Renal, GI, and Peripheral Nerves: Evidence-Based Recommendations for the Management of Symptoms and Care for Patients With Multiple Myeloma
Background: A majority of patients with multiple myeloma experience damage to the kidneys and peripheral nerves either at diagnosis or throughout the disease. Symptoms of diarrhea, nausea, vomiting, or constipation can also occur. Prevention and management of disease- and treatment-related side effects are essential to treatment adherence.
Objectives: This article aims to provide insight into prevention and management of organ health–related toxicities and give best practice recommendations for optimal nursing care.
Methods: The authors performed a literature search, and articles were reviewed for relevance to each organ-related topic: renal, gastrointestinal, and peripheral nerves.
Findings: Prevention and management strategies include close monitoring and prompt intervention when organ damage develops to reverse impairment and maintain adherence to therapy when indicated.
Jump to a section
For most patients diagnosed with multiple myeloma (MM), organ damage involving the kidneys, bones, or peripheral nerves has occurred at diagnosis. Organ damage contributes to a high symptom burden and low health-related quality of life. Attention to organ health at diagnosis and throughout the disease continuum is important. Patients with MM, physicians, advanced practice providers, and nurses share the responsibility of monitoring, preventing, and addressing acute or chronic organ damage. Timely correction or reversal of existing organ damage, preservation of function, and management of symptoms arising from MM or its treatment are essential goals of MM nursing care. In this article, the International Myeloma Foundation (IMF) Nurse Leadership Board (NLB) aims to highlight important findings on MM- and treatment-related symptoms. The IMF NLB believes that nurses have the ability to identify patients at risk for organ damage as a result of MM or its treatment and to institute preventive and therapeutic interventions that can minimize patient morbidity. This article reviews the most recent, significant findings about MM- and treatment-related symptoms involving the kidney, gastrointestinal (GI) tract, and peripheral nerves. The article also provides state-of-the-art, evidence-based recommendations on management of these symptoms.
Methods
To obtain highly relevant literature on organ health in MM, the authors searched the PubMed and CINAHL® Complete databases using the Medical Subject Heading (MeSH) terms multiple myeloma, symptoms, and management and the Boolean operator AND (multiple myeloma AND symptoms AND management). The search yielded 31 articles from PubMed and 53 articles from CINAHL Complete. The following inclusion criteria were used in selecting the articles for final analysis: population of patients with MM; novel therapies; supportive therapies; and symptom management for renal, GI, and peripheral neuropathy (PN)–related symptoms because of MM or its treatment. The literature search was limited to articles published in English from January 2008 to March 2017.
The IMF NLB used Melnyk and Fineout-Overholt’s (2011) Levels of Evidence as a systematic framework for the appraisal and grading of the IMF NLB’s consensus statements and evidence-based recommendations. The rating system has seven levels that are inclusive of evidence from qualitative studies, case-control and cohort studies, reports of expert committees, and opinions of authorities in the field. This article reviews organ health related to MM, including renal, GI, and PN-related symptoms, and provides evidence-based recommendations to manage renal symptoms in the care of patients with MM.
Myeloma-Induced Renal Impairment
The kidneys are vital organs that filter the blood to remove waste materials, balance fluids and electrolytes, release hormones, and eliminate harmful chemicals from the body, including chemotherapeutic drugs. The terms renal impairment and renal insufficiency are often interchangeable and refer to the kidneys’ inability to function at their full capacity. Renal impairment is a relatively common presenting symptom among patients with MM and can occur throughout the duration of the disease. Mild renal insufficiency or renal failure occurs in about 50% of patients with MM at any time during the disease course (Finkel, Cohen, Shirali, & Abudayyeh, 2016; Gavriatopoulou, Terpos, Kastritis, & Dimopoulos, 2016). Multiple factors may affect renal function, including advancing age, development of new or worsening comorbid illnesses, MM disease burden, and cumulative toxicity from chemotherapy. Risk- and patient-related factors that may affect renal function should be assessed routinely at each clinic visit.
Adequate renal function in patients with MM must be safeguarded at all times because acute kidney injury (AKI), as evidenced by elevations in serum creatinine during a short period of time, is a significant factor in the development of chronic kidney disease and increases the risk of approaching end-stage renal disease and dialysis. (Hsu & Hsu, 2016). Therefore, prompt attention to new or worsening renal insufficiency and immediate identification and correction of the underlying cause are critical.
Clinical Presentation and Pathogenesis
Renal impairment may be acute or chronic and may be related to the disease or treatment. To classify AKI in adults, several important classification guidelines are used: RIFLE (risk, injury, failure, loss, and end-stage renal disease) system; the Acute Kidney Injury Network criteria; Kidney Disease: Improving Global Outcomes staging; and grades 0–3 staging (Dimopoulos, Sonneveld, et al., 2016; Thomas et al., 2015) (see Figure 1). Across each of these classification guidelines, the degree of renal impairment (regardless of whether it is acute or chronic) is determined by an increased serum creatinine level and reduced glomerular filtration rate (GFR) (Dimopoulos, Sonneveld, et al., 2016; Faiman, Mangan, Spong, & Tariman, 2011). 
Mild renal insufficiency is unlikely to produce symptoms, whereas anemia, fatigue, malaise, fluid and electrolyte imbalances, and decreased urination can occur with moderate to severe renal impairment. In patients who experience AKI, temporary elevations in serum creatinine or a sudden decline in GFR may be seen in patients who are dehydrated as a result of nausea, vomiting, infection, and decreased fluid volume intake. Figure 2 presents additional conditions or drugs that lead to acute renal insufficiency. Common medications used to treat MM and renal considerations can be found in Table 1. Acute or chronic renal impairment can occur at any time and clinical presentations largely depend on the etiology or pathogenesis (Hsu & Hsu, 2016). 
Distinctive Causes
Immunoglobulin light chain proteins are produced by malignant plasma cells in the bone marrow and eventually reach the nephrons where they combine with a potent immunomodulatory molecule, Tamm–Horsfall protein, and cause cast formation. Myeloma kidney (also called “cast nephropathy”) can be observed microscopically when crystalline precipitates of monoclonal light chains, surrounded by multinucleated giant cells, form casts in the distal and collecting tubules. These large, dense, tubular casts may obstruct and rupture the tubular epithelium, resulting in tubulointerstitial damage that manifests clinically as increased serum creatinine levels, electrolyte imbalance, and decreased GFR (Faiman et al., 2011).
[[{"type":"media","view_mode":"media_original","fid":"36016","field_deltas":{"1":{}},"link_text":null,"fields":{},"attributes":{"height":"768","width":"748","class":"media-image media-element file-media-original","data-delta":"1"}}]]
Diseases that promote glomerular injury, such as amyloid light chain amyloidosis or light chain deposition disease, should be considered in patients with MM when nephrotic-range proteinuria (defined as urine total protein levels greater than 3.5 g per 24 hours) occurs with or without significant renal impairment (Kelepouris & Rovin, 2017). Amyloid light chain amyloidosis is characterized by the deposition of fibrils consisting of monoclonal light chains in the glomeruli (Gertz, 2016). A diagnostic algorithm developed by Gertz (2016) underscores the importance of positive serum and urine immunofixation results and serum free light chain levels. A biopsy of the target organ (e.g., kidney, heart) is required if immunofixation electrophoresis of the serum or urine reveals monoclonal immunoglobulin light chains and if the clinical suspicion for amyloidosis is high (Brayer, Hussein, & Baz, 2017; Gertz, 2016). Tissue from kidney, bone marrow, or subcutaneous fat requires Congo red staining, which produces an apple-green birefringence when viewed under polarized light (Picken, 2015). The most accurate diagnosis of the presence and type of amyloidosis is obtained by mass spectroscopy. If this is not available, immunohistochemical staining is useful (Brayer et al., 2017). Determining the type of amyloid deposition patients have is essential because the selection of therapy depends on an accurate diagnosis (Picken, 2015). Light chain deposition disease is characterized by the presence of immunoglobulin light chains, usually kappa. In contrast to amyloidosis, these infiltrative light chains fail to stain with Congo red (Ardalan, 2013).
Acute Tubular Necrosis in Multiple Myeloma
Acute tubular necrosis is a common cause of acute renal impairment in MM. It may be precipitated by several factors, including dehydration or excessive diuretics, hypercalcemia (which occurs in as many as 10%–15% of patients), nonsteroidal anti-inflammatory drugs, and aminoglycoside antibiotics. Prompt diagnosis often leads to renal recovery (Doshi, Lahoti, Danesh, Batuman, & Sanders, 2016).
Contributors to Chronic Kidney Disease
Adequate kidney function is required for optimal bone repair and remodeling (Wei, Yin, & Xie, 2016). In patients with chronic kidney disease and MM, bone loss and bone damage may be profound long-term consequences of CKD. Bisphosphonates, such as zoledronic acid and pamidronate, are potent inhibitors of bone resorption. However, bisphosphonate use in patients with renal insufficiency or chronic kidney disease may be toxic to the kidneys (Berenson, 1997). Bisphosphonates are generally contraindicated in stage IV chronic kidney disease, which is characterized by GFR less than 30 ml per minute (Dimopoulos, Roussou, et al., 2016). In patients with hypercalcemia, nurses should ensure that patients with monoclonal light chains (also called Bence Jones proteinuria) drink enough fluids to produce 3 liters of urine daily. A diagnosis of primary hyperparathyroidism must be excluded in patients with persistent hypercalcemia because elevated serum parathyroid hormone levels are more common in the general population (Lewis, Hendrickson, & Moynihan, 2011).
Evidence-Based Recommendations for Acute Kidney Injury
Even mild renal insufficiency or impairment can place patients with MM at risk for chronic kidney disease. It is within the nurse’s scope of practice to assess and intervene when kidney health is at risk.
Level of Evidence I
• The IMF NLB recommends that immediate identification and treatment of reversible causes of AKI, such as dehydration, be corrected as quickly as possible. Adequate IV or oral hydration is essential (with caution to avoid overhydration) to help reverse AKI or renal failure (Moos, van Vemde, Stoker, & Bipat, 2013; Okusa & Rosner, 2017).
• Healthcare providers must be certain that dehydration is corrected prior to oral or IV iodinated contrast dye for radiologic procedures. The risk of AKI following the use of iodinated contrast dyes is increased in patients with chronic kidney disease, diabetes, malignancy, and heart failure, and in older adults (Moos et al., 2013; Mussap & Merlini, 2014).
• The IMF NLB recommends measurement of serum creatinine prior to any radiologic procedure that requires an iodinated contrast dye. Once a creatinine level is available, an estimated GFR can be calculated by using the Chronic Kidney Disease-Epidemiology (CKD-EPI) equation, which accounts for the patient’s age, creatinine level, gender, and race (National Institute of Diabetes and Digestive and Kidney Diseases [NIDDKD], 2015).
• The IMF NLB recommends that contrast-induced nephropathy risk assessment and prophylactic strategies should be based on estimated GFR using the CKD-EPI equation (Faiman et al., 2011) rather than solely relying on the absolute value of creatinine. The European Renal Best Practice Ad-Hoc Working Group on contrast-induced nephropathy (Fliser et al., 2012) and a study by Terpos et al. (2013) showed better detection of stages III–V chronic kidney disease (stage III with estimated GFR 30–60 ml per minute per 1.73 m2) in patients with newly diagnosed MM with the CKD-EPI equation than with the absolute creatinine value alone.
Level of Evidence II
• The IMF NLB recommends plasmapheresis in conjunction with anti-MM therapies and the use of high-cutoff dialyzers. Plasmapheresis removes damaging monoclonal light chains and has been shown to improve or reverse renal failure (Burnette, Leung, & Rajkumar, 2011; Clark, 2007; Finkel et al., 2016; Hutchison et al., 2007). The use of high-cutoff dialyzers may effectively remove serum free light chains and can lead to improved renal function (Finkel et al., 2016; Hutchison et al., 2007, 2009) or independence from hemodialysis among patients receiving a bortezomib-based regimen (Bridoux et al., 2016). Temporary or permanent hemodialysis is indicated if symptomatic uremia becomes an issue.
Evidence-Based Recommendations for Simultaneous Renal Impairment and Active Multiple Myeloma
Level of Evidence I
• The IMF NLB recommends prompt initiation of effective MM therapy. In line with International Myeloma Working Group recommendations that renal impairment should be treated with bortezomib-based regimens (Dimopoulos, Sonneveld, et al., 2016), the IMF NLB recommends the use of bortezomib as the cornerstone treatment in the management of these patients.
Level of Evidence I-III
• The IMF NLB recommends prompt treatment using classes of drugs approved by the U.S. Food and Drug Administration to treat patients with relapsed or refractory MM and renal impairment. These include immunomodulatory agents like the following:
– Level of evidence I: Thalidomide; proteasome inhibitors, such as bortezomib (Dimopoulos, Sonneveld, et al., 2016; Picot, Cooper, Bryant, & Clegg, 2011)
– Level of evidence III: Lenalidomide and pomalidomide; carfilzomib and ixazomib; monoclonal antibodies, such as elotuzumab and daratumumab
Evidence-Based Recommendations for Supportive Care
Level of Evidence I
• The IMF NLB recommends bisphosphonate therapy to prevent skeletal-related events, with caution paid to serum creatinine levels (Mhaskar et al., 2010). In patients with chronic kidney disease, the IMF NLB recommends monitoring of the serum creatinine level and estimated GFR using the CKD-EPI calculator (NIDDKD, 2015) prior to each dose, in addition to periodic monitoring with 24-hour urine to assess for albuminuria.
• The IMF NLB recommends that patients who are taking bisphosphonates should also take daily calcium and vitamin D supplementation. Recommendations vary according to vitamin D status and nutritional intake, but supplements of 600 mg calcium and 800 IU vitamin D daily are generally accepted (Body et al., 2016). Calcium supplementation remains contraindicated in patients with hypercalcemia (Body et al., 2016; Dimopoulos, Sonneveld, et al., 2016; Faiman et al., 2011; Mhaskar et al., 2010).
Level of Evidence III
• The IMF NLB recommends the use of erythropoiesis-stimulating agents (ESAs). Anemia, defined in patients with MM as having a hemoglobin value 2 g/dl below the institutional limits of normal, is often present in patients with moderate to severe renal dysfunction, as well as MM itself. The use of erythropoiesis-stimulating agents in renal disease is common practice. However, the use of these drugs in MM and chronic kidney disease remains controversial. An increased risk of stroke and thromboembolic events may occur in patients with MM receiving ESAs (Anaissie et al., 2012). Interventions to minimize this risk, such as venous thromboembolism precautions (e.g., ambulation, antiplatelet and anticoagulant therapy when appropriate), should be initiated prior to use of ESAs. According to the National Comprehensive Cancer Network (2016) clinical practice guidelines for patients with MM, chronic kidney disease is not a contraindication for receiving ESAs. Obtaining baseline serum iron, ferritin, and serum total iron binding capacity is recommended prior to initiating oral iron therapy (Khwaja, 2012).
Evidence-Based Recommendations for Monitoring of Disease Status and Renal Function
Level of Evidence V
• The IMF NLB recommends close monitoring of a patient’s MM disease burden and renal insufficiency by ensuring that they obtain routine complete blood count, blood chemistries, and measurement of monoclonal protein levels. In patients with stable disease, laboratory monitoring should be performed either monthly or according to institutional schedules for routine clinic follow-up. During laboratory monitoring, MM-specific biomarkers should include serum and urine protein electrophoresis, quantitative immunoglobulins, 24-hour urine for protein electrophoresis and serum free light chain assay (Kurtin, Bertolotti, Brigle, & Verina, 2016). The frequency of laboratory testing may change depending on the degree of renal failure and the patient’s response to therapy (Kurtin et al., 2016). See Figures 3 and 4 for renal care plans, including patient monitoring. 
Evidence-Based Recommendations for Patient Education
Level of Evidence V
• The IMF NLB recommends that patients and their caregivers be taught about the additional burden of certain comorbidities on renal functions and that comorbidities be monitored closely and managed effectively in partnership with patients, caregivers, and nephrologists to avoid any additional injury to the kidney. Patients with MM are at an increased risk of renal failure not only from MM itself, but also from aging, because MM occurs in older adults (Zweegman, Palumbo, Bringhen, & Sonneveld, 2014). Diabetes and hypertension are the leading causes of end-stage renal disease, and diabetes mellitus is the number one cause of kidney failure (United States Renal Data System, 2016). About half of all new patients receiving dialysis have diabetes, making it the fastest-growing risk factor for kidney disease. Uncontrolled hypertension is a cause and complication of chronic kidney disease and should be carefully treated and controlled in all patients (Song, Cong, & Wilson, 2016). Other factors that may increase the risk of renal failure include racial or ethnic minority status in the United States and family history of chronic kidney disease.
Level of Evidence VI
• The IMF NLB recommends that oncology clinicians advise all patients, including those with MM, to actively engage in weight control, exercise, smoking cessation, and taking medications for adequate blood pressure or blood sugar control. Adequate health maintenance and preventive screenings for hypertension, diabetes, obesity, and smoking-related health conditions may prevent or slow progression to kidney failure (Faiman et al., 2011; Tariman et al., 2016).
[[{"type":"media","view_mode":"media_original","fid":"36026","field_deltas":{"2":{}},"link_text":null,"fields":{},"attributes":{"height":"621","width":"746","class":"media-image media-element file-media-original","data-delta":"2"}}]]
Gastrointestinal Symptoms
GI symptoms are commonly experienced among patients who undergo active treatment of MM, particularly with novel therapies. Inadequately addressed symptoms, such as constipation, diarrhea, nausea, vomiting, or anorexia, can negatively affect the patient physically and psychologically. Adequate management of GI symptoms has the potential to increase patient adherence to treatment regimens, decrease physiologic impairment, and improve quality of life (Smith, Bertolotti, Curran, & Jenkins, 2008). The following evidence-based recommendations address the GI toxicities of constipation, diarrhea, nausea, and vomiting and will allow the healthcare provider, as part of an interprofessional team, to be better prepared to manage these toxicities associated with novel therapies for MM.
Clinical Presentations and Pathophysiology
The GI tract is a complex system responsible for the disintegration of solid foods, bioavailability of nutrients, and excretion of unnecessary waste (Faiman, 2016; Kong & Singh, 2008). Multiple factors can contribute to abnormal functioning of the GI tract and include disruption of the normal intestinal flora (by chemotherapy or autoimmunity) or changes to GI motility, which allows for excretion of foods (Nitzan, Elias, Chazan, Raz, & Saliba, 2013). For patients with MM, medications used for disease management or supportive care can interrupt the normal functioning of the GI system and lead to diarrhea, constipation, nausea, or vomiting.
The incidence of GI toxicities in patients with MM varies greatly with type of therapy. Diarrhea, defined as an abnormal increase in the amount of fluid in stool, is the most common GI symptom that patients with MM experience. Diarrhea can occur because of infection, inflammation, malignancy, or an autoimmune phenomenon (Faiman, 2016). Diarrhea is also an immediate side effect of bortezomib, ixazomib, and other agents prescribed for the treatment of MM. Late-onset diarrhea occurs in as many as 45% of individuals from exposure to oral lenalidomide, possibly because of an immune effect and a fairly common long-term side effect (Celgene Corporation, 2010; Faiman, 2016; Tariman, 2017). The consequences of diarrhea can be mild and transient or, for some, severe and life-threatening. Figure 5 presents the approach to assessment and differential diagnosis of diarrhea in patients with MM.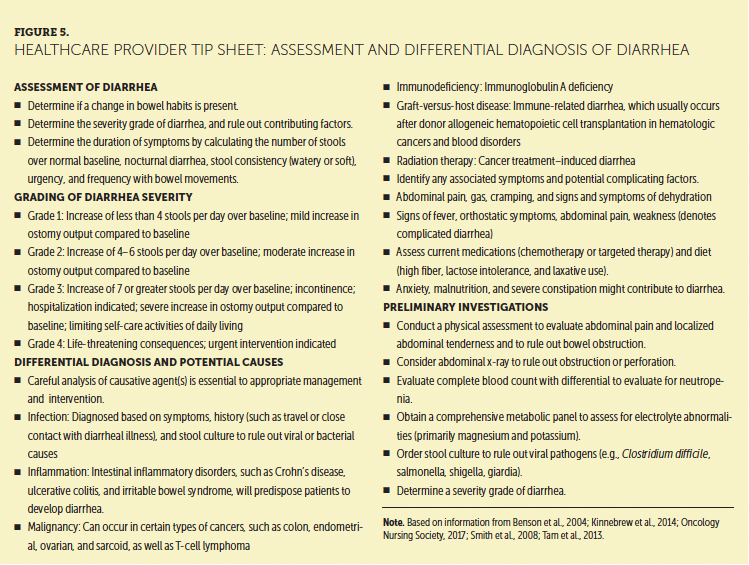
Constipation is defined as a decreased frequency of defecation, usually less than three bowel movements per week, with accompanying abdominal discomfort. This is a common problem in patients with cancer who have poor oral intake or take drugs like opioids or antiemetics that slow intestinal transit time (Smith et al., 2008).
Nausea is an uncomfortable feeling that occurs in the throat or stomach and may or may not result in vomiting. Acute nausea usually occurs within a few minutes to several hours after administration of anticancer agents and often resolves within the first 24 hours. Delayed nausea occurs more than 24 hours after administration of anticancer agents, peaks 48–72 hours after treatment, and can last 6–7 days. Patients may also experience anticipatory nausea, which is a conditioned response that occurs before they receive an anticancer treatment. Once a patient has been determined to have or to be at risk for nausea, it is appropriate to recommend prophylactic and therapeutic interventions and to implement an effective nausea management plan.
Some antiemetic therapies may be helpful for nausea and vomiting (see Figure 6). Antiemetics for older adult patients should have a low risk of drug–drug interactions and a simple, convenient dosing regimen (Smith et al., 2008). 
Vomiting is a separate act from nausea and often a result of an episode of nausea (Smith et al., 2008). Fortunately, outside of transplantation with high-dose chemotherapy, vomiting is rare among patients with MM who receive newer classes of drugs.
Anorexia and weight loss are common and unique GI side effects of an emerging therapy currently in clinical trials, selinexor (Vogl et al., 2016). Selinexor is a first-in-class exportin 1 (XPO1) inhibitor that induces nuclear retention and activation of tumor suppressor proteins and the glucocorticoid receptors and suppresses oncoprotein expression (Turner et al., 2016; Vogl et al., 2016). In a clinical trial, selinexor was found to be effective in combination with corticosteroids in patients who were no longer responding to treatment with five prior therapies (i.e., lenalidomide, bortezomib, carfilzomib, daratumumab, and isatuximab). Despite premedication with a 5-HT3 receptor antagonist, patients in the study experienced nausea (72% overall, 6% for grade 3), anorexia (49% overall, 3% for grade 3), vomiting (43% overall, 4% for grade 3), diarrhea (42% overall, 5% for grade 3), and weight loss (33% overall, 1% for grade 3) (Vogl et al., 2016). Although management of diarrhea, nausea, and vomiting has been associated with newer agents, it is important to emphasize that anorexia and weight loss with selinexor are unique side effects that can occur, and patients taking this drug should be effectively monitored.
Guidance for strategies to manage GI side effects of selinexor can be modeled from clinical trials experience. For patients who experience nausea, ensure they are taking proton pump inhibitor drugs, such as omeprazole, lansoprazole, or esomeprazole. For moderate to severe nausea, vomiting, anorexia, diarrhea, or weight loss, holding selinexor until the symptom improves and resuming at a lower dose level are recommended. Nurses can intervene if anorexia and weight loss are observed by advising patients to eat small, high-caloric meals consisting of food that is not greasy, acidic, fried, or spicy to minimize stomach upset. Patients can proceed to appetite stimulants if initial dietary interventions are not successful. As with any severe toxicity, such as weight loss of greater than 10% (can be more than 20–40 lbs depending on the size of the individual), dietary supplements for weight gain and holding of the drug until the symptom improves or resolves should be considered (Vogl et al., 2016).
Smith et al. (2008) previously presented patient characteristics and clinical features of GI complications in patients with MM treated with typical agents (http://bit.ly/2wEzJ2d). Figure 7 contains information for managing GI side effects with novel agents used to treat MM. 
Evidence-Based Recommendations for Gastrointestinal Symptoms
Level of Evidence II
• The IMF NLB recommends dose modifications of causative agents when GI symptoms from MM therapies, such as uncontrolled nausea, vomiting, diarrhea, constipation, and weight loss, persist despite employing best practices in preventive and supportive care.
• Selinexor represents a new class of drugs with weight loss, anorexia, nausea, vomiting, and diarrhea as common side effects. Optimizing antiemetic, antidiarrheal, and dietary strategies is the best recommended first-line management. If side effects persist and affect quality of life, holding or decreasing the dose of selinexor may allow patients to stay on therapy longer.
Peripheral Neuropathy Symptoms
PN is commonly cited as a major side effect of MM therapy that can negatively affect a patient’s quality of life (Richardson et al., 2012; Sonneveld et al., 2013). PN symptoms can occur in as many as 75% of individuals secondary to disease progression or after prolonged exposure to neurotoxic therapies, such as thalidomide and bortezomib (Richardson et al., 2012). Patients with PN experience a variety of symptoms, such as mild discomfort, loss of sensation, painful burning, muscle cramping, heightened sensitivity to temperature variations, impaired fine-motor skills, and paralysis in the most severe cases (Hershman et al., 2014; Richardson et al., 2012; Tariman, Love, McCullagh, & Sandifer, 2008). These deleterious effects can be cumulative over time and can cause significant morbidity, limit a patient’s ability to receive the appropriate doses of anticancer medication, and lead to decreased cancer treatment compliance (Kaplan, Torcun, Grune, Ozer, & Karademir, 2017). Paying attention to the drugs and conditions that can cause PN and preserving nerve function are critical (Richardson et al., 2012).
Clinical Presentations and Pathophysiology
To understand the pathology of PN, the normal neurophysiologic function of the peripheral nervous system must be reviewed. Each neuron consists of a cell body, dendrites that synapse with other nerves, and an axon that is often surrounded by Schwann cells that form a myelin sheath. The cell bodies of sensory neurons are bundled together within the dorsal root ganglia. Motor nerve cell bodies are in the ventral spinal cord. The motor and sensory neurons are responsible for different actions. Motor neurons carry signals from the central nervous system to muscles and glands. Large, myelinated sensory neurons are responsible for the perception of vibration, proprioception (body position), and light touch, whereas small, myelinated sensory neurons transmit impulses for temperature. Pain impulses can be transmitted by either small myelinated or unmyelinated nerve fibers (Poncelet, 1998).
PN occurs when peripheral nerves are damaged or inflamed or when degeneration has occurred, leading to changes in the way the nerves function (Ferrier, Pereira, Busserolles, Authier, & Balayssac, 2013). The mechanism of PN in MM is not well characterized but is thought to be because of a combination of direct damage to nerve cells, toxicity to the dorsal root ganglion, and decreased nerve blood flow (Ferrier et al., 2013; Richardson et al., 2012).
Diagnosis
There is no gold standard for the diagnosis or measurement of chemotherapy-induced PN; however, patient self-report of symptoms remains the most common diagnostic technique (Tofthagen & Irwin, 2014; Visovsky et al., 2012). Electromyography is a technique that involves the placement of a needle into various muscles to record different stages of muscle activity, including rest, contraction, and activity. This technique can be useful in detecting motor neuropathy but is less accurate in detecting the sensory changes most commonly encountered in MM; however, detection methods for PN, including electromyography, are lacking (Preston & Shapiro, 2005).
Nerve conduction studies can also be performed to diagnose PN and distinguish from other disorders. These studies measure the velocity and size of the signal along a nerve, allowing neurologists to evaluate nerve myelination and muscle contraction amplitude. This procedure can aid in categorizing the pathophysiology of the PN as demyelinating, axonal, or mixed (Preston & Shapiro, 2005).
PN assessment and monitoring are largely based on patient self-report of symptoms, which commonly include pain, numbness, tingling, or all three of those symptoms. The Functional Assessment of Cancer Therapy/Gynecologic Oncology Group–Neurotoxicity is an 11-item questionnaire that addresses patient-reported concerns likely to be associated with chemotherapy-induced PN (Calhoun et al., 2003; Cella et al., 1993; Cella & Webster, 1999). This instrument, with scores ranging from 0–44 on a continuous scale (with higher scores indicating greater impairment), has been validated in patients with neurotoxicity associated with ovarian cancer chemotherapy, and has been used in several MM studies (Richardson et al., 2012).
Evidence-Based Recommendations for Management of Peripheral Neuropathy
The IMF NLB has reviewed over-the-counter preventive and treatment strategies for PN. These recommendations can be found in Figure 8. 
Level of Evidence II
• The IMF NLB recommends dose reduction of the causative agent or a change to subcutaneous route of administration, and discontinuation of bortezomib with the onset of moderate PN (Richardson, Sonneveld, et al., 2009). To date, few interventions have been documented to prevent or treat neuropathy in patients with MM (Faiman et al., 2013; Moreau et al., 2011; Richardson et al., 2012; Richardson, Xie, et al., 2009; Takeda Pharmaceutical Company, 2016; Tsubaki et al., 2015; Velasco et al., 2010). However, dose reduction or discontinuation may come at the expense of inferior disease control.
Level of Evidence III
• The IMF NLB recommends to consider the use of glutamine for prevention of PN in patients with MM receiving a neurotoxic agent, such as bortezomib. Glutamine, a naturally occurring nonessential amino acid, is easy to use, well absorbed, and cost effective. Two key studies (Hall, Heel, & McCauley, 1996; Klimberg et al., 1992) support the safety and efficacy of oral glutamine in preventing cancer treatment–induced PN. The glutamine group had a lower incidence of severe (grades 3–4) PN than the placebo group and, therefore, less need to reduce the chemotherapy dose. In one small trial of patients with colorectal cancer, response rates for the glutamine and nonglutamine groups (52.4% and 47.8%, respectively) were not significantly different, suggesting that glutamine does not interact with the efficacy of the chemotherapy regimen (Wang et al., 2007). A second randomized pilot study of oral glutamine in MM demonstrated a medium effect size (0.43) of glutamine on neuropathy symptoms and a large effect on quality of life (1.09). In this study, diarrhea and nausea were mild (grade 1) in both study groups. Drug-related grade 3 or 4 toxicities did not occur in any study participant (Faiman, Zhang, Moore, Daly, & Reu, 2015).
• The IMF NLB recommends the use of the most widely studied over-the-counter supplements with varying suggested benefits for CIPN. In addition to glutamine, these include B vitamins, magnesium, potassium, and glutathione (Cascinu et al., 2002; Hershman et al., 2014; Vahdat et al., 2001), alpha-lipoic acid (Ziegler, Nowak, Kempler, Vargha, & Low, 2004); acetyl-L-carnitine (Bianchi et al., 2005); and vitamin E (Pace et al., 2003). These supplements may have activity in the prevention and treatment of PN. 
Conclusion
Symptoms related to renal, GI, or peripheral nerve organ damage in patients with MM should be assessed and treated promptly if they are present at initial diagnosis and throughout therapy. Preventive measures should be initiated at diagnosis and continue throughout the disease course. Evidence-based recommendations for the management of organ damage–related symptoms should be updated as the need arises. The prevention of acute and long-term organ damage gives patients with MM more therapeutic options and offers them the opportunity to achieve a longer overall survival and a better quality of life.
The authors gratefully acknowledge Robert Kyle, MD, Brian G.M. Durie, MD, and Diane P. Moran, RN, MA, EdM, at the International Myeloma Foundation for their review of this manuscript.
About the Author(s)
Beth Faiman, PhD, MSN, APRN-BC, AOCN®, is a nurse practitioner in the Department of Hematology and Medical Oncology at the Cleveland Clinic Taussig Cancer Institute in Ohio; Deborah Doss, RN, OCN®, is a multiple myeloma research nurse and Kathleen Colson, RN, BSN, BS, is a clinical research nurse, both at the Dana-Farber Cancer Institute in Boston, MA; Patricia Mangan, RN, MSN, APRN-BC, is a nurse lead of the hematologic malignancies program at the Abramson Cancer Center of the University of Pennsylvania in Philadelphia; Tracy King, RN, MN, is a myeloma clinical nurse consultant at the Sydney Local Health District in Australia; and Joseph D. Tariman, PhD, RN, ANP-BC, FAAN, is an assistant professor in the School of Nursing at DePaul University in Chicago, IL. The authors take full responsibility for this content. This supplement was supported by the International Myeloma Foundation, with funding from Celgene Corporation, Karyopharm Therapeutics, and Takeda Oncology. Writing and editorial support was provided by Eubio Medical Communications. Faiman consults and serves on speakers bureaus for Amgen, Bristol-Myers Squibb, Celgene Corporation, and Takeda Oncology, and has received support from Celgene Corporation and Takeda Oncology. Doss has previously served on speakers bureaus for Celgene Corporation, Takeda Pharmaceuticals, and Bristol-Myers Squibb, and has received additional support from Bristol-Myers Squibb and Celgene Corporation. Colson has previously consulted for Celgene Corporation and the International Myeloma Foundation Advisory Board, has served on speakers bureaus for the National Comprehensive Cancer Network, Multiple Myeloma Research Foundation, and Axis Medical. Mangan has previously consulted for Kite Pharmaceuticals and serves on speakers bureaus for Amgen, Celgene Corporation, and Takeda Oncology. King has previously served on speakers bureaus for Janssen India. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Faiman can be reached at faimanb@ccf.org, with copy to CJONEditor@ons.org. (Submitted June 2017. Accepted July 20, 2017.)
References
Amgen. (2016). Kyprolis® (carfilzomib) [Package insert]. Retrieved from http://pi.amgen.com/~/media/amgen/repositorysites/pi-amgen-com/kyprolis…
Anaissie, E.J., Coleman, E.A., Goodwin, J.A., Kennedy, R.L., Lockhart, K.D., Stewart, C.B., . . . Barlogie, B. (2012). Prophylactic recombinant erythropoietin therapy and thalidomide are predictors of venous thromboembolism in patients with multiple myeloma: Limited effectiveness of thromboprophylaxis. Cancer, 118, 549–557. https://doi.org/10.1002/cncr.26302
Ardalan, M.R. (2013). Light chain deposition disease; there are reasons for confusion. Journal of Renal Injury Prevention, 2, 127–128. https://doi.org/10.12861/jrip.2013.41
Badros, A.Z., Vij, R., Martin, T., Zonder, J.A., Kunkel, L., Wang, Z., . . . Niesvizky, R. (2013). Carfilzomib in multiple myeloma patients with renal impairment: Pharmacokinetics and safety. Leukemia, 27, 1707–1714. https://doi.org/10.1038/leu.2013.29
Benson, A.B., III, Ajani, J.A., Catalano, R.B., Engelking, C., Komblau, S.M., Martenson, J.A., Jr., . . . Wadler, S. (2004). Recommended guidelines for the treatment of cancer treatment-induced diarrhea. Journal of Clinical Oncology, 22, 2918–2926.
Berenson, J.R. (1997). Bisphosphonates in multiple myeloma. Cancer, 80(Suppl. 8), 1661–1667. https://doi.org/10.1002/(SICI)1097-0142(19971015)80:8+<1661::AID-CNCR16…
Bianchi, G., Vitali, G., Caraceni, A., Ravaglia, S., Capri, G., Cundari, S., . . . Gianni, L. (2005). Symptomatic and neurophysiological responses of paclitaxel- or cisplatin-induced neuropathy to oral acetyl-L-carnitine. European Journal of Cancer, 41, 1746–1750. https://doi.org/10.1016/j.ejca.2005.04.028
Body, J.J., Terpos, E., Tombal, B., Hadji, P., Arif, A., Young, A., . . . Coleman, R. (2016). Bone health in the elderly cancer patient: A SIOG position paper. Cancer Treatment Reviews, 51, 46–53. https://doi.org/10.1016/j.ctrv.2016.10.004
Brayer, J., Hussein, M.A., & Baz, R. (2017) Uncommon presentations of plasma cell dyscrasias. In D. Raghavan, M.S. Ahluwalia, C.D.Blanke, J. Brown, E.S. Kim, G.H. Reaman, & M.A. Sekeres (Eds.), Textbook of uncommon cancer (5th ed., pp. 776–792). Hoboken, NJ: Wiley-Blackwell.
Bridoux, F., Pegourie, B., Augeul-Meunier, K., Royer, B., Joly, B., Lamy, T., . . . Fermand, J.P. (2016). Treatment of myeloma cast nephropathy (MCN): A randomized trial comparing intensive haemodialysis (HD) with high cut-off (HCO) or standard high-flux dialyzer in patients receiving a bortezomib-based regimen: The MYRE Study, by Intergroupe Francophone du Myélome (IFM) and the French Society of Nephrology (SFNDT). Blood, 128, 978. Retrieved from http://www.bloodjournal.org/content/128/22/978
Burnette, B.L., Leung, N., & Rajkumar, S.V. (2011). Renal improvement in myeloma with bortezomib plus plasma exchange. New England Journal of Medicine, 364, 2365–2366. https://doi.org/10.1056/NEJMc1101834
Calhoun, E.A., Welshman, E.E., Chang, C.H., Lurain, J.R., Fishman, D.A., Hunt, T.L., & Cella, D. (2003). Psychometric evaluation of the Functional Assessment of Cancer Therapy/Gynecologic Oncology Group-Neurotoxicity (Fact/GOG-Ntx) questionnaire for patients receiving systemic chemotherapy. International Journal of Gynecological Cancer, 13, 741–748.
Cascinu, S. Catalano, V., Cordella, L., Labianca, R., Giordani, P., Baldelli, A.M., . . . Catalano, G. (2002). Neuroprotective effect of reduced glutathione on oxaliplatin-based chemotherapy in advanced colorectal cancer: A randomized, double-blind, placebo-controlled trial. Journal of Clinical Oncology, 20, 3478–3483. https://doi.org/10.1200/jco.2002.07.061
Chanan-Khan, A.A., Kaufman, J.L., Mehta, J., Richardson, P.G., Miller, K.C., Lonial, S., . . . Singhal, S. (2007). Activity and safety of bortezomib in multiple myeloma patients with advanced renal failure: A multicenter retrospective study. Blood, 109, 2604–2606. https://doi.org/10.1182/blood-2006-09-046409
Celgene Corporation. (2010). Revlimid® (lenalidomide) [Package insert]. Summit, NJ: Author. Retrieved from http://www.revlimid.com/wp-content/uploads/full-prescribing-information…
Cella, D., & Webster, K.A. (1999). Quality of life and treatment value in the management of hematologic malignancies. Seminars in Oncology, 26(5, Suppl. 14), 34–42.
Cella, D.F., Tulsky, D.S., Gray, G., Sarafian, B., Linn, E., Bonomi, A., . . . Brannon, J. (1993). The Functional Assessment of Cancer Therapy scale: Development and validation of the general measure. Journal of Clinical Oncology, 11, 570–579. https://doi.org/10.1200/JCO.1993.11.3.570
Clark, W.F. (2007). Correction: Plasma exchange when myeloma presents as acute renal failure. Annals of Internal Medicine, 146, 471.
Conticello, C., Parisi, M., Romano, A., Calafiore, V., Ancora, F., La Fauci, A., . . . Di Raimondo, F. (2017). Pomalidomide experience: An effective therapeutic approach with immunomodulatory drugs in a patient with relapsed-refractory multiple myeloma. Future Oncology, 13(5s), 3–6. https://doi.org/10.2217/fon-2016-0368
Dimopoulos, M.A., Cheung, M.C., Roussel, M., Liu, T., Gamberi, B., Kolb, B., . . . Facon, T. (2016). Impact of renal impairment on outcomes with lenalidomide and dexamethasone treatment in the FIRST trial, a randomized, open-label phase 3 trial in transplant-ineligible patients with multiple myeloma. Haematologica, 101, 363–370. https://doi.org/10.3324/haematol.2015.133629
Dimopoulos, M.A., Roussou, M., Gavriatopoulou, M., Psimenou, E., Eleutherakis-Papaiakovou, E., Migkou, M., . . . Kastritis, E. (2016). Bortezomib-based triplets are associated with a high probability of dialysis independence and rapid renal recovery in newly diagnosed myeloma patients with severe renal failure or those requiring dialysis. American Journal of Hematology, 91, 499–502. https://doi.org/10.1002/ajh.24335
Dimopoulos, M.A., Sonneveld, P., Leung, N., Merlini, G., Ludwig, H., Kastritis, E., . . . Terpos, E. (2016). International Myeloma Working Group recommendations for the diagnosis and management of myeloma-related renal impairment. Journal of Clinical Oncology, 34, 1544–1557. https://doi.org/10.1200/JCO.2015.65.0044
Doshi, M., Lahoti, A., Danesh, F.R., Batuman, V., & Sanders, P.W. (2016). Paraprotein–related kidney disease: Kidney injury from paraproteins—What determines the site of injury? Clinical Journal of the American Society of Nephrology, 11, 2288–2294. https://doi.org/10.2215/cjn.02560316
Faiman, B. (2016). Diarrhea in multiple myeloma: A review of the literature [Online exclusive]. Clinical Journal of Oncology Nursing, 20, E100–E105. https://doi.org/10.1188/16.CJON.E100-E105
Faiman, B., Elson, P., Smith, M., Dean, R.M., Valent, J., Samaras, C., . . . Bruening, K. (2013). Neuropathy and efficacy of weekly subcutaneous bortezomib in myeloma and AL amyloidosis. Blood, 122, 1975. Retrieved from http://www.bloodjournal.org/content/122/21/1975
Faiman, B.M., Mangan, P., Spong, J., & Tariman, J.D. (2011). Renal complications in multiple myeloma and related disorders: Survivorship care plan of the International Myeloma Foundation Nurse Leadership Board. Clinical Journal of Oncology Nursing, 15(Suppl.), 66–76. https://doi.org/10.1188/11.CJON.S1.66-76
Faiman, B., Zhang, A., Moore, S., Daly, B., & Reu, F.J. (2015). A pilot placebo controlled study to estimate the effect size of glutamine to prevent peripheral neuropathy in multiple myeloma. Clinical Lymphoma, Myeloma and Leukemia, 15(Suppl. 3), e318. https://doi.org/10.1016/j.clml.2015.08.091
Ferrier, J., Pereira, V., Busserolles, J., Authier, N., & Balayssac, D. (2013). Emerging trends in understanding chemotherapy-induced peripheral neuropathy. Current Pain and Headache Reports, 17(10), 364. https://doi.org/10.1007/s11916-013-0364-5
Finkel, K.W., Cohen, E.P., Shirali, A., & Abudayyeh, A. (2016). Paraprotein-related kidney disease: Evaluation and treatment of myeloma cast nephropathy. Clinical Journal of the American Society of Nephrology, 11, 2273–2279. https://doi.org/10.2215/cjn.01640216
Fliser, D., Laville, M., Covic, A., Fouque, D., Vanholder, R., Juillard, L., & Van Biesen, W. (2012). A European Renal Best Practice (ERBP) position statement on the kidney disease improving global outcomes (KDIGO) clinical practice guidelines on acute kidney injury: Part 1: Definitions, conservative management and contrast-induced nephropathy. Nephrology Dialysis, Transplantation, 27, 4263–4272. https://doi.org/10.1093/ndt/gfs375
Gavriatopoulou, M., Terpos, E., Kastritis, E., & Dimopoulos, M.A. (2016). Current treatments for renal failure due to multiple myeloma. Expert Opinion on Pharmacotherapy, 17, 2165–2177. https://doi.org/10.1080/14656566.2016.1236915
Gertz, M.A. (2016). Immunoglobulin light chain amyloidosis: 2016 update on diagnosis, prognosis, and treatment. American Journal of Hematology, 91, 947–956. https://doi.org/10.1002/ajh.24433
Hall, J.C., Heel, K., & McCauley, R. (1996). Glutamine. British Journal of Surgery, 83, 305–312.
Hershman, D.L., Lacchetti, C., Dworkin, R.H., Lavoie Smith, E.M., Bleeker, J., Cavaletti, G., . . . Loprinzi, C.L. (2014). Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. Journal of Clinical Oncology, 32, 1941–1967. https://doi.org/10.1200/JCO.2013.54.0914
Hingorani, S. (2016). Renal complications of hematopoietic-cell transplantation. New England Journal of Medicine, 374, 2256–2267. https://doi.org/10.1056/NEJMra1404711
Hsu, R.K., & Hsu, C.Y. (2016). The role of acute kidney injury in chronic kidney disease. Seminars in Nephrology, 36, 283–292. https://doi.org/10.1016/j.semnephrol.2016.05.005
Hutchison, C.A., Bradwell, A.R., Cook, M., Basnayake, K., Basu, S., Harding, S., . . . Cockwell, P. (2009). Treatment of acute renal failure secondary to multiple myeloma with chemotherapy and extended high cut-off hemodialysis. Clinical Journal of the American Society of Nephrology, 4, 745–754. https://doi.org/10.2215/cjn.04590908
Hutchison, C.A., Cockwell, P. Reid, S., Chandler, K., Mead, G.P., Harrison, J., . . . Bradwell, A.R. (2007). Efficient removal of immunoglobulin free light chains by hemodialysis for multiple myeloma: In vitro and in vivo studies. Journal of the American Society of Nephrology, 18, 886–895. https://doi.org/10.1681/asn.2006080821
Jakubowiak, A., Offidani, M., Pégourie, B., De La Rubia, J., Garderet, L., Laribi, K., . . . Plaumbo, A. (2016). Randomized phase 2 study: Elotuzumab plus bortezomib/dexamethasone vs bortezomib/dexamethasone for relapsed/refractory MM. Blood, 127, 2833–2840. https://doi.org/10.1182/blood-2016-01-694604
Kaplan, G.S., Torcun, C.C., Grune, T., Ozer, N.K., & Karademir, B. (2017). Proteasome inhibitors in cancer therapy: Treatment regimen and peripheral neuropathy as a side effect. Free Radical Biology and Medicine, 103, 1–13. https://doi.org/10.1016/j.freeradbiomed.2016.12.007
Kelepouris, E. & Rovin, B.H. (2017). Overview of heavy proteinuria and the nephrotic syndrome. UpToDate®. Retrieved from https://www.uptodate.com/contents/overview-of-heavy-proteinuria-and-the…
Khwaja, A. (2012). KDIGO clinical practice guidelines for acute kidney injury. Nephron Clinical Practice, 120(4), c179–c184. https://doi.org/10.1159/000339789
Kim, S.J., Kim, K., Do, Y.R., Bae, S.H., Yang, D.H., & Lee, J.J. (2011). Low-dose acyclovir is effective for prevention of herpes zoster in myeloma patients treated with bortezomib: A report from the Korean Multiple Myeloma Working Party (KMMWP) retrospective study. Japanese Journal of Clinical Oncology, 41, 353–357. https://doi.org/10.1093/jjco/hyq194
Kinnebrew, M.A., Lee, Y.J., Jenq, R.R., Lipuma, L., Littmann, E.R., Gobourne, A., . . . Taur, Y. (2014). Early clostridium difficile infection during allogeneic hematopoietic stem cell transplantation. PLOS ONE, 9(3), e90158. https://doi.org/10.1371/journal.pone.0090158
Klimberg, V.S., Pappas, A.A., Nwokedi, E., Jensen, J.C., Broadwater, J.R., Lang, N.P., & Westbrook, K.C. (1992). Effect of supplemental dietary glutamine on methotrexate concentrations in tumors. Archives of Surgery, 127, 1317–1320. https://doi.org/10.1001/archsurg.1992.01420110063013
Kong, F., & Singh, R.P. (2008). Disintegration of solid foods in human stomach. Journal of Food Science, 73(5), R67–R80. https://doi.org/10.1111/j.1750-3841.2008.00766.x
Kurtin, S., Bertolotti, P., Brigle, K., & Verina, D. (2016). Updates in the diagnosis and monitoring of multiple myeloma. Journal of the Advanced Practitioner in Oncology, 7, 59–70. https://doi.org/10.6004/jadpro.2016.7.2.15
Kwon, D., & Niesvizky, R. (2013). Treatment of multiple myeloma with carfilzomib in patients with renal injury. Clinical Advances in Hematology and Oncology, 11, 605–606.
Laubach, J.P., Paba Prada, C.E., Richardson, P.G., & Longo, D.L. (2017). Daratumumab, elotuzumab, and the development of therapeutic monoclonal antibodies in multiple myeloma. Clinical Pharmacology and Therapeutics, 101, 81–88. https://doi.org/10.1002/cpt.550
Lewis, M.A., Hendrickson, A.W., & Moynihan, T.J. (2011). Oncologic emergencies: Pathophysiology, presentation, diagnosis, and treatment. CA: A Cancer Journal for Clinicians, 61, 287–314.
Libby, E.N., Becker, P.S., Burwick, N., Green, D.J., Holmberg, L., & Bensinger, W.. (2015). Panobinostat: A review of trial results and future prospects in multiple myeloma. Expert Review of Hematology, 8, 9–18. https://doi.org/10.1586/17474086.2015.983065
Lonial, S., Dimopoulos, M., Palumbo, A., White, D., Grosicki, S., Spicka, I., . . . Richardson, P. (2015). Elotuzumab therapy for relapsed or refractory multiple myeloma. New England Journal of Medicine, 373, 621–631. https://doi.org/10.1056/NEJMoa1505654
Mateos, M.V., Granell, M., Oriol, A., Martinez-Lopez, J., Blade, J., Hernandez, M.T., . . . San-Miguel, J. (2016). Elotuzumab in combination with thalidomide and low-dose dexamethasone: A phase 2 single-arm safety study in patients with relapsed/refractory multiple myeloma. British Journal of Haematology, 175, 448–456. https://doi.org/10.1111/bjh.14263
Melnyk, B.M., & Fineout-Overholt, E. (2011). Evidence-based practice in nursing and healthcare: A guide to best practice (2nd ed., p. 13). Philadelphia, PA: Lippincott Williams and Wilkins.
Mhaskar, R., Redzepovic, J., Wheatley, K., Clark, O.A., Miladinovic, B., Glasmacher, A., . . . Djubegovic, B. (2010). Bisphosphonates in multiple myeloma. Cochrane Database of Systematic Reviews, 3, CD003188. https://doi.org/10.1002/14651858.CD003188.pub2
Mikhael, J.R., Reeder, C.B., Libby, E.N., Costa, L.J., Bergsagel, P.L., Buadi, F., . . . Stewart, A.K. (2015). Phase Ib/II trial of CYKLONE (cyclophosphamide, carfilzomib, thalidomide and dexamethasone) for newly diagnosed myeloma. British Journal of Haematology, 169, 219–227. https://doi.org/10.1111/bjh.13296
Millennium Pharmaceuticals. (2017). Velcade® (bortezomib) [Package insert]. Cambridge, MA: Author. Retrieved from http://www.velcade.com/files/PDFs/VELCADE_PRESCRIBING_INFORMATION.pdf
Moos, S.I., van Vemde, D.N., Stoker, J., & Bipat, S. (2013). Contrast induced nephropathy in patients undergoing intravenous (IV) contrast enhanced computed tomography (CECT) and the relationship with risk factors: A meta-analysis. European Journal of Radiology, 82(9), e387–e399. https://doi.org/10.1016/j.ejrad.2013.04.029
Moreau, P., Pylypenko, H., Grosicki, S., Karamanesht, I., Leleu, X., Grishunina, M., . . . Harousseau, J.L. (2011). Subcutaneous versus intravenous administration of bortezomib in patients with relapsed multiple myeloma: A randomised, phase 3, non-inferiority study. Lancet Oncology, 12, 431–440. https://doi.org/10.1016/S1470-2045(11)70081-X
Mussap, M., & Merlini, G. (2014). Pathogenesis of renal failure in multiple myeloma: Any role of contrast media? BioMed Research International, 2014, 167125. https://doi.org/10.1155/2014/167125
National Cancer Institute. (2010). Common terminology criteria for adverse events [v.4.03]. Retrieved from http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_…
National Comprehensive Cancer Network. (2016). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Multiple myeloma [v.3.2017]. Retrieved from https://www.nccn.org/professionals/physician_gls/PDF/myeloma.pdf
National Institute of Diabetes and Digestive and Kidney Diseases. (2015). CKD-EPI adults (conventional units). Retrieved from https://www.niddk.nih.gov/health-information/health-communication-progr…
Nitzan, O., Elias, M., Chazan, B., Raz, R., & Saliba, W. (2013). Clostridium difficile and inflammatory bowel disease: Role in pathogenesis and implications in treatment. World Journal of Gastroenterology, 19, 7577–7585. https://doi.org/10.3748/wjg.v19.i43.7577
Novartis. (2015). FARYDAK® (panobinostat) [Package insert]. Retrieved from http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/205353s000lbl…
Okusa, M.D., & Rosner, M.H. (2017). Overview of the management of acute kidney injury (acute renal failure). UpToDate®. Retrieved from https://www.uptodate.com/contents/overview-of-the-management-of-acute-k…
Oncology Nursing Society. (2017). Chemotherapy-induced diarrhea. Retrieved from https://www.ons.org/practice-resources/pep/diarrhea/chemotherapy-induce…
Pace, A., Savarese, A., Picardo, M., Maresca, V., Pacetti, U., Del Monte, G., . . . Bove, L. (2003). Neuroprotective effect of vitamin E supplementation in patients treated with cisplatin chemotherapy. Journal of Clinical Oncology, 21, 927–931.
Palumbo, A., Ambrosini, M.T., Benevolo, G., Pregno, P., Pescosta, N., Callea, V., . . . Cavallo, F. (2007). Bortezomib, melphalan, prednisone, and thalidomide for relapsed multiple myeloma. Blood, 109, 2767–2772. https://doi.org/10.1182/blood-2006-08-042275
Palumbo, A., Bringhen, S., Larocca, A., Rossi, D., Di Raimondo, F., Magarotto, V., . . . Cavo, M. (2014). Bortezomib-melphalan-prednisone-thalidomide followed by maintenance with bortezomib-thalidomide compared with bortezomib-melphalan-prednisone for initial treatment of multiple myeloma: Updated follow-up and improved survival. Journal of Clinical Oncology, 32, 634–640 https://doi.org/10.1200/JCO.2013.52.0023
Palumbo, A., Chanan-Khan, A., Weisel, K., Nooka, A.K., Masszi, T., Beksac, M., . . . Sonneveld, P. (2016). Daratumumab, bortezomib, and dexamethasone for multiple myeloma. New England Journal of Medicine, 375, 754–766. https://doi.org/10.1056/NEJMoa1606038
Palumbo, A., Facon, T., Sonneveld, P., Bladè, J., Offidani, M., Gay, F., . . . Harousseau, J.L. (2008). Thalidomide for treatment of multiple myeloma: 10 years later. Blood, 111, 3968–3977. https://doi.org/10.1182/blood-2007-10-117457
Picken, M.M. (2015). Proteomics and mass spectrometry in the diagnosis of renal amyloidosis. Clinical Kidney Journal, 8, 665–672. https://doi.org/10.1093/ckj/sfv087
Picot, J., Cooper, K., Bryant, J., & Clegg, A.J. (2011). The clinical effectiveness and cost-effectiveness of bortezomib and thalidomide in combination regimens with an alkylating agent and a corticosteroid for the first-line treatment of multiple myeloma: A systematic review and economic evaluation. Health Technology Assessment, 15(41), 1–204. https://doi.org/10.3310/hta15410
Poncelet, A.N. (1998). An algorithm for the evaluation of peripheral neuropathy. American Family Physician, 57, 755–764.
Preston, D., & Shapiro, B. (2005). Electromyography and neuromuscular disorders: Clinical-electrophysiologic correlations (2nd ed.). Boston, MA: Butterworth-Heinemann.
Rajkumar, S.V., Blood, E., Vesole, D., Fonseca, R., & Greipp, P.R. (2006). Phase III clinical trial of thalidomide plus dexamethasone compared with dexamethasone alone in newly diagnosed multiple myeloma: A clinical trial coordinated by the Eastern Cooperative Oncology Group. Journal of Clinical Oncology, 24, 431–436. https://doi.org/10.1200/JCO.2005.03.0221
Richardson, P.G., Delforge, M., Beksac, M., Wen, P., Jongen, J.L., Sezer, O., . . . Sonneveld, P. (2012). Management of treatment-emergent peripheral neuropathy in multiple myeloma. Leukemia, 26, 595–608. https://doi.org/10.1038/leu.2011.346
Richardson, P.G., Hungria, V.T., Yoon, S.S., Beksac, M., Dimopoulos, M.A., Elghandour, A., . . . San-Miguel, J.F. (2016). Panobinostat plus bortezomib and dexamethasone in previously treated multiple myeloma: Outcomes by prior treatment. Blood, 127, 713–721. https://doi.org/10.1182/blood-2015-09-665018
Richardson, P.G., Moreau, P., Laubach, J.P., Gupta, N., Hui, A.M., Anderson, K.C., . . . Kumar, S. (2015). The investigational proteasome inhibitor ixazomib for the treatment of multiple myeloma. Future Oncology, 11, 1153–1168. https://doi.org/10.2217/fon.15.9
Richardson, P.G., Siegel, D.S., Vij, R., Hofmeister, C.C., Baz, R., Jagannath, S., . . . Anderson, K.C. (2014). Pomalidomide alone or in combination with low-dose dexamethasone in relapsed and refractory multiple myeloma: A randomized phase 2 study. Blood, 123, 1826–1832. https://doi.org/10.1182/blood-2013-11-538835
Richardson, P.G., Sonneveld, P., Schuster, M.W., Stadtmauer, E.A., Facon, T., Harousseau, J.L., . . . San Miguel, J. (2009). Reversibility of symptomatic peripheral neuropathy with bortezomib in the phase III APEX trial in relapsed multiple myeloma: Impact of a dose-modification guideline. British Journal of Haematology, 144, 895–903. https://doi.org/10.1111/j.1365-2141.2008.07573.x
Richardson, P.G., Xie, W., Mitsiades, C., Chanan-Khan, A.A., Lonial, S., Hassoun, H., . . . Amato, A.A. (2009). Single-agent bortezomib in previously untreated multiple myeloma: Efficacy, characterization of peripheral neuropathy, and molecular correlations with response and neuropathy. Journal of Clinical Oncology, 27, 3518–3525. https://doi.org/10.1200/jco.2008.18.3087
Roy, V., Stewart, A.K., Bergsagel, P.L., Dispenzieri, A., Laumann, K., Allred, J., . . . Rajkumar, S.V. (2015). Phase I/II study of melphalan, prednisone and lenalidomide combination for patients with newly diagnosed multiple myeloma who are not candidates for stem cell transplantation. Blood Cancer Journal, 5, e294. https://doi.org/10.1038/bcj.2015.23
San-Miguel, J.F., Hungria, V.T., Yoon, S.S., Beksac, M., Dimopoulos, M.A., Elghandour, A., . . . Richardson, P.G. (2014). Panobinostat plus bortezomib and dexamethasone versus placebo plus bortezomib and dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma: A multicentre, randomised, double-blind phase 3 trial. Lancet Oncology, 15, 1195–1206. https://doi.org/10.1016/S1470-2045(14)70440-1
San Miguel, J.F., Schlag, R., Khuageva, N.K., Dimopoulos, M.A., Shpilberg, O., Kropff, M., . . . Richardson, P.G. (2008). Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. New England Journal of Medicine, 359, 906–917. https://doi.org/10.1056/NEJMoa0801479
Shah, J.J. (2013). Incidence and management of renal adverse events in patients with relapsed and/or refractory multiple myeloma treated with single-agent carfilzomib. Oncology, 27(Suppl. 3), 19–23.
Siegel, D.S., Weisel, K.C., Dimopoulos, M.A., Baz, R., Richardson, P., Delforge, M., . . . Palumbo, A. (2016). Pomalidomide plus low-dose dexamethasone in patients with relapsed/refractory multiple myeloma and moderate renal impairment: A pooled analysis of three clinical trials. Leukemia and Lymphoma, 57, 2833–2838. https://doi.org/10.1080/10428194.2016.1177181
Singhal, S., Mehta, J., Desikan, R., Ayers, D., Roberson, P., Eddlemon, P., . . . Barlogie, B. (1999). Antitumor activity of thalidomide in refractory multiple myeloma. New England Journal of Medicine, 341, 1565–1571. https://doi.org/10.1056/NEJM199911183412102
Smith, L.C., Bertolotti, P., Curran, K., & Jenkins, B. (2008). Gastrointestinal side effects associated with novel therapies in patients with multiple myeloma: Consensus statement of the IMF Nurse Leadership Board. Clinical Journal of Oncology Nursing, 12(Suppl. 3), S37–S52. https://doi.org/10.1188/08.CJON.S1.37-51
Song, X., Cong, Z., & Wilson, K. (2016). Real-world treatment patterns, comorbidities, and disease-related complications in patients with multiple myeloma in the United States. Current Medical Research and Opinion, 32, 95–103. https://doi.org/10.1185/03007995.2015.1105202
Sonneveld, P., Verelst, S.G., Lewis, P., Gray-Schopfer, V., Hutchings, A., Nixon, A., & Petrucci, M.T. (2013). Review of health-related quality of life data in multiple myeloma patients treated with novel agents. Leukemia, 27, 1959–1969. https://doi.org/10.1038/leu.2013.185
Stewart, A.K. (2015). Carfilzomib for the treatment of patients with relapsed and/or refractory multiple myeloma. Future Oncology, 11, 2121–2136. https://doi.org/10.2217/fon.15.123
Stewart, A.K., Rajkumar, S.V., Dimopoulos, M.A., Masszi, T., Špička, I., Oriol, A., . . . Palumbo, A. (2015). Carfilzomib, lenalidomide, and dexamethasone for relapsed multiple myeloma. New England Journal of Medicine, 372, 142–152. https://doi.org/10.1056/NEJMoa1411321
Swaika, A., Paulus, A., Miller, K.C., Sher, T., Almyroudis, N.G., Ball, D., . . . Chanan-Khan, A.A. (2012). Acyclovir prophylaxis against varicella zoster virus reactivation in multiple myeloma patients treated with bortezomib-based therapies: A retrospective analysis of 100 patients. Journal of Supportive Oncology, 10(4), 155–159. https://doi.org/10.1016/j.suponc.2011.10.006
Takeda Pharmaceutical Company. (2015). Velcade® (bortezomib) [Package insert]. Retrieved from http://www.velcade.com/files/PDFs/VELCADE_PRESCRIBING_INFORMATION.pdf
Takeda Pharmaceutical Company. (2016). Ninlaro®(ixazomib) [Package insert]. Retrieved from https://www.ninlarohcp.com/pdf/prescribing-information.pdf
Tam, C.C., Viviani, L., Rodrigues, L.C., & O’Brien, S.J. (2013). The second study of infectious intestinal disease (IID2): increased rates of recurrent diarrhoea in individuals aged 65 years and above. BMC Public Health, 13, 739. https://doi.org/10.1186/1471-2458-13-739
Tariman, J.D. (2017). Changes in cancer treatment: Mabs, mibs, mids, nabs, and nibs. Nursing Clinics of North America, 52, 65–81. https://doi.org/10.1016/j.cnur.2016.10.004
Tariman, J.D., Gleason, C., Faiman, B., Doss, D., Catamero, D., Bishop-Royse, J., . . . Lonial, S. (2016). Lack of health maintenance examinations and risk in myeloma patients. Cancer Medicine, 5, 1425–1435. https://doi.org/10.1002/cam4.716
Tariman, J.D., Love, G., McCullagh, E., & Sandifer, S. (2008). Peripheral neuropathy associated with novel therapies in patients with multiple myeloma: Consensus statement of the IMF Nurse Leadership Board. Clinical Journal of Oncology Nursing, 12(Suppl. 3), S29–S36. https://doi.org/10.1188/08.CJON.S1.29-35
Terpos, E., Christoulas, D., Kastritis, E., Katodritou, E., Pouli, A., Michalis, E., . . . Dimopoulos, M.A. (2013). The Chronic Kidney Disease Epidemiology Collaboration cystatin C (CKD-EPI-CysC) equation has an independent prognostic value for overall survival in newly diagnosed patients with symptomatic multiple myeloma; is it time to change from MDRD to CKD-EPI-CysC equations? European Journal of Haematology, 91, 347–355. https://doi.org/10.1111/ejh.12164
Thomas, M.E., Blaine, C., Dawnay, A., Devonald, M.A., Ftouh, S., Laing, C., . . . Ostermann, M. (2015). The definition of acute kidney injury and its use in practice. Kidney International, 87, 62–73. https://doi.org/10.1038/ki.2014.328
Tofthagen, C., & Irwin, M. (2014). Peripheral neuropathy. In M. Irwin & L.A. Johnson (Eds.), Putting evidence into practice: A pocket guide to cancer symptom management (pp. 201–210). Pittsburgh, PA: Oncology Nursing Society.
Tsubaki, M., Takeda, T., Tani, T., Shimaoka, H., Suzuyama, N., Sakamoto, K., . . . Nishida, S. (2015). PKC/MEK inhibitors suppress oxaliplatin-induced neuropathy and potentiate the antitumor effects. International Journal of Cancer, 131, 243–250. https://doi.org/10.1002/ijc.29367
Turner, J.G., Dawson, J.L., Grant, S., Shain, K.H., Dalton, W.S., Dai, Y., . . . Sullivan, D.M. (2016). Treatment of acquired drug resistance in multiple myeloma by combination therapy with XPO1 and topoisomerase II inhibitors. Journal of Hematology and Oncology, 9, 73. https://doi.org/10.1186/s13045-016-0304-z
United States Renal Data System. (2016). Chapter 1: Incidence, prevalence, patient characterisics, and treatment modalities. Retrieved from https://www.usrds.org/2016/view/v2_01.aspx
Vahdat, L., Papadopoulos, K., Lange, D., Leuin, S., Kaufman, E., Donovan, D., . . . Balmaceda, C. (2001). Reduction of paclitaxel-induced peripheral neuropathy with glutamine. Clinical Cancer Research, 7, 1192–1197.
Velasco, R., Petit, J., Clapés, V., Verdú, E., Navarro, X., & Bruna, J. (2010). Neurological monitoring reduces the incidence of bortezomib-induced peripheral neuropathy in multiple myeloma patients. Journal of the Peripheral Nervous System, 15, 17–25. https://doi.org/10.1111/j.1529-8027.2010.00248.x
Visovsky, C., Haas, M., Faiman, B., Kurtin, S. , Shaftic, A.M., Lyden, E., & Rice, J. (2012). Nurse self-evaluation of assessment of chemotherapy-induced peripheral neuropathy in patients with cancer. Journal of the Advanced Practitioner in Oncology, 3, 319–325.
Vogl, D.T., Dingli, D., Cornell, R.F., Huff, C.A., Jagannath, S., Bhutani, D., . . . Stewart, A.K. (2016). Selinexor and low dose dexamethasone (Sd) in patients with lenalidomide, pomalidomide, bortezomib, carfilzomib and anti-CD38 ab refractory multiple myeloma (MM): STORM Study. Blood, 128, 491. Retrieved from http://www.bloodjournal.org/content/128/22/491
Wang, W.S., Lin, J.K., Lin, T.C., Chen, W.S., Jiang, J.K., Wang, H.S., . . . Chen, P.M. (2007). Oral glutamine is effective for preventing oxaliplatin-induced neuropathy in colorectal cancer patients. Oncologist, 12, 312–319. https://doi.org/10.1634/theoncologist.12-3-312
Weber, D.M., Chen, C., Niesvizky, R., Wang, M., Belch, A., Stadtmauer, E.A., . . . Knight, R.D. (2007). Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. New England Journal of Medicine, 357, 2133–2142. https://doi.org/10.1056/NEJMoa070596
Wei, K., Yin, Z., & Xie, Y. (2016). Roles of the kidney in the formation, remodeling and repair of bone. Journal of Nephrology, 29, 349–357. https://doi.org/10.1007/s40620-016-0284-7
Zannetti, B.A., Zamagni, E., Santostefano, M., De Sanctis, L.B., Tacchetti, P., Mancini, E., . . . Cavo, M. (2015). Bortezomib-based therapy combined with high cut-off hemodialysis is highly effective in newly diagnosed multiple myeloma patients with severe renal impairment. American Journal of Hematology, 90, 647–652. https://doi.org/10.1002/ajh.24035
Ziegler, D., Nowak, H., Kempler, P., Vargha, P., & Low, P.A. (2004). Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: A meta-analysis. Diabetic Medicine, 21, 114–121.
Ziogas, D.C., Kastritis, E., Terpos, E., Roussou, M., Migkou, M., Gavriatopoulou, M., . . . Dimopoulos, M.A. (2017). Hematologic and renal improvement of monoclonal immunoglobulin deposition disease after treatment with bortezomib-based regimens. Leukemia and Lymphoma, 58, 1832–1839. https://doi.org/10.1080/10428194.2016.1267349
Zweegman, S., Palumbo, A., Bringhen, S., & Sonneveld, P. (2014). Age and aging in blood disorders: Multiple myeloma. Haematologica, 99, 1133–1137. https://doi.org/10.3324/haematol.2014.110296




