Symptom Distress: Implementation of Palliative Care Guidelines to Improve Pain, Fatigue, and Anxiety in Patients With Advanced Cancer
Background: The integration of palliative care into standard oncology care is supported by research to improve quality of life and symptom distress in patients with advanced cancer. In 2016, the American Society of Clinical Oncology (ASCO) released practice guidelines for oncology palliative care that emphasized interprofessional assessment and management of this patient population.
Objectives: The purpose of this study was to evaluate the effect of clinical guidelines on symptom distress in patients with advanced cancer.
Methods: In two oncology palliative care clinics, the Edmonton Symptom Assessment Scale (ESAS) scores for pain, fatigue, and anxiety were measured prior to consultation (T1) and at two subsequent visits (T2 and T3). A standardized documentation template was used to measure fidelity for key guideline components.
Findings: Pain, fatigue, and anxiety ESAS scores were statistically lower from T1 to T3. The frequency of patients having a decrease of 2 or more points for all symptoms increased compared to baseline data. There was 100% compliance to the documentation template during the guideline implementation.
Jump to a section
About 1.68 million people are diagnosed with cancer annually in the United States, and about 50% of these patients are expected to have uncontrolled distressing symptoms, such as pain, fatigue, and anxiety, that negatively affect quality of life (American Cancer Society, 2017). Distressing symptoms are even more prevalent in patients with advanced cancers, which are defined as cancers with distant metastatic disease, end-stage disease, cancer that is life-limiting, or with a prognosis of 6–24 months (Aboshaiqah et al., 2016; Ferrell et al., 2017). Uncontrolled pain, fatigue, and emotional distress (anxiety and/or depression) are the most commonly reported symptoms and often are reported as severe (Basch et al., 2016; DeShields, Potter, Olsen, & Liu, 2014; Donovan, Thompson, & Jacobsen, 2012; Hwang, Cho, & Yoo, 2016; Stark, Tofthagen, Visovky, & McMillan, 2012). These symptoms may result from emotional, psychosocial, or spiritual distress, as well as the physical burden of the disease; therefore, comprehensive care of these patients requires an interprofessional approach. Nurses, in particular, have a significant opportunity to contribute to comprehensive care and to coordinate the many needs of the patients (Baker, Krok-Schoen, & McMillan, 2016; DeShields et al., 2014; Didwaniya, Tanco, de la Cruz, & Bruera, 2015; Ferrell, Smith, Levit, & Balogh, 2014; Li, Xiao, Yang, & Zhao, 2017).
As cancer treatments continue to advance, patients are living longer with distressing symptoms, and there is a need to provide concurrent palliative care with evidence-based oncology care to address the effect of cancer and its treatment on quality of life (Dionne-Odom et al., 2015; Ferrell et al., 2014, 2017; Haun et al., 2017; Walling at el., 2016). The integration of palliative care into the treatment plan of patients with advanced cancer has been recognized by multiple national organizations, including the American Society of Clinical Oncology, the National Comprehensive Cancer Network, the Hospice and Palliative Nurses Association, and the Oncology Nursing Society. Integrating palliative care planning is recognized as an essential component in providing quality cancer care to best meet quality-of-life needs and to assist with the management of distressing symptoms (Dans et al., 2017; Ferrell et al., 2014, 2017). With palliative care as an accepted component of quality cancer care, the effect of care can best be evaluated by using standard measures. Such measurements include objective changes in symptom distress, healthcare resource use, and the effect on the patient’s experience. This allows a better understanding of the direct impact of palliative care within the context of the delivery of care for patients with advanced cancer.
In 2016, evidence-based guidelines were released by the American Society of Clinical Oncology (ASCO) specific to the provision of oncology palliative care, with an increased focus on the psychosocial and interprofessional components of the consultation, including coping assessment, care coordination, and caregiver needs (Ferrell et al., 2017). Unique to these guidelines are the recommended key components of each palliative care consultation to directly address multiple areas of psychosocial and caregiver concerns that have been found to influence a patient’s quality of life and physical suffering. The guidelines are composed of the following specific care components (Ferrell et al., 2017):
• Rapport and relationship building with patient and caregiver
• Symptom distress and functional status management
• Exploration of understanding of diagnosis and prognosis
• Clarification of treatment goals
• Assessment and support for coping needs
• Care coordination with other care providers
• Referrals to other care providers as indicated
The specific aims of the study were that the patients seen in the outpatient palliative care clinic would (a) receive the guideline-based consultation evidenced by fidelity (100% compliance and completion) with a standardized documentation template; (b) rate their pain, fatigue, and anxiety prior to their initial consultation (T1) in the palliative care clinic and prior to the two follow-up visits (T2 and T3); and (c) experience pain, fatigue, and anxiety. It was expected that, with the incorporation of the practice guidelines and an increase in interprofessional care, the patient symptom distress would improve, evidenced by a decrease in patient Edmonton Symptom Assessment Scale (ESAS) scores for pain, fatigue, and anxiety.
Methods
A within-subjects pre-/post-test design was used to measure the self-reported patient symptom scores related to pain, fatigue, and anxiety before and after a standardized oncology palliative care consultation.
Setting
The study was conducted at two part-time palliative care clinics in the Levine Center Institute (LCI), part of the Carolinas Healthcare System (CHS), from April to August 2017. Palliative care services were provided through collaboration with Carolinas Palliative Care and Hospice Group (CPCHG), a medical group within CHS. The palliative care teams included a physician (MD), a nurse practitioner (NP), an RN, and a licensed clinical social worker (LCSW). An RN navigator and registered dietitian were also available upon referral and worked within the same clinical area.
Participants
All new patients with advanced cancer referred to the palliative care clinics were evaluated for inclusion in the study. Inclusion criteria were being aged 18 years or older, able to speak English, and diagnosed with any type of advanced cancer. Exclusion criteria were patients who were non-English speaking and patients with low-stage, curable cancer (not defined as advanced). All three ESAS scores (T1, T2, and T3) had to be obtained for the analysis; therefore, only patients who completed all three visits were included.
Procedure and Measurements
This study was reviewed by the institutional review board committee at CHS and was determined to be exempt from review. For the guidelines to be followed in detail, a documentation template was created within the electronic health record (EHR) because each visit and fidelity was monitored (see Figure 1). The components of the template included specific attention to the essential components of care that are listed within the ASCO practice guidelines. 
To introduce use of these guidelines, this study was implemented to evaluate the effect of symptom distress on patients with advanced cancer after the incorporation of the ASCO palliative care clinical guidelines used within an oncology palliative care consultation. Symptom distress scores were measured at T1, T2, and T3. The ESAS tool was initially validated in a palliative care setting and subsequently has been shown to have success in documenting the symptom experience of patients with cancer (Bruera, Kuehn, Miller, Selmser, & MacMillan, 1991; Chang, Hwang, & Feuerman, 2000; Hui & Bruera, 2017). However, the ESAS is not without limitations because tracking symptom distress and subsequent worsening or improvement is still not a standardized practice for all clinicians in this field (Hui & Bruera, 2017; Rauenzahn, Schmidt, Jones, Aduba, & Tenner, 2016). Specific attention was focused on the ESAS scores for pain, fatigue, and anxiety for all new patient referrals.
Before being seen by the MD or NP in the palliative care clinic, each patient completed an ESAS tool to rank the presence and intensity of 10 symptoms (pain, fatigue, anxiety, depression, insomnia, well-being, drowsiness, appetite, nausea, and shortness of breath). As shown in Figure 2, the clinic flow was streamlined to allow translation (i.e., transferability/replication for other clinic settings). The patient was educated on completion of the tool by the medical assistant or RN who placed the patient in an examination room and assisted with the tool. This process took 5–10 minutes and was incorporated into the rooming process. This was repeated prior to each of the two follow-up visits, and scores were compared with specific attention to pain, fatigue, and anxiety. The provider used the tool to guide the evaluation of the patient based on the symptom reports and scores to determine the plan of care and necessary interprofessional referrals while completing the comprehensive assessment of the patient and family members. Data were then extracted from the EHR and maintained within a secure database for analysis. 
Statistical Analysis
Descriptive statistics were conducted on patient demographic information and the primary cancer diagnosis. Repeated-measures analyses of variance (ANOVAs) with post hoc pairwise comparisons were conducted to evaluate pain, fatigue, and anxiety across the three time points. The dependent variables for the study included the symptom scores of pain, fatigue, and anxiety, and the standardized palliative care consultation served as the independent variable. Pearson correlation coefficients were conducted among pain, fatigue, and anxiety at each timepoint and between change scores. In addition, change scores from T1 to T3 were calculated and Pearson r correlations were conducted on change scores among pain, fatigue, and anxiety. ESAS scores that decreased by 2 or more points indicated clinical significance (Zhao et al., 2014). Statistical analyses were performed with IBM SPSS Statistics, version 24.0, with alpha set to 0.05.
Results
Sample Characteristics
A total of 50 patients were included for initial evaluation within the two clinics during the project time period, and 31 (62%) completed all three visits for final inclusion in the sample. Twelve patients did not have both follow-up visits (did not show or were scheduled to follow-up only as needed), six patients were referred to hospice during the project time period, and one patient died. Thirty-one patients were included in the final sample. The mean time between T1 and T2 was 28 days (median = 28 days), and the mean time between T2 and T3 was 38 days (median = 36 days). Table 1 displays descriptive statistics for demographic characteristics and cancer type. 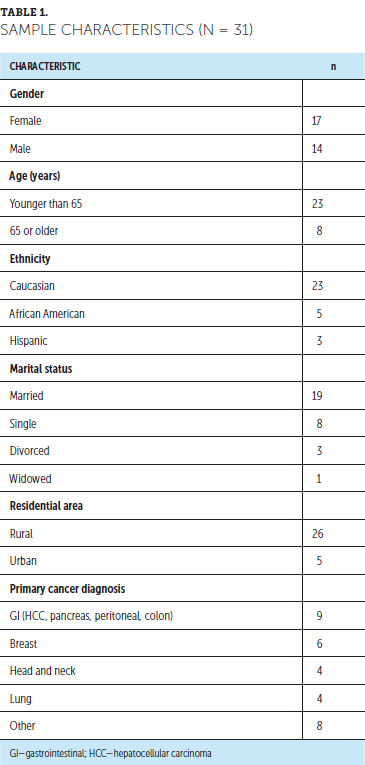
Pain
A repeated-measures ANOVA with a Greenhouse–Geisser correction determined that ESAS pain scores differed among the three time points (F[1.56, 46.69] = 4.648, p = 0.021). Post-hoc tests using the Bonferroni correction revealed a decrease from T1 (mean = 5, SD = 3.183) to T2 (mean = 4.29, SD = 2.877), which was not statistically significant (p = 0.278), and a decrease from T2 to T3 (mean = 3.39, SD = 2.848), which was not statistically significant (p = 0.28). The decrease from T1 to T3 was statistically significant (p = 0.005) (see Table 2). 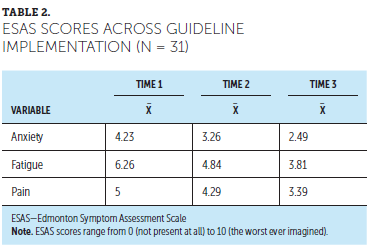
Anxiety
A repeated-measures ANOVA with a Greenhouse-Geisser correction showed a statistically significant difference among the three time points (F[1.84, 55.11] = 5.62, p = 0.007). Post-hoc tests using the Bonferroni correction revealed a decrease from T1 (mean = 4.23, SD = 3.27) to T2 (mean = 3.26, SD = 2.7), which was not statistically significant (p = 0.077), and a decrease from T2 to T3 (mean = 2.49, SD = 2.55), which was also not statistically significant (p = 0.192). However, the decrease in ESAS anxiety scores from T1 to T3 was statistically significant (p < 0.001).
Fatigue
A repeated-measures ANOVA with a Greenhouse-Geisser correction showed a statistically significant decrease in ESAS fatigue scores across the three time points (F[1.81, 54.32] = 8.352, p = 0.001). Post-hoc tests using the Bonferroni correction revealed a decrease from T1 (mean = 6.26, SD = 2.94) to T2 (mean = 4.84, SD = 2.97), which was statistically significant (p = 0.011), and a decrease from T2 to T3 (mean = 3.81, SD = 3.16), which was not statistically significant (p = 0.085). The decrease from T1 to T3 was statistically significant (p = 0.001).
Correlations Among ESAS Symptom Change Scores
Pearson correlation coefficient was used to determine the presence of any relationships between pain, anxiety, and fatigue change scores. A statistically significant correlation was found for anxiety change and fatigue change (r[34] = 0.39, p = 0.019). The correlations between pain and fatigue change scores, as well as pain and anxiety changes scores, were not statistically significant (p = 0.148 and 0.383, respectively).
ESAS Score Comparison
To evaluate clinical significance as per methods used in Zhao et al. (2014), descriptive statistics (n and %) were conducted to determine the percentage of patients who experienced a decrease of 2 or more points in ESAS scores within each symptom between T1 and T3. These percentages were then compared to the percentage of patients who had a decrease of 2 or more points in symptom scores during the pre-project period (which involved delivery of standard care). A greater percentage of patients had a decrease of 2 or more points in their ESAS scores following the incorporation of the ASCO practice guidelines compared to the pre-project period (see Table 3). 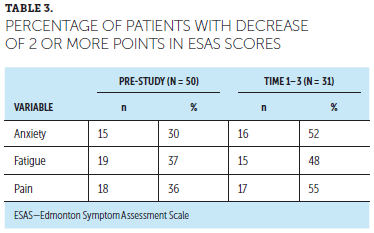
Discussion
The purpose of this study was to implement the 2016 ASCO oncology palliative care practice guidelines into the care of patients with advanced cancer within outpatient palliative care clinic settings. The results of this study support previous findings of the complex symptom-related needs of patients with advanced cancer and the need for a comprehensive strategy to positively affect quality of life (Didwaniya et al., 2015; Donovan et al., 2012; Ferrell et al., 2014; Hui & Bruera, 2017). The two aims of the study were met, which included having patients seen in the outpatient palliative care clinic receive the guideline-based consultation with a standardized documentation template and rate their pain, fatigue, and anxiety at T1, T2, and T3. The results from this study reported ESAS scores for pain, fatigue, and anxiety decreasing by 2 or more points only occurred for the symptom of fatigue (mean = 2.45). The mean difference of the three scores was 1.61 for pain and 1.74 for anxiety, which is trending toward a decrease of 2 points.
The ESAS score change measurements of all three symptoms was statistically significant from T1 to T3. Fatigue was the only symptom that decreased after one follow-up visit (T2), whereas pain and anxiety scores required three visits for a statistically significant change. Because of the elements of the guidelines, there was intent on the part of the nurses, in particular, to specifically assess and address caregiver needs and the coping skills of patients and their caregivers. During the study, patients indicated that knowing their caregivers were supported improved their physical feelings of distress. In addition, involving social workers, navigators, and dietitians early in the consultation was felt to be helpful in improving any psychosocial or non-medical elements of pain, fatigue, and anxiety. This collaboration would be worthy of further research and outcome measurement.
Prior to the implementation of this study and the incorporation of the practice guidelines, the ESAS scores decreased 2 or more points in 36% of patients for pain, 37% for fatigue, and 30% for anxiety. After the implementation of the practice guidelines, a higher percentage of patients did have a decrease in ESAS scores of 2 or more points. This represents additional substantial improvement in the patient’s reported experience, providing additional evidence to support the integration of interprofessional outpatient palliative care.
Limitations
Although this study did achieve its aims with statistical improvement of patient ESAS scores in pain, fatigue, and anxiety following a newly developed guideline-based palliative care consultation, there were some limitations that should be acknowledged. Limitations include variability of provider practices and patient reporting. Despite education and scripting for the providers participating in the project, retrospective reflection of the providers revealed their approach to the discussion of the ESAS scores was inconsistent. Depending on clinic flow and scheduling, the reflection on previous scores with the patient and correlating the score to the subjective report of the patient was not consistent. For example, it was found through the data collection process that several unchanged scores between visits did not reflect the subjective report documented in the medical note. Their score was an unchanged 10 of 10; however, the note recorded a patient response of improved, controlled pain. It could be suggested to remind patients of their previous score; however, this may be viewed as leading for their report.
Second, with the mean and median times between visits ranging from 28 to 38 and 28 to 36 days, respectively, a significant time was demonstrated between visits which could allow multiple changes in the patient’s disease status and/or treatment plan to occur. This could have interfered with the true causality of each symptom distress score and occurred because of multiple reasons, including limited capacity of a part-time clinic, patient schedule changes, and multiple obligations of the patient and family.
Implications for Research
Future investigations with larger samples and an extended time period would be beneficial to determine what time interval could more significantly decrease ESAS scores and patient symptom distress. Because of the labile nature of the advanced cancer trajectory, to improve multiple symptom scores in a more significant way, either more frequent assessments or a longer time period of measurement may be needed. Correlating the symptom scores to specific interventions of the interprofessional team would add to the understanding of the impact on these symptoms and determine what role within the team led to improvement and how to strategically continue the care design. Opportunities and implications for nurses are robust, although somewhat dependent on resources and the structure of the clinic and team. Understanding how the integration of the guidelines affects the caregiver’s experience would be beneficial for a clearer understanding of caregiver needs and resources and how that ultimately improves the patient’s quality of life. 
Conclusion
The integration of the ASCO oncology palliative care guidelines into an interprofessional outpatient palliative care setting demonstrated a significant decrease in pain, anxiety, and fatigue in patients with advanced cancer over time. A comprehensive, interprofessional palliative care approach is recommended to improve symptom distress and quality of life in this vulnerable patient population. [[{"fid":"49666","view_mode":"default","fields":{"format":"default","alignment":"","field_file_image_alt_text[und][0][value]":false,"field_file_image_title_text[und][0][value]":false},"link_text":null,"type":"media","field_deltas":{"1":{"format":"default","alignment":"","field_file_image_alt_text[und][0][value]":false,"field_file_image_title_text[und][0][value]":false}},"attributes":{"class":"media-element file-default","data-delta":"1"}}]]
About the Author(s)
Niki Koesel, DNP, ANP, ACHPN, FPCN, is a medical science liaison at Helsinn Therapeutics in Charlotte, NC; Christine Tocchi, PhD, APRN, GNP, is an assistant professor in the School of Nursing at Duke University in Chapel Hill, NC; Larry Burke, MD, is a medical director at Atrium Health in Charlotte, NC; Tracey Yap, PhD, RN, FGSA, FAAN, is an associate professor in the School of Nursing at Duke University; and Alison Harrison, DNP, RN, NP-C, ACHPN, is a nurse practitioner at Mercy North Iowa Palliative Care in Mason City. The authors take full responsibility for this content. Koesel has previously served on speakers bureaus for the Lung Cancer Alliance and as a board member for the Hospice and Palliative Care Credentialing Center. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Koesel can be reached at niki.koesel@helsinn.com, with copy to CJONEditor@ons.org. (Submitted July 2018. Accepted November 18, 2018.)
References
Aboshaiqah, A., Al-Saedi, T.S., Abu-Al-Ruyhaylah, M.M., Aloufi, A.A., Alharbi, M.O., Alharbi, S.S., . . . Al-Erwi, A.F. (2016). Quality of life and satisfaction with care among palliative cancer patients in Saudi Arabia. Palliative and Supportive Care, 14, 621–627. https://doi.org/10.1017/S1478951516000432
American Cancer Society. (2017). Cancer facts and figures, 2017. Retrieved from https://www.cancer.org/research/cancer-facts-statistics/all-cancer-fact…
Baker, T.A., Krok-Schoen, J.L., & McMillan, S.C. (2016). Identifying factors of psychological distress on the experience of pain and symptom management among cancer patients. BMC Psychiatry, 4, 52. https://doi.org/10.1186/s40359-016-0160-1
Basch, E., Deal, A.M., Kris, M.G., Scher, H.I., Hudis, C.A., Sabbatini, P., . . . Schrag, D. (2016). Symptom monitoring with patient-reported outcomes during routine cancer treatment: A randomized controlled trial. Journal of Clinical Oncology, 34, 557–565.
Bruera, E., Kuehn, N., Miller, M.J., Selmser, P., & MacMillan K. (1991). The Edmonton Symptom Assessment System (ESAS): A simple method for the assessment of palliative care patients. Journal of Palliative Care, 7(2), 6–9.
Chang, V.T., Hwang, S.S., & Feuerman, M. (2000). Validation of the Edmonton Symptom Assessment Scale. Cancer, 88, 2164–2171.
Dans, M., Smith, T., Back, A., Baker, J.N., Bauman, J.R., Beck, A.C., . . . Scavone, J.L. (2017). NCCN guidelines insights: Palliative care. Journal of the National Comprehensive Cancer Network, 15, 989–997. https://doi.org/10.6004/jnccn.2017.0132
DeShields, T.L., Potter, P., Olsen, S., & Liu, J. (2014). The persistence of symptom burden: Symptom experience and quality of life of cancer patients across one year. Supportive Care in Cancer, 22, 1089–1096. https://doi.org/10.1007/s00520-013-2049-3
Didwaniya, N., Tanco, K., de la Cruz, M., & Bruera, E. (2015). The need for a multidisciplinary approach to pain management in advanced cancer: A clinical case. Palliative and Supportive Care, 13, 389–394. https://doi.org/10.1017/S1478951514000108
Dionne-Odom, J.N., Azuero, A., Lyons, K.D., Hull, J.G., Tosteson, T., Li, Z., . . . Bakitas, M.A. (2015). Benefits of early versus delayed palliative care to informal family caregivers of patients with advanced cancer: Outcomes from the ENABLE III randomized controlled trial. Journal of Clinical Oncology, 33, 1446–1452. https://doi.org/10.1200/JCO.2014.58.7824
Donovan, K.A., Thompson, L.M.A., & Jacobsen, P.B. (2012). Pain, depression, and anxiety in cancer: Biobehavioral approaches for the life course. In R.J. Moore (Eds.), Handbook of pain and palliative care (pp. 615–637). New York, NY: Springer.
Ferrell, B.R., Smith, T.J., Levit, L., & Balogh, E. (2014). Improving the quality of cancer care: Implications for palliative care. Journal of Palliative Medicine, 17, 393–399. https://doi.org/10.1089/jpm.2013.0536
Ferrell, B.R., Temel, J.S., Temin, S., Alsi, E.R., Balboni, T.A., Basch, E.M., . . . Smith, T.J. (2017). Integration of palliative care into standard oncology care: American Society of Clinical Oncology clinical practice guideline update. Journal of Clinical Oncology, 35, 96–112. https://doi.org/10.1200/JCO.2016.70.1474
Gerrits, M.M., van Marwijk, H.W., van Oppen, P., van der Horst, H., & Penninx, B.W. (2015). Longitudinal association between pain, and depression and anxiety over four years. Journal of Psychosomatic Research, 78, 64–70. https://doi.org/10.1016/j.jpsychores.2014.10.011
Haun, M.W., Estel, S., Rücker, G., Friedrich, H., Villalobos, M., Thomas, M., & Hartmann, M. (2017). Early palliative care for adults with advanced cancer. Cochrane Database of Systematic Reviews, 6, CD011129. https://doi.org/10.1002/14651858.CD011129.pub2
Hui, D., & Bruera, E. (2017). The Edmonton Symptom Assessment System 25 years later: Past, present, and future developments. Journal of Pain and Symptom Management, 53, 630–643. https://doi.org/10.1016/j.jpainsymman.2016.10.370
Hwang, K.H., Cho, O.H., & Yoo, Y.S. (2016). Symptom clusters of ovarian cancer patients undergoing chemotherapy, and their emotional status and quality of life. European Journal of Oncology Nursing, 21, 215–222. https://doi.org/10.1016/j.ejon.2015.10.007
Li., X.M., Xiao, W.H., Yang, P., & Zhao, H.X. (2017). Psychological distress and cancer pain: Results from a controlled cross-sectional survey in China. Scientific Reports, 7, 39397. https://doi.org/10.1038/srep39397
Rauenzahn, S.L., Schmidt, S., Jones, J., Aduba, I., & Tenner, L.L. (2016). Integrating palliative care in oncology clinics: An application of the Edmonton Symptom Assessment System (ESAS) [Abstract 168]. Journal of Clinical Oncology, 34(Suppl. 26), 168.
Stark, L., Tofthagen, C., Visovsky, C., & McMillan, S.C. (2012). The symptom experience of patients with cancer. Journal of Hospice and Palliative Nursing, 14, 61–70. https://doi.org/10.1097/NJH.0b013e318236de5c
Walling, A.M., Tisnado, D., Ettner, S.L., Asch, S.M., Dy, S.M., Pantoja, P., . . . Lorenz, K.A. (2016). Palliative care specialist consultation is associated with supportive care quality in advanced cancer. Journal of Pain and Symptom Management, 52, 507–514. https://doi.org/10.1016/j.jpainsymman.2016.04.005
Zhao, F., Chang, V.T., Cleeland, C., Cleary, J.F., Mitchell, E.P., Wagner, L.I., & Fisch, M.J. (2014). Determinants of pain severity changes in ambulatory patients with cancer: An analysis from Eastern Cooperative Oncology Group (E2Z02). Journal of Clinical Oncology, 32, 312–319.




