Management Across Settings: An Ambulatory and Community Perspective for Patients Undergoing CAR T-Cell Therapy in Multiple Care Settings
Background: Many patients are referred to chimeric antigen receptor (CAR) T-cell therapy programs from outside their primary oncology setting and community. Collaboration between the referring and treating providers is required to coordinate safe and effective care.
Objectives: This article presents an overview of key considerations for referring providers and institutions prior to and following CAR T-cell therapy.
Methods: Definitions of the consultation and workup, leukapheresis, bridging, lymphodepleting chemotherapy, infusion and monitoring, and long-term follow-up phases are presented, along with specific considerations for referring centers.
Findings: Although CAR T-cell therapy is limited to select centers, the process of supporting and educating patients and their caregivers requires a partnership between referring and treating providers. As CAR T-cell indications expand, management of patients in diverse settings requires a collaborative and evidence-based approach to support safe and effective care.
Jump to a section
Approval of chimeric antigen receptor (CAR) T-cell therapies by the U.S. Food and Drug Administration ([FDA], 2017a, 2017b) has extended this standard-of-care treatment to adult and pediatric populations with B-cell malignancies. A limited number of institutions are certified to offer this therapy. The majority of these centers have existing blood and marrow transplantation programs because a similar infrastructure is required (Taylor, Rodriguez, Reese, & Anderson, 2019). For treatment centers to become qualified to offer CAR T-cell therapy, they must go through a training and management program under the Risk Evaluation and Mitigation Strategy (REMS), which is a requirement of the FDA and developed by the pharmaceutical company that manufactures the drug (Novartis Pharmaceuticals, 2018c).
Although this number has grown from 59 programs in May 2018 to 160 programs certified as of February 2019 to provide one or both of the FDA-approved CAR T-cell agents, many patients continue to seek treatment outside of their community, state, or country (ASCO Post, 2018; Novartis Pharmaceuticals, 2018a). This process involves collaborative relationships between the referring provider (a primary oncologist), the treating provider (a CAR T-cell specialist at an approved treatment facility), and community providers (individuals providing care for recipients of CAR T-cell therapy beyond the oncology setting, such as primary care or emergency providers). Patients are co-managed between their primary oncologists and CAR T-cell specialists at a certified center. They are then transitioned back to the primary oncologist with specific instructions and contact information from the treating center in the event support is required postinfusion. As a result, care coordination between referring and providing institutions is critical to the successful treatment and management of eligible patients leading up to and following CAR T-cell therapy. As part of an interprofessional care team, nurses are instrumental to ensuring care coordination through education and communication with patients and their caregivers.
This article presents an overview of considerations for referring centers and community-based care providers, separated into six phases involved in CAR T-cell therapy. Recommendations for referring institutions are explored in the context of the consultation and workup, leukapheresis, bridging, lymphodepleting chemotherapy, infusion and monitoring, and long-term follow-up phases (see Table 1). 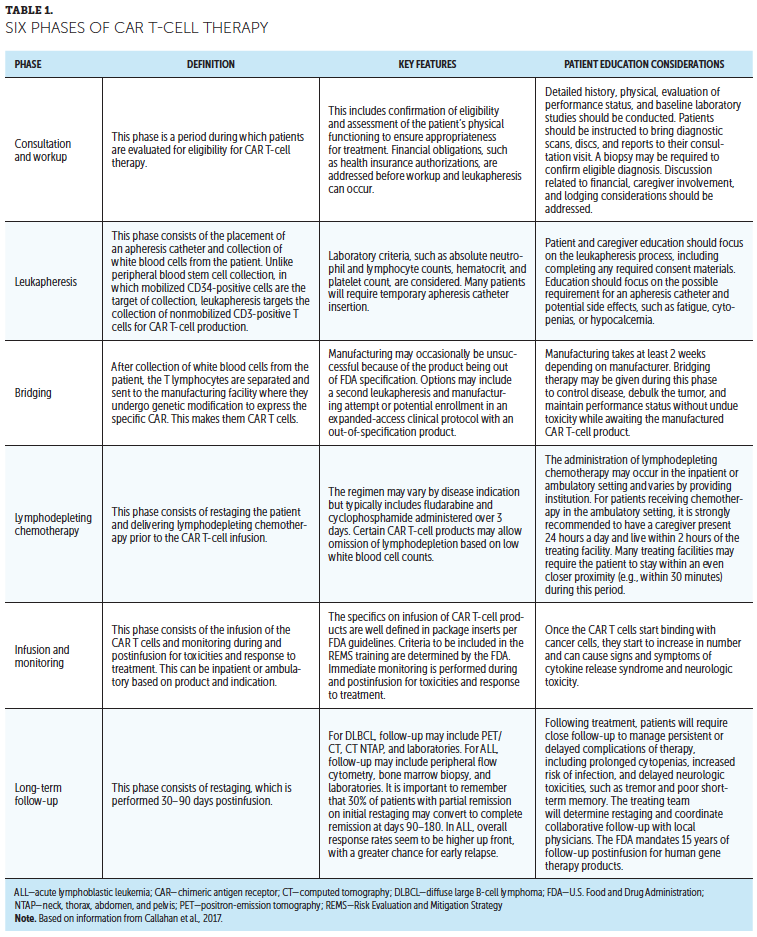
Patient Management Considerations
Consultation and Workup
Definition and key features: The consultation and workup phase is a period during which patients are evaluated for eligibility for CAR T-cell therapy. This consists of three components: confirming that patients meet the indication and eligibility criteria for treatment, ensuring patients are physiologically stable to proceed to treatment, and confirming insurance/self-pay authorization and logistical requirements.
In diffuse large B-cell lymphoma (DLBCL) and acute lymphoblastic leukemia (ALL), eligibility includes confirming CD19-positive status using flow cytometry or immunohistochemical staining. CD19 status should be determined, particularly if there was prior treatment with a CD19-targeted agent, such as blinatumomab. CD19 antigen loss is an established mechanism of disease relapse after CD19-directed therapy (Ruella & Maus, 2016). However, CD19-positive status, including density, is not clearly associated with outcomes in DLBCL and ALL CAR T-cell trials (Maude et al., 2014; Neelapu et al., 2017; Schuster et al., 2019).
Concurrent with confirmation of eligibility is an assessment of the patient’s physical functioning to ensure appropriateness for treatment. A detailed history and physical, evaluation of performance status, and baseline laboratory studies should be conducted (see Figure 1). Ultimately, any patient who meets the FDA-approved indications should be seen at a certified CAR T-cell center for evaluation. 
Referring center considerations: The referring center is pivotal in providing current patient records, including original and most recent diagnostic scans and pathology reports, along with a complete history and physical that can be used to evaluate treatment eligibility.
In the consultation phase, it is important for referring centers to be knowledgeable of the eligibility criteria for CAR T-cell therapy. Patients and caregivers will be educated on the financial and logistical considerations associated with treatment (e.g., travel, lodging, caregiver assistance), all of which can be introduced by the nursing team members (e.g., clinical, advanced practice, and navigator nurses) at each certified treatment center.
Leukapheresis
Definition and key features: Once confirmed as eligible, patients proceed to the leukapheresis phase. This phase consists of the placement of an apheresis catheter and collection of white blood cells from the patient, which are then sent to the manufacturer for processing for CAR T-cell production. Unlike peripheral blood stem cell apheresis, in which mobilized CD34-positive cells are the target, leukapheresis targets the collection of nonmobilized CD3-positive T lymphocytes for CAR T-cell production. The target number of cells is specific to each CAR T-cell product. For tisagenlecleucel (Kymriah®), it is 0.2 to 5 x 106 CAR-positive viable T cells per kg body weight (50 kg or less) or 0.1 to 2.5 x 108 CAR-positive viable T cells per kg body weight (greater than 50 kg) (Novartis Pharmaceuticals, 2018b). For axicabtagene ciloleucel (Yescarta®), the target dose is 2 × 106 CAR-positive viable T cells per kg body weight (minimum of 1 x 106 CAR-positive viable T cells per kg body weight) (Kite Pharma, 2017). The collection can usually be completed in one session lasting two to three hours (McGuirk et al., 2017).
Referring center considerations: Apheresis may occur at the treatment center or at an external apheresis center. Patient assessment should include the following considerations: history of stem cell transplantation, review of current medications, complete blood count (CBC), and vascular access.
For patients who have undergone allogeneic stem cell transplantation, leukapheresis for CAR T-cell therapy is discouraged within three months of transplantation because of the risk for graft-versus-host disease. An evaluation of current medications is important to reduce risk of apheresis and treatment complications, with specific recommended holding periods prior to the apheresis procedure presented in Table 2. In addition, review of a CBC with attention to required minimum hematocrit (greater than 27%) and platelet (greater than 20) values should be completed prior to the procedure to reduce the risk of syncope and bleeding. Assessment for venous access is critical to the efficacy of the leukapheresis procedure, with the potential need for a temporary or permanent dialysis-grade catheter in pediatric patients and appropriate central venous access for adolescent and adult patients. 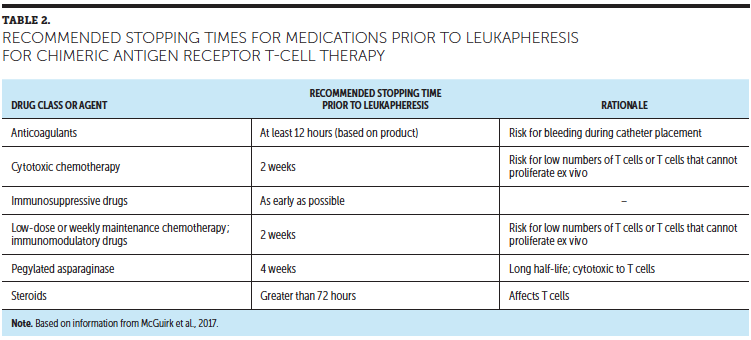
Nurses from the certified treatment center provide patient and caregiver education focused on the leukapheresis process, including potential side effects (e.g., cytopenia, hypocalcemia, cardiovascular events) (Stenzinger & Bonig, 2018). The patients review and complete required consent materials. It is also important to educate patients about the expected period for manufacturing of the CAR T-cell product, which takes at least two weeks (Levine, Miskin, Wonnacott, & Keir, 2017). Patients are informed that manufacturing might not be successful in meeting FDA specification requirements, resulting in some cases requiring a second leukapheresis and re-attempt at manufacturing. Alternatively, some institutions may offer expanded access protocols to allow off-label administration under close monitoring.
Bridging
Definition and key features: The bridging phase corresponds with the manufacturing period required to produce the CAR T-cell product, a period of at least two weeks, depending on the product for infusion (Levine et al., 2017; Perica, Curran, Brentjens, & Giralt, 2018). Because of aggressive disease, patients must be closely monitored during this period for disease progression or other complications. Although efforts are being made to reduce this wait time, the implication for patients with aggressive malignancies is that their disease may progress, and performance status may decline to the point where the CAR T cells cannot be safely given. For this reason, bridging therapy may be given during this surveillance phase to control disease, debulk tumor, and maintain performance status without undue toxicity while awaiting the manufactured CAR T-cell product. The optimal bridging therapy has not been defined and may vary by underlying diagnosis and patient-specific indications.
In lymphoma, the ZUMA-1 clinical trial (axi-cel) only allowed bridging with steroids, whereas the JULIET trial (tis-gen) allowed bridging per investigator choice in 92% of patients (Neelapu et al., 2017). Assessment of patients’ prior responses to chemotherapy, the overall tumor burden, and the anatomy of patient tumors are all important considerations. Typical lymphoma bridging options include IV chemotherapy (e.g., rituximab-gemcitabine-oxaliplatin), oral chemotherapy (e.g., variants of predisone, etoposide, procarbazine, and cyclophosphamide [PEP-C]; oral cyclophosphamide 100 mg daily), high-dose steroids (e.g., dexamethasone 40 mg for four days, repeated as needed) or radiation therapy to symptomatic or large masses (Coleman et al., 2008; Jain et al., 2018; Mounier et al., 2013).
For leukemia, bridging regimens are typically adapted from known B-cell ALL therapies, but lower-intensity therapies are preferred because of fewer infectious complications (Gupta et al., 2018). Novel and targeted agents may also be used, but consideration is given to whether the drug is likely to start working within one to two weeks and if there are lasting effects that may interact with subsequent CAR T-cell therapy. Whichever strategy is used, therapy begins after leukapheresis so that T-cell quality is not affected. Before lymphodepleting chemotherapy conditioning begins (administration of fludarabine and cyclophosphamide), patients during this bridging phase are monitored closely (i.e., weekly) for infection and toxicities.
Referring center considerations: Surveillance and bridging treatment takes place either at the treating or referring center, requiring critical communication among providers. Patient education focuses on the reason for bridging therapy and the importance of frequent monitoring and follow-up, including blood work, imaging, and other procedures to monitor patients’ disease state.
Following the bridging phase, patients will transition, if they have not already done so, to the treating institution for the lymphodepleting chemotherapy and infusion and monitoring phases of the process. Nursing education provided to patients and caregivers about this handoff to the treating center, including the role that the referring provider will have during and following treatment, is important to reinforce the collaborative approach to CAR T-cell therapy and to ensure that the patient experiences continuity of care between these centers.
Lymphodepleting Chemotherapy
Definition and key features: The chemotherapy phase consists of restaging the patient and delivering lymphodepleting chemotherapy prior to CAR T-cell infusion. Restaging is particularly important if the patient received bridging chemotherapy. A clearance period prior to the start of chemotherapy also includes a review of laboratory values and the placement of a catheter for treatment (if not already in place). The administration of lymphodepleting chemotherapy may occur in the inpatient or ambulatory setting and varies by providing institution. The agents may vary by disease type but typically include fludarabine and cyclophosphamide (Callahan, Baniewicz, & Ely, 2017). This chemotherapy will be given over three to four days, depending on diagnosis and product selection. The purpose of this chemotherapy is to prepare the body for CAR T-cell infusion by depleting T cells and creating an optimal environment for the CAR T cells to expand (Turtle et al., 2016). If patients are receiving chemotherapy in the ambulatory setting, it is strongly recommended that they have a caregiver present with them 24 hours a day and that they live within two hours of the treating facility; however, the treating facility may require the patient to stay within closer proximity (e.g., within 30 minutes) during this period.
The caregiver, whether a family member, friend, or other individual, is pivotal in supporting the patient during this period and should be educated on expectations of them during this phase of treatment (see Figure 2). At the completion of lymphodepleting chemotherapy, the patient will proceed to CAR T-cell infusion, which may occur in an inpatient or ambulatory setting, depending on the treating institution. 
Referring center considerations: Although the lymphodepleting chemotherapy phase is conducted at the treating facility, the referring provider can expect communication of the treatment plan, and the patient’s status may be reported out by the nurses at the completion of the CAR T-cell therapy as the patient transitions back to the referring provider for follow-up.
Infusion and Monitoring
Definition and key features: The infusion and monitoring phase consists of infusion of the CAR T cells and immediate monitoring during and postinfusion for toxicities and response to treatment. The process of CAR T-cell infusion is well defined in the product inserts and REMS training (Novartis Pharmaceuticals, 2018b, 2018c).
The two most commonly observed acute toxicities following CAR T-cell infusion are cytokine release syndrome (CRS), which clinically presents as high fevers, tachycardia, hypotension, and hypoxia and may progress to multiorgan failure, and neurologic toxicities, presenting as an encephalopathic syndrome characterized by confusion, tremor, and delirium to seizures and cerebral edema, potentially leading to coma (Bonifant, Jackson, Brentjens, & Curran, 2016). These are primarily managed inpatient with reversal agents, such as tociluzumab and high-dose steroids (Bonifant et al., 2016). A comprehensive overview of acute toxicities is presented in this supplement by Anderson and Latchford (2019).
Once initial toxicities have recovered to grade 1 or less, patients are discharged for close outpatient management. Pancytopenia is observed in most patients. Although initially expected given the lymphodepleting chemotherapy, delayed cytopenias (past day 28) were observed in 24% of patients. Transfusions and intermittent granulocyte–colony-stimulating factor injections (e.g., filgrastim) are treatments to manage cytopenias but are not recommended by the manufacturer within three weeks of CAR T-cell therapy.
Because most B cells express CD19 on their cell surface, infusion of anti-CD19 CAR T cells frequently leads to B-cell aplasia. Although this has often been presumed to correlate with persistence of the CAR T-cell activity, two-year follow-up from the ZUMA-1 trial found that 54% of patients with complete remission at 12 months also had full B-cell recovery, indicating that persistence may not be a requisite for sustained remissions (Locke et al., 2017). Clinically, the destruction of these healthy B cells leads to an immunocompromised state and a risk for frequent infections. Monthly IV immunoglobulin G supplementation is often recommended for patients who experience frequent infections or empirically in the pediatric population.
Restaging positron-emission tomography/computed tomography (PET/CT) scans are performed 30–90 days postinfusion and every three months for the first two years post-therapy. In patients with DLBCL, CAR T-cell therapy is considered definitive therapy, with durable remissions of about 42% at two-year follow-up (Locke et al., 2017). In ALL, the overall response rates seem to be higher up front, with a greater chance for early relapse. Therefore, in certain settings, consolidative allogeneic stem cell transplantation can be considered. This infusion and monitoring phase occurs over about 30 days or when toxicities return to grade 1 or less, at which point the patient may be transferred back to the referring provider.
Long-term follow-up: Handoff procedures to transition patients from the treating to the referring provider allows successful continuity of care and includes a clearly delineated plan for follow-up (Perica et al., 2018). After CAR T-cell therapy, patients will require close follow-up to manage possible delayed complications of therapy, including prolonged cytopenias, increased risk of infection, and delayed neurologic toxicities (Park et al., 2018). Antiviral prophylaxis with acyclovir is recommended for the first year post-treatment, as well as trimethoprim-sulfamethoxazole or pentamidine as prophylaxis for Pneumocystis jirovecii pneumonia for six months post-treatment. Follow-up with the local physician will vary by individual patient; however, management of cytopenias requiring blood product transfusions, growth factors, or IV immunoglobulin infusions may necessitate frequent visits (McConville et al., 2017). Because most patients will follow up with certified CAR T-cell facilities at intervals over time, continuing communication among referring and treating provider teams will ensure coordinated care. Once the patient is stable and counts have recovered, the patient can start following preventive screening guidelines that are recommended for individual age groups. The treating providers will determine follow-up with restaging, and referring providers can recommend follow-up frequency between these staging visits. The FDA (2018) currently mandates 15-year follow-up postinfusion for human gene therapy products.
Referring center considerations: Referring providers should anticipate receipt of medical records related to the full course of treatment (FDA, 2018) (see Figure 3). Nurses from the certified treatment center should provide patient education that emphasizes the potential persistent or long-term toxicities of CAR T-cell therapy, including those relating to infection and cytopenia, which may persist for months following infusion. Examples of these side effects are sleepiness, dizziness, and coordination problems, which present for pediatric (Callahan et al., 2019) and adult (Buitrago, Adkins, Hawkins, Iyamu, & van Oort, 2019) patients. Patients are recommended to abstain from driving and/or operating heavy machinery for eight weeks after CAR T-cell infusion (Kite Pharma, 2017). Nurses from the certified treatment center can provide a patient wallet card to patients after treatment, which provides examples of symptoms that may occur post-treatment, as well as the treating medical center contact information. In any patient treated with CAR T-cell therapy who presents to an external emergency department or ambulatory care setting, presentation of the wallet card and communication with the treating center should be emphasized, particularly in patients with neurologic findings. The academic community continues to learn about potential toxicities of CAR T-cell therapy, including rare entities (e.g., hemophagocytic lymphohistiocytosis), and continues to refine toxicity scoring and monitoring (Lee et al., 2018). 
CAR T-cell therapy recovery involves unique monitoring and management. B-cell aplasia may persist for months to years and will predispose these patients to community-acquired infections (Park et al., 2018). Equally contributing to the risk of infection, patients may experience prolonged neutropenia, in addition to anemia and thrombocytopenia. The etiology of prolonged cytopenia is not well understood but widely recognized. Close attention to peripheral blood counts for transfusion requirements, growth factor support, and consideration of platelet recombination factors can all be considered in this phase. A comprehensive overview of late effects, monitoring, and management is presented for pediatric and adult patients. Although CAR T-cell therapy is associated with durable remissions, close surveillance for disease recurrence is an important part of long-term care for these patients. Unexplained fevers or chills, new pain, or lymphadenopathy should warrant reimaging with PET/CT to rule out relapse. Nurses from the certified treatment center may provide education to patients and caregivers regarding toxicities, expected symptoms, how to care for themselves, and when to call the healthcare provider (Bayer et al., 2017). 
Conclusion
Caring for patients receiving CAR T-cell therapy requires a collaborative approach to care management. Establishing expectations for the referring and providing teams, as well as ensuring clear and open communication, is important to synchronize care (Callahan et al., 2017). Nurses are leaders in care coordination, particularly in navigation roles to ensure transition within and between care settings (Riley & Riley, 2016). The use of immunotherapy identification cards has been recommended for patients to communicate to non-oncology providers about their treatment history (Brahmer, Lacchetti, & Thompson, 2018). As CAR T-cell therapies and their indications are approved, this collaborative approach will ensure care coordination across diverse treatment settings.
About the Author(s)
Alix Beaupierre, BSN, RN, OCN®, is a transplant nurse coordinator, Rachel Lundberg, PA-C, is a physician’s assistant, and Leslie Marrero, BSN, RN, OCN®, BMTCN®, is a transplant nurse coordinator, all at the Moffitt Cancer Center in Tampa, FL; Michael Jain, MD, PhD, is an assistant member at Moffitt Cancer Center and an assistant professor in the Morsani College of Medicine at the University of South Florida in Tampa; Trent Wang, DO, MPH, is an assistant professor of clinical medicine in the Department of Medicine in the School of Medicine at the University of Miami in Florida; and Maritza C. Alencar, DNP, MBA, APRN-BC, BMTCN®, is the director of clinical operations oncology service line in the Sylvester Comprehensive Cancer Center at the University of Miami. The authors take full responsibility for this content. This supplement was supported by an independent educational grant from Bristol-Myers Squibb. Beaupierre and Alencar serve on speakers bureaus for Kite Pharma, and Jain has previously consulted for Kite Pharma. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Beaupierre can be reached at alix.beaupierre@moffitt.org, with copy to CJONEditor@ons.org. (Submitted January 2019. Accepted January 31, 2019.)
References
Anderson, K., & Latchford, T. (2019). Associated toxicities: Assessment and management related to CAR T-cell therapy. Clinical Journal of Oncology Nursing, 23(Suppl. 1), 13–19. https://doi.org/10.1188/19.CJON.S1.13-19
ASCO Post. (2018, May 25). Treatment centers authorized to administer CAR T-cell therapy. Retrieved from http://www.ascopost.com/issues/may-25-2018/treatment-centers-authorized…
Bayer, V., Amaya, B., Baniewicz, D., Callahan, C., Marsh, L., & McCoy, A.S. (2017). Cancer immunotherapy: An evidence-based overview and implications for practice. Clinical Journal of Oncology Nursing, 21(Suppl. 2), 13–21. https://doi.org/10.1188/17.CJON.S2.13-21
Bonifant, C.L., Jackson, H.J., Brentjens, R.J., & Curran, K.J. (2016). Toxicity and management in CAR T-cell therapy. Molecular Therapy-Oncolytics, 3, 16011. https://doi.org/10.1038/mto.2016.11
Brahmer, J.R., Lacchetti, C., & Thompson, J.A. (2018). Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology clinical practice guideline summary. Journal of Oncology Practice, 14, 247–249. https://doi.org/10.1200/JOP.18.00005
Buitrago,J., Adkins, S., Hawkins, M., Iyamu, K., & van Oort, T. (2019). Adult survivorship: Considerations following CAR T-cell therapy. Clinical Journal of Oncology Nursing, 23(Suppl. 1), 42–48. https://doi.org/10.1188/19.CJON.S1.42-48
Callahan, C., Baniewicz, D., & Ely, B. (2017). CAR T-cell therapy: Pediatric patients with relapsed and refractory acute lymphoblastic leukemia. Clinical Journal of Oncology Nursing, 21(Suppl. 2), 22–28. https://doi.org/10.1188/17.CJON.S2.22-28
Callahan, C., Barry, A., Fooks-Parker, S., Smith, L., Baniewicz, D., & Hobbie, W. (2019). Pediatric survivorship: Considerations following CAR T-cell therapy. Clinical Journal of Oncology Nursing, 23(Suppl. 1), 35–41. https://doi.org/10.1188/19.CJON.S1.35-41
Coleman, M., Martin, P., Ruan, J., Furman, R., Niesvizky, R., Elstrom, R., . . . Leonard, J.P. (2008). Prednisone, etoposide, procarbazine, and cyclophosphamide (PEP-C) oral combination chemotherapy regimen for recurring/refractory lymphoma: Low-dose metronomic, multidrug therapy. Cancer, 112, 2228–2232. https://doi.org/10.1002/cncr.23422
Gupta, S., Alexander, S., Zupanec, S., Athale, U., Bassal, M., Edwards, E., . . . Krueger, J. (2018, December). High vs. low-intensity bridging chemotherapy in children with acute lymphoblastic leukemia awaiting chimeric antigen receptor T-cell therapy: A population-based study from Ontario, Canada [1410]. Poster presented at the American Society of Hematology annual meeting, San Diego, CA.
Jain, M.D., Chavez, J.C., Shah, B.D., Khimani, F., Lazaryan, A., Davila, M.L., . . . Locke, F.L. (2018, December). Radiation therapy as a bridging strategy for refractory diffuse large B cell lymphoma patients awaiting CAR T manufacturing of axicabtagene ciloleucel. Poster presented at the American Society of Hematology annual meeting, San Diego, CA.
Kite Pharma. (2017). Yescarta® (axicabtagene ciloleucel) [Package insert]. Retrieved from https://www.fda.gov/downloads/UCM581226.pdf
Lee, D.W., Santomasso, B.D., Locke, F.L., Ghobadi, A., Turtle, C.J., Brudno, J.N., . . . Neelapu, S.S. (2018). ASBMT consensus grading for cytokine release syndrome and neurological toxicity associated with immune effector cells. Biology of Blood and Marrow Transplantation. Advance online publication. https://doi.org/10.1016/j.bbmt.2018.12.758
Levine, B.L., Miskin, J., Wonnacott, K., & Keir, C. (2017). Global manufacturing of CAR T cell therapy. Molecular Therapy—Methods and Clinical Development, 4, 92–101. https://doi.org/10.1016/j.omtm.2016.12.006
Locke, F.L., Neelapu, S.S., Bartlett, N.L., Siddiqi, T., Chavez, J.C., Hosing, C.M., . . . Go, W.Y. (2017). Phase 1 results of ZUMA-1: A multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Molecular Therapy, 25, 285–295. https://doi.org/10.1016/j.ymthe.2016.10.020
Maude, S.L., Frey, N., Shaw, P.A., Aplenc, R., Barrett, D.M., Bunin, N.J., . . . Grupp, S.A. (2014). Chimeric antigen receptor T cells for sustained remissions in leukemia. New England Journal of Medicine, 371, 1507–1517. https://doi.org/10.1056/NEJMoa1407222
McConville, H., Harvey, M., Callahan, C., Motley, L., Difilippo, H., & White, C. (2017). CAR T-cell therapy effects: Review of procedures and patient education [Online exclusive]. Clinical Journal of Oncology Nursing, 21(3), E79–E86. https://doi.org/10.1188/17.CJON.E79-E86
McGuirk, J., Waller, E.K., Qayed, M., Abhyankar, S., Ericson, S., Holman, P., . . . Myers, G.D. (2017). Building blocks for institutional preparation of CTL019 delivery. Cytotherapy, 19, 1015–1024. https://doi.org/10.1016/j.jcyt.2017.06.001
Mounier, N., El Gnaoui, T., Tilly, H., Canioni, D., Sebban, C., Casasnovas, R.O., . . . Haioun, C. (2013). Rituximab plus gemcitabine and oxaliplatin in patients with refractory/relapsed diffuse large B-cell lymphoma who are not candidates for high-dose therapy. A phase II lymphoma study association trial. Haematologica, 98, 1726–1731. https://doi.org/10.3324/haematol.2013.090597
Neelapu, S.S., Locke, F.L., Bartlett, N.L., Lekakis, L.J., Miklos, D.B., Jacobson, C.A., . . . Go, W.Y. (2017). Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. New England Journal of Medicine, 377, 2531–2544. https://doi.org/10.1056/NEJMoa1707447
Novartis Pharmaceuticals. (2018a). Find a Kymriah® treatment center. Retrieved from https://www.us.kymriah.com/treatment-center-locator/
Novartis Pharmaceuticals. (2018b). Kymriah® (tisagenlecleucel) [Package insert]. Retrieved from https://www.fda.gov/downloads/UCM573941.pdf
Novartis Pharmaceuticals. (2018c). Kymriah® (tisagenlecleucel) Risk Evaluation and Mitigation Strategy (REMS). Retrieved from http://www.kymriah-rems.com
Park, J.H., Rivière, I., Gonen, M., Wang, X., Sénéchal, B., Curran, K.J., . . . Sadelain, M. (2018). Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. New England Journal of Medicine, 378, 449–459. https://doi.org/10.1056/NEJMoa1709919
Perica, K., Curran, K.J., Brentjens, R.J., & Giralt, S.A. (2018). Building a CAR garage: Preparing for the delivery of commercial CAR T cell products at Memorial Sloan Kettering Cancer Center. Biology of Blood and Marrow Transplantation, 24, 1135–1141. https://doi.org/10.1016/j.bbmt.2018.02.018
Riley, S., & Riley, C. (2016). The role of patient navigation in improving the value of oncology care. Journal of Clinical Pathways, 2, 41–47.
Ruella, M., & Maus, M.V. (2016). Catch me if you can: Leukemia escape after CD19-directed T cell immunotherapies. Computional and Structural Biotechnology Journal, 14, 357–362. https://doi.org/10.1016/j.csbj.2016.09.003
Schuster, S.J., Bishop, M.R., Tam, C.S., Waller, E.K., Borchmann, P., McGuirk, J.P., . . . Maziarz, R.T. (2019). Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. New England Journal of Medicine, 380, 45–56. https://doi.org/10.1056/NEJMoa1804980
Stenzinger, M., & Bonig, H. (2018). Risks of leukapheresis and how to manage them—A non-systematic review. Transfusion and Apheresis Science, 57, 628–634. https://doi.org/10.1016/j.transci.2018.09.008
Taylor, L., Rodriguez, E., Reese, A., & Anderson, K. (2019). Building a program: Implications for infrastructure, nursing education, and training for CAR T-cell therapy. Clinical Journal of Oncology Nursing, 23(Suppl. 1), 20–26. https://doi.org/10.1188/19.CJON.S1.20-26
Turtle, C.J., Hanafi, L.A., Berger, C., Hudecek, M., Pender, B., Robinson, E., . . . Maloney, D.G. (2016). Immunotherapy of non-Hodgkin’s lymphoma with a defined ratio of CD8+ and CD4+ CD19-specific chimeric antigen receptor-modified T cells. Science Translational Medicine, 8(355), 355ra116. https://doi.org/10.1126/scitranslmed.aaf8621
U.S. Food and Drug Administration. (2017a). FDA approval brings first gene therapy to the United States [Press release]. Retrieved from https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm574058.htm
U.S. Food and Drug Administration. (2017b). FDA approves CAR-T cell therapy to treat adults with certain types of large B-cell lymphoma [Press release]. Retrieved from https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm581216.htm
U.S. Food and Drug Administration. (2018). Long term follow-up after administration of human gene therapy products. Retrieved from https://www.fda.gov/downloads/BiologicsBloodVaccines/GuidanceCompliance…




