., Kozjek, N.R., & Strojan, P. (2015). Malnutrition and cachexia in patients with head and neck cancer treated with (chemo)radiotherapy. Reports of Practical Oncology and Radiotherapy, 20(4), 249–258.
Krishnasamy, K., Yoong, T.L., Chan, C.M., Choong, L.P., & Chinna, K. (2017). Identifying malnutrition: Nutritional status in newly diagnosed patients with cancer. Clinical Journal of Oncology Nursing, 21(1), E23–E29. https://doi.org/10.1188/17.CJON.E23-E29
Lim, S.L., Ong, K.C.B., Chan, Y.H., Loke, W.C., Ferguson, M., & Daniels, L. (2012). Malnutrition and its impact on cost of hospitalization, length of stay, readmission and 3-year mortality. Clinical Nutrition, 31(3), 345–350. https://doi.org/10.1016/j.clnu.2011.11.001
Lis, C.G., Gupta, D., Lammersfeld, C.A., Markman, M., & Vashi, P.G. (2012). Role of nutritional status in predicting quality of life outcomes in cancer—A systematic review of the epidemiological literature. Nutrition Journal, 11, 27. https://doi.org/10.1186/1475-2891-11-27
Melnyk, B.M., & Fineout-Overholt, E. (2015). Evidence-based practice in nursing and healthcare: A guide to best practice (3rd ed.). Wolters Kluwer.
Müller-Richter, U., Betz, C., Hartmann, S., & Brands, R.C. (2017). Nutrition management for head and neck cancer patients improves clinical outcome and survival. Nutrition Research, 48, 1–8. https://doi.org/10.1016/j.nutres.2017.08.007
Paccagnella, A., Morello, M., Da Mosto, M.C., Baruffi, C., Marcon, M.L., Gava, A., . . . Marchiori. C. (2010). Early nutritional intervention improves treatment tolerance and outcomes in head and neck cancer patients undergoing concurrent chemoradiotherapy. Supportive Care in Cancer, 18(7), 837–845. https://doi.org/10.1007/s00520-009-0717-0
Prevost, V., Joubert, C., Heutte, N., & Babin, E. (2014). Assessment of nutritional status and quality of life in patients treated for head and neck cancer. European Annals of Otorhinolaryngology, Head and Neck Diseases, 131(2), 113–120. https://doi.org/10.1016/j.anorl.2013.06.007
Reber, E., Gomes, F., Vasiloglou, M.F., Schuetz, P., & Stanga, Z. (2019). Nutritional risk screening and assessment. Journal of Clinical Medicine, 8(7), 1065. https://doi.org/10.3390/jcm8071065
Shahvazi, S., Onvani, S., Heydari, M., Mehrzad, V., Nadjarzadeh, A., & Fallahzadeh, H. (2017). Assessment of nutritional status using abridged scored Patient-Generated Subjective Global Assessment in cancer patient. Journal of Cancer Research and Therapeutics, 13(3), 514–518. https://doi.org/10.4103/0973-1482.177500
Talwar, B., Donnelly, R., Skelly, R., & Donaldson, M. (2016). Nutritional management in head and neck cancer: United Kingdom national multidisciplinary guidelines. Journal of Laryngology and Otology, 130(S2), S32–S40. https://doi.org/10.1017/S0022215116000402
van Veen, M.R., Hoedjes, M., Versteegen, J.J., van de Meulengraaf-Wilhelm, N., Kampman, E., & Beijer, S. (2017). Improving oncology nurses’ knowledge about nutrition and physical activity for cancer survivors. Clinical Journal of Oncology Nursing, 44(4), 488–496. https://doi.org/10.1188/17.ONF.488-496




