CAR T-Cell Therapy: Update on the State of the Science
Background: Chimeric antigen receptor (CAR) T-cell therapy leverages the power of the patient’s own immune system by serving as a bridge to connect genetically modified T cells to the surface antigens of tumor cells based on targeted ligands. Clinical trials have demonstrated compelling overall response and survival rates in individuals with B-cell malignancies. The current approved agents target CD19, an antigen commonly overexpressed in B-cell hematologic and other malignancies.
Objectives: This article provides information on the current state of the science related to commercially available CAR T-cell products and examines how CAR T-cell science is evolving.
Methods: An overview of pathophysiology, indications, and nursing implications of the currently approved CAR T-cell agents is presented. Future directions for CAR T-cell development and treatment indications are discussed.
Findings: Tisagenlecleucel (Kymriah®) and axicabtagene ciloleucel (Yescarta®) received approval in 2017 for the treatment of B-cell precursor acute lymphoblastic leukemia in pediatric and young adult patients, and relapsed or refractory diffuse large B-cell lymphoma after two or more lines of systemic therapy in adult patients, respectively. Additional indications have since been approved, and new agents are in development.
Jump to a section
The use of cellular therapies to treat cancer is a rapidly growing and promising new branch in the field of immunotherapy. Immune effector cell therapy is broadly defined as agents that can be leveraged to produce an immune response against targeted tumor cells, and currently include natural killer cells, cytotoxic T lymphocytes, regulatory T cells, dendritic cells, and engineered T-cell receptors (Maus & Nikiforow, 2017). Chimeric antigen receptor (CAR) T cells are described as a living drug; the use of genetically modified T cells, expanded ex vivo, to produce an antitumor effect (Fesnak, June, & Levine, 2016). Unlike many oncologic treatments, these cells are designed to target a single surface antigen, resulting in remarkable efficacy at eradicating malignancy. The efficacy of T cells is dependent on the interaction between the CAR T cell and this expressed target, allowing the CAR T cell to bind and initiate an immune response toward the underlying malignancy (Fesnak et al., 2016). Two agents directed toward CD19, a protein commonly overexpressed in many B-cell malignancies, received U.S. Food and Drug Administration (FDA) approval for the treatment of acute lymphoblastic leukemia (ALL) and diffuse large B-cell lymphoma (DLBCL) (Bach, Giralt, & Saltz, 2017; Bouchkouj et al., 2018). Additional studies are ongoing to explore the use of CAR T cells and other immune effector cells directed to hematologic and solid tumor malignancies, with increasing products and indications anticipated for approval (Jackson, Rafiq, & Brentjens, 2016; Newick, O’Brien, Moon, & Albelda, 2017). This article presents an evidence-based overview of the current state of the science, including the pathophysiology of CAR T-cell therapy, current and potential future indications, and implications for nursing practice.
Pathophysiology
The premise for CAR T-cell therapy lies in the interactions between the receptors on the surface of healthy T cells and the co-stimulatory ligands on the surface of the antigen-presenting or tumor cell (Fesnak et al., 2016). The co-stimulatory signal from the tumor serves as an on or off switch to the body’s innate T-cell response. T-cell receptors serve to either activate or inhibit that immune response, and cancer cells are inherently designed to overcome the body’s immune response to proliferate. CAR T cells consist of three modules, extracellular target binding, transmembrane, and intracellular signaling domain (Guedan et al., 2018). These modules work as a bridge, expressing targeted receptors that increase binding to particular types of tumor cells and further unleash the antitumor effect of the T cells that are specifically programmed to engage with these particular tumor cells (Fesnak et al., 2016). A graphical representation of this process can be found in Figure 1. 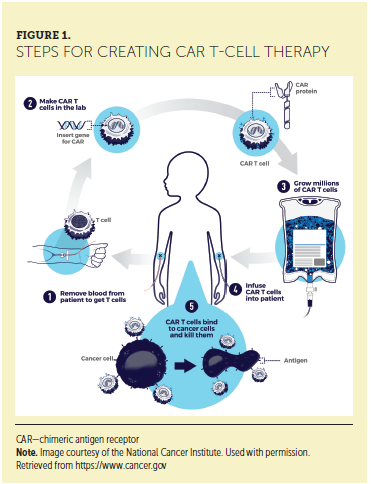
Several generations of CARs have been produced, seeking the ideal signaling domains to target to elicit the maximum antitumor effect. Early CARs were ligand-based and targeted a single signaling domain focused on the CD3z chain (Fesnak et al., 2016). Second-generation CARs have expanded to focus on co-stimulatory domains using single-chain fragment variable with an emphasis on CD28 or 4-1BB ligand (Fesnak et al., 2016). The single-chain fragment variable serves as a link, allowing the CAR to bind to the targeted surface antigen of the tumor cell to initiate the immune response (Fesnak et al., 2016). Third- and fourth-generation CARs are under review to evaluate how additional signaling domains may interact to produce an enhanced disease-specific antitumor effect.
Current Indications
Tisagenlecleucel (Kymriah®) received initial FDA approval in 2017 for the treatment of patients who are aged 25 years or younger with B-cell precursor ALL that is refractory or in second or later relapse (FDA, 2017a). This was followed by a 2018 approval for treatment of adult patients with relapsed or refractory DLBCL after two or more lines of systemic therapy (Novartis Pharmaceuticals, 2018a). In late 2017, FDA approval was granted for axicabtagene ciloleucel (Yescarta®) for the treatment of adult patients with relapsed or refractory DLBCL after two or more lines of systemic therapy (FDA, 2017b; Kite Pharma, 2017). Tisagenlecleucel and axicabtagene ciloleucel are CD19-directed autologous T-cell products. CD19 is the antigen expressed on the surface of B cells and overly expressed on malignant B cells (Maude, Teachey, Porter, & Grupp, 2015). The primary difference in the products is the co-stimulatory domain used to enhance expansion. Tisagenlecleucel uses the 4-1BB ligand (Bach et al., 2017), while axicabtagene ciloleucel uses the CD28 protein (Bouchkouj et al., 2018). Both co-stimulatory domains act in conjunction with the CD3z protein to activate the CAR T cells (Fesnak et al., 2016). Additional trials using these CD19-directed products for other B-cell malignancies are in progress (Jackson et al., 2016).
A product developed by Celgene (in collaboration with Bluebird Bio) for multiple myeloma, a disease with no known curative therapies, is expected to be the next FDA-approved CAR T-cell product. This product targets B-cell maturation antigen, which is expressed on the surface of plasma cells (Celgene, 2018).
Manufacturing Process
Manufacturing of autologous CAR T cells is a carefully defined and regulated process (Hollyman et al., 2009; Levine, Miskin, Wonnacott, & Keir, 2016; Wang & Rivière, 2016). The process for treating patients with these products is complex and can be time-intensive. The first step in the process is collecting T cells from the patient to be treated. This is accomplished via apheresis, during which blood is withdrawn from the body and separated using centrifuge (Levine et al., 2016). All blood components except the one desired—in this case the buffy coat—are then returned to the patient. The buffy coat, including the desired T cells, is then shipped to the processing facility. Clinical concerns for the patient during this stage of the process include large-bore IV access, pain, and infiltration. If peripheral large-bore access is not an option, a central venous catheter is needed (Allen et al., 2017) and may be preferred based on institutional policy. Once in the laboratory, the T cells from the patient’s product are isolated (Levine et al., 2016; Wang & Rivière, 2016). These cells are selected via exposure to anti–CD3/CD28-coated beads that trigger ex vivo T-cell activation (Levine et al., 2016; Wang & Rivière, 2016). Various selection options are available, including super-paramagnetic beads and biodegradable beads, which couple to CD3 and CD28 antibodies, allowing for the selection and activation of T cells (Wang & Rivière, 2016). The CAR gene is then attached to the patient’s T cells through incubation using an inactivated viral vector, which enters the T cell and introduces the genetically modified material (Levine et al., 2016; Wang & Rivière, 2016). Following development of the CAR T cells, they are expanded in vitro to generate a sufficient number of cells for effective treatment. Target cell volume is product specific and can involve weight-based dosing of the patient. After expansion, the beads are removed and the product is cryopreserved before being shipped back to the treating facility. This process, from collection of the autologous product to return of the CAR T-cell product, takes at least two weeks and is dependent on the rate of cellular reproduction to produce the replication-defective viral vector (Levine et al., 2016).
Because underlying disease and prior treatment can affect the quality of the collected lymphocytes, defined parameters for leukapheresis are followed (Allen et al., 2017). Leukapheresis should occur at least three months after previous stem cell transplantation, with recommended infusion of tisagenlecleucel at least six months after transplantation because of risk for alloreactive lymphocytes (Buechner, Kersten, Fuchs, Salmon, & Jäger, 2018). This time off treatment, combined with time needed for product manufacturing, can require bridge therapy for the patient’s disease between collection and CAR T-cell infusion because of the aggressive nature of the underlying malignancy (Beaupierre et al., 2019; Buechner et al., 2018; Shah et al., 2017). Steroids may also need to be limited during this period because of concern for their lympholytic properties, particularly T-cell suppression (Neelapu et al., 2018).
Prior to infusion of the CAR T-cell product, the patient receives lymphodepleting chemotherapy. Study results recommend a combination of cyclophosphamide and fludarabine, often via three daily doses of 250–500 mg/m2 and 25–30 mg/m2, respectively (Bot et al., 2015; Kochenderfer et al., 2017). The regimen is designed to deplete regulatory T cells to benefit the expansion of the CAR T-cell product (Wieczorek & Uharek, 2013). The cell product is infused approximately two days after completion of the lymphodepleting chemotherapy (Kochenderfer et al., 2017). Patients are premedicated according to institutional or protocol parameters, typically with acetaminophen and diphenhydramine, to reduce the risk for reaction to the product (Shank et al., 2017). Infusion of the product should occur within 30 minutes of thawing (Kite Pharma, 2017; Novartis Pharmaceuticals, 2018a). Once infused, CAR T cells continue to proliferate, reaching their peak expansion within two weeks postinfusion, with evidence of continued proliferation at one year or greater (Locke et al., 2017). This immune persistence has been associated with durable complete remission, allowing for continued immune surveillance by the activated T cells to promote eradication of current and potentially future malignancy (Guedan et al., 2018).
New Indications
The early successes of CAR T cells in ALL and DLBCL provided a pathway to explore other hematologic and nonhematologic malignant indications. By its mechanism of action, the efficacy and utility of a CAR T cell is dependent on identification of a target surface protein to which the CAR can be directed. Although early CAR T-cell trials have been directed at the CD19 and CD22 proteins on the B-cell surface known to be responsible for B-cell ALL and DLBCL, new target proteins are being evaluated (Jackson et al., 2016). To date, more than 180 clinical trials are ongoing, targeting proteins (CD19 and 22 or CD19 and 123) which are known to be overexpressed proteins on the surface of B cells responsible for leukemias and lymphomas (Leukemia and Lymphoma Society, 2018).
Of the hematologic malignant indications currently being evaluated, refractory or relapsed multiple myeloma holds the greatest promise. Late-stage clinical trials assessing the use of CARs directed at the B-cell maturing antigen protein, which is found on the surface of multiple myeloma cells, have demonstrated efficacy in achieving durable responses with a toxicity profile like those found in leukemia and lymphoma indications (Cho, Anderson, & Tai, 2018).
Solid tumors present a more complex array of surface proteins, which makes finding a common target for CARs more challenging. One common cell surface protein found in pancreatic, lung, ovarian, and mesothelioma tumors is mesothelin (Morello, Sadelain, & Adusumilli, 2016). Early-phase clinical trials are evaluating mesothelin-directed CAR T cells in this group of patients and has demonstrated some early efficacy and a tolerable safety profile. Larger, later-stage clinical studies for these indications are ongoing. In 2019, funding was awarded by the National Cancer Institute to broadly explore the application of CAR T-cell therapy in solid tumor malignancies, specifically lung cancer and mesothelioma (Albelda, 2019).
Other solid tumor indications currently in clinical trials include neuroblastoma, glioblastoma, gliomas, and hepatocellular carcinomas. These tumors have identified targetable antigens or protein expression, such as EGFRvIII, GD2, or glypican-3, that can be CAR-directed (D’Aloia, Zizzari, Sacchetti, Pierelli, & Alimandi, 2018). Additional studies evaluate the delivery methodology of CAR T-cell therapy. Although most of the CAR T-cell infusion products are delivered via IV, studies continue to evaluate intraventricular administration for brain tumor indications (Migliorini et al., 2018) and direct administration to the tumor site, such as for hepatocellular tumors (Newick et al., 2017).
Off-the-Shelf CARs
One of the key benefits of CAR T-cell therapy is its use of an autologous patient-specific product, eliminating the potential for graft-versus-host disease found with allogeneic infusions. However, this process is lengthy and involves multiple steps for collection and manufacturing. To make the treatment more readily accessible with greater production efficacy, alternative methods are being explored. Multiple academic and biotechnology laboratories are investigating the development of an off-the-shelf CAR T-cell product (Cooper et al., 2018; Poirot et al., 2015). This method involves creating a master cell bank derived from induced pluripotent stem cells that are grown from donor cells. Using gene-editing techniques, these blood cells are reprogrammed to become immature (undifferentiated) stem cells that provide a blank slate for creating an immune cell line that can be expanded and easily reproduced (Ruella & Kenderian, 2017). To avoid the immune response that would be expected by using donor cells, the T-cell receptor is removed during the gene-editing process, making these cells incapable of attacking recipient T cells. This process renders universal use of the new cells. These new T cells can be transduced to insert the desired CAR for the proposed disease indication or target and then cryopreserved and stored for ready access for patients.
Although the methodology of creating these off-the-shelf CAR T-cell products has demonstrated feasibility, the safety, efficacy, and comparability to existing autologous CAR T-cell products continues to be explored. To date, trials evaluating these new methods are still in early phases of clinical development. 
Implications for Nursing
As CAR T-cell therapy approvals expand agents and indications, as well as first line-therapy applications for disease progression, nurses will be expected to provide safe and effective patient care. Safe nursing practice is based on continuing education about these evolutions in care. Familiarization with the language of CAR T-cell therapy, including common terms associated with treatment, is important (see Table 1). Although CAR T-cell administration is currently limited to select centers throughout the United States, patients return to primary oncology and community-based care settings for follow-up or management of other acute and chronic illnesses. This requires education for nurses practicing in oncology and non-oncology settings, including emergency centers (Hanley, 2018), to ensure preparation to care for these individuals. Nurses in the apheresis setting are fundamental to the safe and effective collection of CD3-positive cells and should, therefore, be included in comprehensive education about the CAR T-cell collection process. For those administering the drug or managing patients during and immediately following administration, the FDA requires Risk Evaluation and Mitigations Strategy (REMS) education for each approved agent (Kite Pharma, 2018; Novartis Pharmaceuticals, 2018b). Nursing knowledge can translate to enhanced education for patients and their caregivers about what to expect throughout the CAR T-cell therapy process (McConville et al., 2017). Resources for nurse and patient education are listed in Figure 2. 
CAR T-cell therapy is FDA approved for treatment across the lifespan and, as such, care that is sensitive to the unique physiologic and psychosocial needs of patients across developmental stages is important. Family-based care is recommended, particularly in the pediatric population, to ensure the needs of the patient and the family caregivers are met (Callahan, Baniewicz, & Ely, 2017; Callahan et al., 2019). Establishing defined guidelines, standard operating procedures, and CAR T-cell–specific plans of care can also help to support transitions throughout the various stages of CAR T-cell therapy from cellular collection, through conditioning chemotherapy and CAR T-cell infusion, and ultimately through survivorship (Beaupierre et al., 2019; Buitrago, Adkins, Hawkins, Iyamu, & van Oort, 2019; Halton, Llerandi, Diamonte, Quintanilla, & Miale-Mayer, 2017; Taylor, Rodriguez, Reese, & Anderson, 2019). Toxicity management is a significant component of the care management and is well addressed in other articles (Anderson & Latchford, 2019; McConville et al., 2017). 
Conclusion
The efficacy of CAR T-cell therapy shows much promise. These products are characterized as living drugs because of their intrinsic mechanisms with the capacity for long-term persistence of the therapy. However, the same mechanisms of activation, expansion, and persistence are believed to be responsible for the potentially severe complications of cytokine release syndrome and neurologic toxicity often associated with these therapies. New and improved management techniques for these toxicities are receiving much attention in the literature as use of CAR T-cell therapy expands (Anderson & Latchford, 2019). Nurses are integral to the safe and effective care of these patients and can support best practice through continuing education as these therapies evolve.
About the Author(s)
Misty Lamprecht, MS, APRN-CNS, AOCN®, BMTCN®, is a clinical nurse specialist in the Blood and Marrow Transplant Program in the Arthur G. James Cancer Hospital and Richard J. Solove Research Institute at the Ohio State University in Columbus; and Colleen Dansereau, MSN, RN, CPN, is the director of clinical research nursing and director of clinical operations for the Gene Therapy Program at the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center in Boston, MA. The authors take full responsibility for this content. The supplement was supported by an independent educational grant from Bristol-Myers Squibb. Dansereau has previously served on advisory committees for Bluebird Bio and Novartis Pharmaceuticals. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Lamprecht can be reached at misty.lamprecht@osumc.edu, with copy to CJONEditor@ons.org. (Submitted January 2019. Accepted January 31, 2019.)
References
Albelda, S.M. (2019, January 2). NCI grant to fund study of CAR T-cell therapy for solid tumors. HemOnc Today. Retrieved from https://www.healio.com/hematology-oncology/lung-cancer/news/online/%7Be…
Allen, E.S., Stroncek, D.F., Ren, J., Eder, A.F., West, K.A., Fry, T.J., . . . Conry-Cantilena, C. (2017). Autologous lymphapheresis for the production of chimeric antigen receptor T cells. Transfusion, 57, 1133–1141. https://doi.org/10.1111/trf.14003
Bach, P.B., Giralt, S.A., & Saltz, L.B. (2017). FDA approval of tisagenlecleucel: Promise and complexities of a $475,000 cancer drug. JAMA, 318, 1861–1862.
Bot, A., Rossi, J.M., Jiang, Y., Navale, L., Shen, Y.W., Sherman, M., . . . Roberts, M.R. (2015, December). Cyclophosphamide and fludarabine conditioning chemotherapy induces a key homeostatic cytokine profile in patients prior to CAR T cell therapy [4426]. Paper presented at the American Society of Hematology 57th Annual Meeting and Exposition, Orlando, FL.
Bouchkouj, N., Kasamon, Y.L., de Claro, R.A., George, B., Lin, X., Lee, S., . . . Pazdur, R. (2018). FDA approval summary: Axicabtagene ciloleucel for relapsed or refractory large B-cell lymphoma. Clinical Cancer Research. Advance online publication. https://doi.org/10.1158/1078-0432.CCR-18-2743
Buechner, J., Kersten, M.J., Fuchs, M., Salmon, F., & Jäger, U. (2018). Chimeric antigen receptor-T cell therapy: Practical considerations for implementation in Europe. HemaSphere, 2(1), e18. https://doi.org/10.1097/HS9.0000000000000018
Callahan, C., Baniewicz, D., & Ely, B. (2017). CAR T-cell therapy: Pediatric patients with relapsed and refractory acute lymphoblastic leukemia. Clinical Journal of Oncology Nursing, 21(2, Suppl.), 22–28. https://doi.org/10.1188/17.CJON.S2.22-28
Celegene. (2018). bb2121 (Anti-BCMA CAR T). Retrieved from https://www.researchoncology.com/translational-research/bb2121-car-t
Cho, S.F., Anderson, K.C., & Tai, Y.T. (2018). Targeting B cell maturation antigen (BCMA) in multiple myeloma: Potential uses of BCMA-based immunotherapy. Frontiers in Immunology, 9, 1821. https://doi.org/10.3389/fimmu.2018.01821
Cooper, M.L., Choi, J., Staser, K., Ritchey, J.K., Devenport, J.M., Eckardt, K., . . . DiPersio, J.F. (2018). An “off-the-shelf” fratricide-resistant CAR-T for the treatment of T cell hematologic malignancies. Leukemia, 32, 1970–1983. https://doi.org/10.1038/s41375-018-0065-5
D’Aloia, M.M., Zizzari, I.G., Sacchetti, B., Pierelli, L., & Alimandi, M. (2018). CAR T-cells: The long and winding road to solid tumors. Cellular Death and Disease, 9, 282. https://doi.org/10.1038/s41419-018-0278-6
Fesnak, A.D., June, C.H., & Levine, B.L. (2016). Engineered T cells: The promise and challenges of cancer immunotherapy. Nature Reviews Cancer, 16, 566–581.
Foundation for the Accreditation of Cellular Therapy. (2018). FACT-JACIE international standards for hematopoietic cellular therapy product collection, processing and administration. Retrieved from http://www.factweb.org/forms/store/ProductFormPublic/seventh-edition-fa…
Guedan, S., Posey, A.D., Jr., Shaw, C., Wing, A., Da, T., Patel, P.R., . . . June, C.H. (2018). Enhancing CAR T cell persistence through ICOS and 4-1BB costimulation. JCI Insight, 3(1), 96976. https://doi.org/10.1172/jci.insight.96976
Halton, E., Llerandi, D., Diamonte, C., Quintanilla, H., & Miale-Mayer, D. (2017). Developing infrastructure: Managing patients with cancer undergoing CAR T-cell therapy. Clinical Journal of Oncology Nursing, 21(2, Suppl.), 35–40. https://doi.org/10.1188/17.CJON.2.35-40
Hanley, M. (2018). Increase in cancer patients receiving immunotherapy means ED nurses need to cautiously seek signs. ENA Connection. Retrieved from http://www.enaconnection-digital.org/enaconnection/june_2018/MobilePage…
Hollyman, D., Stefanski, J., Przybylowski, M., Bartido, S., Borquez-Ojeda, O., Taylor, C., . . . Rivière, I. (2009). Manufacturing validation of biologically functional T cells targeted to CD19 antigen for autologous adoptive cell therapy. Journal of Immunotherapy, 32, 169–180. https://doi.org/10.1097/CJI.0b013e318194a6e8
Jackson, H.J., Rafiq, S., & Brentjens, R.J. (2016). Driving CAR T-cells forward. Nature Reviews Clinical Oncology, 13, 370–383. https://doi.org/10.1038/nrclinonc.2016.36
Kite Pharma. (2017). Yescarta® (axicabtagene ciloleucel) [Package insert]. Retrieved from https://www.fda.gov/downloads/UCM581226.pdf
Kite Pharma. (2018). Yescarta® (axicabtagene ciloleucel) Risk Evaluation and Mitigation Strategy? Retrieved from https://www.yescartarems.com
Kochenderfer, J., Somerville, R., Lu, T., Shi, V., Yang, J.C., Sherry, R., . . . Rosenberg, S.A. (2017). Anti-CD19 chimeric antigen receptor T cells preceded by low-dose chemotherapy to induce remissions of advanced lymphoma [Abstract LBA3010]. Journal of Clinical Oncology, 34(18, Suppl.). https://doi.org/10.1200/JCO.2016.34.18_suppl.LBA3010
Leukemia and Lymphoma Society. (2018). Chimeric antigen receptor (CAR) T-cell therapy Retrieved from https://www.lls.org/treatment/types-of-treatment/immunotherapy/chimeric…
Levine, B.L., Miskin, J., Wonnacott, K., & Keir, C. (2016). Global manufacturing of CAR T cell therapy. Molecular Therapy. Methods and Clinical Development, 4, 92–101.
Locke, F.L., Neelapu, S.S., Bartlett, N.L., Siddiqi, T., Chavez, J.C., Hosing, C.M., . . . Go, W.Y. (2017). Phase 1 results of ZUMA-1: A multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Molecular Therapy, 25, 285–295. https://doi.org/10.1016/j.ymthe.2016.10.020
Maude, S.L., Teachey, D.T., Porter, D.L., & Grupp, S.A. (2015). CD19-targeted chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Blood, 125, 4017–4023. https://doi.org/10.1182/blood-2014-12-580068
Maus, M.V., & Nikiforow, S. (2017). The why, what, and how of the new FACT standards for immune effector cells. Journal for Immunotherapy of Cancer, 5, 36. https://doi.org/10.1186/s40425-017-0239-0
McConville, H. Harvey, M., Callahan, C., Motley, L., Difilippo, H., & White, C. (2017). CAR T-cell therapy effects: Review of procedures and patient education [Online exclusive]. Clinical Journal of Oncology Nursing, 21, E79–E86. https://doi.org/10.1188/17.CJON.E79-E86
Medical Dictionary. (n.d.). The free dictionary. Retrieved from https://medical-dictionary.thefreedictionary.com
Migliorini, D., Dietrich, P.Y., Stupp, R., Linette, G.P., Posey, A.D., Jr., & June, C.H. (2018). CAR T-cell therapies in glioblastoma: A first look. Clinical Cancer Research, 24, 535–540. https://doi.org/10.1158/1078-0432.CCR-17-2871
Morello, A., Sadelain, M., & Adusumilli, P.S. (2016). Mesothelin-targeted CARs: Driving T cells to solid tumors. Cancer Discovery, 6, 133–146. https://doi.org/10.1158/2159-8290.CD-15-0583
National Cancer Institute. (n.d.). Dictionary of cancer terms. Retrieved from https://www.cancer.gov/publications/dictionaries/cancer-terms
National Institutes of Health. (n.d.). Stem cell basics VI. Retrieved from https://stemcells.nih.gov/info/basics/6.htm
Neelapu, S.S., Tummala, S., Kebriaei, P., Wierda, W., Gutierrez, C., Locke, F.L., . . . Shpall, E.J. (2018). Chimeric antigen receptor T-cell therapy—assessment and management of toxicities. Nature Reviews. Clinical Oncology, 15, 47–62. https://doi.org/10.1038/nrclinonc.2017.148
Newick, K., O’Brien, S., Moon, E., & Albelda, S.M. (2017). CAR T cell therapy for solid tumors. Annual Review of Medicine, 68, 139–152. https://doi.org/10.1146/annurev-med-062315-120245
Novartis Pharmaceuticals. (2018a). Kymriah® (tisagenlecleucel) [Package insert]. Retrieved from https://www.fda.gov/downloads/UCM573941.pdf
Novartis Pharmaceuticals. (2018b). Kymriah® (tisagenlecleucel) Risk Evaluation and Mitigation Strategy (REMS). Retrieved from http://www.kymriah-rems.com
Poirot, L., Philip, B., Schiffer-Mannioui, C., Le Clerre, D., Chion-Sotinel, I., Derniame, S., . . . Smith, J. (2015). Multiplex genome edited T-cell manufacturing platform for “off-the-shelf” adoptive T-cell immunotherapies. Cancer Research, 75, 3853–3864. https://doi.org/10.1158/0008-5472.CAN-14-3321
Ruella, M., & Kenderian, S.S. (2017). Next-generation chimeric antigen receptor T-cell therapy: Going off the shelf. BioDrugs, 31, 473–481. https://doi.org/10.1007/s40259-017-0247-0
Shah, B., Stock, W., Wierda, W., Topp, M., Kersten, M.J., Houot, R., . . . Rajul, J. (2017). KTE-C19, an anti-CD19 chimeric antigen an anti-CD19 chimeric antigen receptor (CAR) T cell therapy in adult patients (Pts) with relapsed/refractory acute lymphoblastic leukemia (R/R ALL) in the ZUMA-3 trial: Preliminary results of novel safety interventions. Clinical Lymphoma, Myeloma and Leukemia, 17(Suppl. 2), S253. https://doi.org/10.1016/j.clml.2017.07.014
Shank, B.R., Do, B., Sevin, A., Chen, S.E., Neelapu, S.S., & Horowitz, S.B. (2017). Chimeric antigen receptor T cells in hematologic malignancies. Pharmacotherapy, 37, 334–345. https://doi.org/10.1002/phar.1900
U.S. Food and Drug Administration. (2017a). FDA approval brings first gene therapy to the United States [Press release]. Retrieved from https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm574058.htm
U.S. Food and Drug Administration. (2017b). FDA approves CAR-T cell therapy to treat adults with certain types of large B-cell lymphoma [Press release]. Retrieved from https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm581216.htm
Wang, X., & Rivière, I. (2016). Clinical manufacturing of CAR T cells: Foundation of a promising therapy. Molecular Therapy—Oncolytics, 3, 16015. https://doi.org/10.1038/mto.2016.15
Wieczorek, A., & Uharek, L. (2013). Genetically modified T cells for the treatment of malignant disease. Transfusion Medicine and Hemotherapy, 40, 388–402. https://doi.org/10.1159/000357163




