Bone Health, Pain, and Mobility: Evidence-Based Recommendations for Patients With Multiple Myeloma
Background: About 85% of patients with multiple myeloma develop bone disease. In these patients, lytic bone lesions can cause fractures, poor circulation, blood clots, pain, poor mobility, and decreased quality of life.
Objectives: This article presents consensus statements to guide nurses in the assessment and management of bone disease, pain, and mobility in patients with multiple myeloma at varying points in their disease trajectory.
Methods: Members of the International Myeloma Foundation Nurse Leadership Board reviewed previously provided recommendations, current recommendations based on literature review, and evidence-based grading.
Findings: Oncology nurses play a key role in maximizing bone health, minimizing skeletal injury, maximizing pain control, and improving quality of life in patients by enhancing patient mobility and safety. Clinician assessment accompanied by effective interventions reduces patient injury and optimizes functioning in patients with multiple myeloma.
Jump to a section
With improvements in supportive care and treatment, patients with multiple myeloma (MM) are living longer (Kumar et al., 2014). For patients with MM, bone pain is a hallmark symptom that affects function, mobility, and quality of life (QOL) (Rajkumar, 2009). Health-related QOL includes physical and mental health perceptions (e.g., energy level, mood) and their correlates, as well as health risks and comorbidity, functional status, social support, and socioeconomic status (Centers for Disease Control and Prevention, 2016). In this updated evidence-based consensus statement, the International Myeloma Foundation (IMF) Nurse Leadership Board (NLB) presents the most recent information about bone health and functional mobility, along with evidence-based recommendations for the management of bone pain in MM survivors, with a focus on promoting patient QOL. The IMF NLB uses Melnyk and Fineout-Overholt’s (2011) levels of evidence as a systematic framework for the appraisal and grading of the consensus statements and evidence-based recommendations for MM- and treatment-related symptoms. Melnyk and Fineout-Overholt’s (2011) rating system has seven levels that are inclusive of evidence from qualitative studies, case-control and cohort studies, reports of expert committees, and opinion of authorities.
Bone Health in Multiple Myeloma Survivors
Since the 2011 IMF NLB consensus care plan (Miceli, Colson, Faiman, Miller, & Tariman, 2011), the published literature about bone health maintenance has focused on preventing skeletal events through early disease detection. Because most patients with high-risk smoldering MM (which has the potential to evolve into active disease and is considered a premalignant plasma cell proliferative disorder that requires monitoring) progress to active MM within two years, the latest diagnostic criteria for active myeloma now require at least one myeloma defining event (MDE), of which bone disease is one possible type of event (Rajkumar et al., 2014) (see Figure 1). As a result, patients who were classified by previous criteria as having high-risk smoldering MM are now classified as having active MM if they have one MDE (Rajkumar, Larson, & Kyle, 2011). The updated criteria promote starting treatment prior to end-organ damage, such as bone disease (Rajkumar, 2011a; Rajkumar et al., 2014). Consequently, there is growing support for more sensitive diagnostic imaging to identify early bone disease. Early detection and treatment of MM to prevent bone damage, along with already understood methods to promote bone health, are critical to patient QOL. A patient education tip sheet for managing bone health can be found in Figure 2.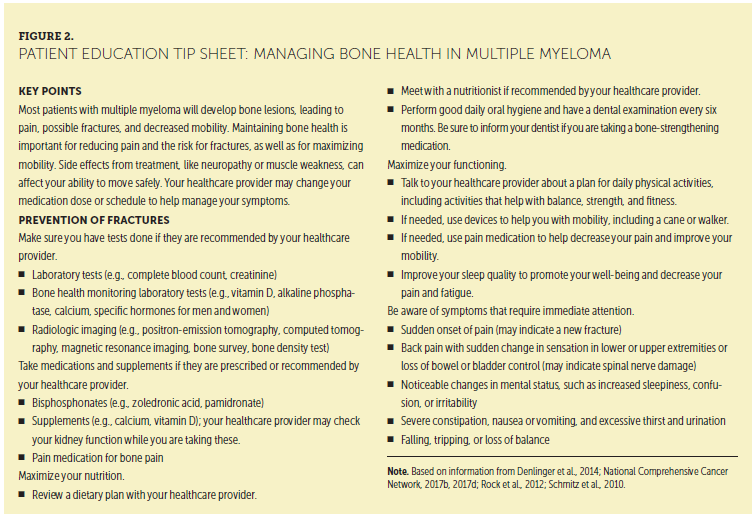
Evidence-Based Recommendations to Manage and Maintain Bone Health
Level of Evidence I
• Based on the International Myeloma Working Group (IMWG) criteria, active MM should be diagnosed when there is more than one lesion with a diameter greater than 5 mm detected by whole-body magnetic resonance imaging (MRI), even in the absence of end-organ damage (Dimopoulos et al., 2015; Rajkumar et al., 2014). For lesions involving the skull and ribs, the metastatic bone survey may still play an important role in evaluating bone disease. However, the more sensitive imaging techniques, such as 2-deoxy-2 [fluorine-18] fluoro-D-glucose positron-emission tomography (F-FDG-PET), MRI, and whole-body computed tomography have greater diagnostic and prognostic value (Regelink et al., 2013).
• MRI is highly sensitive for early detection of myeloma bone marrow infiltration. The IMWG recommends that all individuals with smoldering or asymptomatic MM be evaluated for bone lesions with whole-body MRI (Rajkumar et al., 2014; Terpos et al., 2013).
• An IV bisphosphonate medication (BPM) every 3–4 weeks is recommended for all newly diagnosed patients with MM. BPM may be stopped or its frequency decreased after 12–24 months at the provider’s discretion for patients who have no evidence of bone disease and have achieved a complete response to MM therapy (Terpos et al., 2013).
• With long-term use of BPM, patients are at risk of developing osteonecrosis of the jaw (Miceli et al., 2011; Terpos et al., 2013). For this reason, IMWG guidelines include dental evaluation prior to initiation of therapy and minimum annual dental evaluations after the initiation of BPM. Patients should avoid dental extractions, if possible. If osteonecrosis of the jaw develops, BPM should be held until relapse of the MM occurs.
• Close monitoring of renal function is required, and serum creatinine should be assessed before each BPM treatment; the dose should be held if there is evidence of renal dysfunction as defined as a creatinine clearance less than 30 ml per minute. Patients with a creatinine clearance of 30–60 ml per minute should receive reduced doses of zoledronate with no change to infusion time, whereas pamidronate should be given via a four-hour infusion (Terpos et al., 2013).
• Calcium and vitamin D supplements should be prescribed to promote bone health if no clear contraindications are noted (National Osteoporosis Foundation, 2017). This is relevant to patients with MM because 60% are vitamin D deficient and in need of supplementation to prevent bone resorption, decrease parathyroid hormone levels, and maintain bone health (National Osteoporosis Foundation, 2017; Terpos et al., 2013).
Level of Evidence III
• Balloon kyphoplasty or vertebroplasty is recommended for vertebral compression fractures (Dudeney, Lieberman, Reinhardt, & Hussein, 2002; Hadjipavlou, Tzermiadianos, Katonis, & Szpalski, 2005; Lane et al., 2004).
• Low-dose radiation therapy (as much as 30 Gy) and orthopedic consultation are recommended for the management of vertebral compression fractures, impending or actual long bone fractures, and associated pain (Miceli et al., 2011; Terpos et al., 2013).
• In one prospective cohort study, osteolytic lesions were detected using F-FDG-PET in patients, particularly in patients with smoldering MM, without pain or destructive lesions on metastatic bone survey (Siontis et al., 2015).
Pain Prevention and Management in Multiple Myeloma Survivors
The prevention and management of pain is a priority for the NLB, which has addressed the topic in several publications (Miceli et al., 2011; Richards & Brigle, 2016; Tariman, Love, McCullagh, & Sandifer, 2008). Physician counterparts within the IMWG also place a priority on pain management in several of their publications (Johnson et al., 2011; Moreau et al., 2011; Richardson et al., 2012; Terpos et al., 2013). Tip sheets for patients and for healthcare providers can be found in Figures 3 and 4.
Primary Sources of Multiple Myeloma Pain
Pain can be acute or chronic in patients with MM. Acute pain is of short duration, has an identifiable cause, and functions to warn people about and protect them from tissue damage. In most clinical situations, acute pain resolves. In contrast, chronic pain lasts longer than expected for typical recovery time following tissue injury. In general, chronic pain continues for longer than one to six months or recurs at intervals over time. Patients with MM may experience acute pain, chronic pain, or both, because both types can occur simultaneously depending on the disease state and any ongoing diagnostic procedures or treatments (Miaskowski, 2010).
In many instances, pain associated with MM may be prevented and treated. For this reason, members of the healthcare team should continuously assess patients for pain and modify treatment regimens based on current symptoms. Treatment options are numerous; however, pain management requires constant shared communication between the healthcare team and the patient. Referral to a pain specialist or palliative care service may be necessary if pain persists despite multiple unsuccessful management attempts (Miceli et al., 2011).
Sources of Pain
Cancer can cause pain through several mechanisms, making pain one of the most feared and prevalent symptoms in all patients diagnosed with cancer (Faiman, 2016). Because MM involves bone disease, bone pain is one of the most common symptoms of MM (Kyle & Rajkumar, 2009). In addition, patients with MM may also experience neuropathic and procedural pain. Identifying the underlying source of pain may help optimize treatment for each patient with MM.
Bone Disease as a Source of Pain
MM has a higher incidence of bone involvement than any other malignant disease. For this reason, pain is a presenting characteristic in a high proportion of patients diagnosed with MM. Osteoporosis, osteolytic bone lesions, pathologic fractures, and vertebral compression fractures are common findings at diagnosis and throughout the course of MM. An estimated 80%–90% of all patients with MM experience pain from bone involvement, and 60% develop pathologic fractures (Delgado-Calle, Bellido & Roodman, 2014; Greenberg et al., 2014; Kyle & Rajkumar, 2009).
Neuropathic Pain
Neuropathic pain is caused by abnormal somatosensory processing in the peripheral or central nervous system (Dworkin et al., 2010). It is less common than pain caused by bone involvement but can be unrelenting and influence patient QOL. Neuropathic pain can present as peripheral neuropathy (PN) at the time of MM diagnosis or may be related to comorbid conditions, such as amyloidosis, cryoglobulinemia, spinal cord impingement, or autoimmune mechanisms (Mohty et al., 2010). Symptoms of PN are also common in patients diagnosed with disorders such as diabetes, peripheral vascular disease, nutritional deficiency, alcoholism, postsurgical changes, and inherited diseases (Staff & Windebank, 2014; Yorek, 2015). Postherpetic neuralgia (PHN), another cause of neuropathic pain, is a direct result of reactivation of the varicella-zoster virus (VZV), which may occur in patients with MM because of stress, weakened immunity from active disease, or treatment (Faiman, 2016).
Procedural Pain
Patients with MM may undergo multiple necessary procedures throughout their diagnostic evaluations and treatment courses. Although necessary for assessment of disease activity and treatments, procedures—such as tumor biopsy, central line placement, surgical interventions, venipuncture, and bone marrow biopsies— are all sources of recurrent, acute pain (Rajkumar et al., 2014). Patient-focused approaches to limiting the number of blood draws can be achieved by consolidating laboratory tests from different care providers (e.g., primary care, hematology) into single blood draws (Rajkumar et al., 2014). In addition, conscious sedation may be provided in some institutions for invasive procedures, such as bone marrow biopsies, alleviating this recurrent source of pain and procedure-related anxiety (Hjortholm et al., 2013).
Evidence-Based Recommendations to Manage Pain
Level of Evidence I
• Pain can adversely affect QOL in patients with cancer and should be prevented, when possible, or effectively managed to reduce physical and emotional distress (Mesgarpour et al., 2014; Miaskowski, 2010).
• Comprehensive pain assessment starts with a detailed history, examination of the painful site(s), and a diagnostic evaluation, which may include radiologic imaging, to find a source of musculoskeletal pain. The next steps are to determine the effect of pain on the patient’s QOL and overall function, and to determine what interventions are needed to relieve the pain (Miaskowski, 2010).
• Patients should be questioned about the impact of pain, specifically on their functional status and activities of daily living. Patients should also be questioned about any previous coping strategies they may have had that worked, any financial issues as a result of their functional status, and any concerns about opioid use to manage pain (Greco et al., 2011; Ripamonti, Santini, Maranzano, Berti, & Roila, 2012).
• Clinicians can initiate measures to quickly relieve acute pain while the patient is undergoing evaluation to determine the specific etiology of the pain (Portenoy, 2011).
• Interventions to alleviate pain include pharmacologic and nonpharmacologic strategies, which are chosen based on results of the initial pain assessment. Treatment effectiveness is continuously assessed and changes made based on the clinical situation and the patients’ response (Miaskowski, 2010).
• Neuropathic pain can progress from being mildly uncomfortable to being incapacitating and limiting function and QOL in patients with MM. Healthcare providers must prevent neuropathic pain when possible and, if present, to diagnose and treat it. PN is a neuropathic pain and is associated with various medications, including thalidomide and bortezomib (Mohty et al., 2010; Moulin et al., 2014).
Level of Evidence II
• Conduct a comprehensive assessment to determine the type of pain being experienced and the best interventions for any patient with MM. The initial step in assessing pain is derived from the patient’s self-report, including the description, intensity, duration, etiology, and characteristics of pain. In addition, any aggravating and alleviating factors should be determined. The effectiveness of interventions already attempted, such as medications, rest, activities, heat, or cold compresses, should be determined (National Comprehensive Cancer Network [NCCN], 2017a).
Level of Evidence V
• Hjortholm et al. (2013) provide pharmacologic and nonpharmacologic strategies to reduce procedural pain associated with bone marrow biopsy and aspiration. Many of the nonpharmacologic strategies, such as hypnosis, music, and cognitive behavioral therapy, may be used to reduce other sources of procedural pain and anxiety.
Pharmacologic Interventions for Pain
Level of Evidence I
• Based on the World Health Organization (1990, 2017) analgesic ladder, opioids are the principal analgesics to treat moderate-to-severe pain. Opioids prescribed for chronic pain should be administered on a regular basis. Before an opioid is prescribed and administered, nurses, providers, and pharmacists should have an understanding of the medication’s mechanism of action, common starting doses, equivalence to other opioids, duration of effect, half-life, available routes, and associated adverse effects.
• Breakthrough pain medication may be needed at the end of the extended-release medication schedule or with added activity during the day (McMenamin, 2011; Miaskowski, 2010; NCCN, 2017a). Nonsteroidal anti-inflammatory medications are associated with renal toxicities and, therefore, should be avoided in patients with MM, who are at risk for renal dysfunction (NCCN, 2017c).
• Pharmacologic treatment of metastatic bone disease requires a multi-strategy approach. This includes treatment of the underlying disease, opioid/analgesic medications, and use of bisphosphonate therapy. The use of IV BPM (pamidronate and zoledronic acid) as supportive care for patients with MM may reduce pain, increase calcium levels, decrease skeletal complications secondary to bone involvement, and improve QOL (NCCN, 2017c; Terpos et al., 2013).
The administration of BPM significantly reduces the number of skeletal events. Either pamidronate or zoledronic acid is recommended for patients with MM who have one or more lytic lesions identified radiologically (Pozzi & Raje, 2011).
Level of Evidence II
• Initial opioid titration for opiate-naive patients is done at a slower pace than in opiate-tolerant patients (NCCN, 2017a). When long-acting opioids do not adequately control pain, a short-acting or breakthrough medication should be prescribed. Administration of BPM can decrease pain, improve performance, and preserve QOL (NCCN, 2017c).
Nonpharmacologic Interventions for Pain
Level of Evidence I
• The efficacy of complementary and alternative medicine (CAM) in cancer pain management was systematically reviewed in 2006 (Bardia, Barton, Prokop, Bauer, & Moynihan, 2006). Although the authors mentioned hypnosis, imagery, support groups, acupuncture, and healing touch as promising interventions, given the paucity of rigorous trials, the authors did not recommend these CAM approaches for pain management.
• Two systematic reviews among patients with hematologic cancers revealed limited data to support an improvement in patient QOL associated with yoga and meditation (Felbel, Meerpohl, Monsef, Engert, & Skoetz, 2014; Salhofer, Will, Monsef, & Skoetz, 2016).
Level of Evidence III
• The IMF NLB recommends either vertebroplasty or kyphoplasty for the treatment of vertebral compression fractures. These procedures successfully decrease pain and potentially restore or prevent loss in height. Vertebroplasty is performed by injecting methyl methacrylate into collapsed vertebra, and kyphoplasty is performed by inserting an inflatable balloon followed by the methyl methacrylate injection (Dudeney et al., 2002; Hadjipavlou et al., 2005). Pain relief is generally rapid and can be long-lasting (Lane et al., 2004).
• Localized external radiation is a widely accepted treatment option to control MM pain. The IMF NLB recommends radiation therapy primarily for the treatment of solitary plasmacytoma, symptomatic spinal cord compression, extremely painful lytic lesion, and for the prevention of pathologic fractures. However, radiation therapy may cause delays in systemic therapies because of the combined toxicity associated with aggressive systemic therapy and radiation (Terpos et al., 2015).
Level of Evidence IV
• CAM may offer several options for patients with symptoms related to MM or MM treatment. In the 2007 National Health Interview Survey, individuals with cancer listed the five most common complementary therapies practiced as vitamin or mineral supplement, prayer for self, intercessory prayer, chiropractic/osteopathic manipulation, and herbal therapies (Anderson & Taylor, 2012).
Management and Prevention of Neuropathic Pain
Level of Evidence I
• According to a recent systematic review, pain management is the most studied and established role for cannabinoid therapies (Maida & Daeninck, 2016). Most of the information about cannabis use is derived from patients with symptoms of PN (Maida & Daeninck, 2016). In addition, cannabinoids appear to be safe when used in appropriate doses. A meta-analysis reported that about 1 in every 5–6 patients with PN improved with inhaled cannabis (Andreae et al., 2015).
• A systematic review indicated that cannabinoids are effective adjuvants for opioid therapy in patients whose cancer pain is not completely relieved by opioid therapy alone (Tateo, 2017). However, the author cautions that there is a paucity of high-quality studies to support a stronger conclusion about cannabinoids for cancer pain (Tateo, 2017).
• A systematic review revealed that inhaled cannabis is associated with potential pulmonary issues and, therefore, the IMF NLB recommends ingesting cannabis to avoid these detrimental side effects (Tetrault et al., 2007).
Level of Evidence II
• For patients receiving bortezomib therapy for relapsed MM, subcutaneous was equal in efficacy to IV administration and associated with lower rates of PN (Moreau et al., 2011).
• Dosing and scheduling adjustments of potentially neurotoxic agents can also be made to reduce onset or progression of PN symptoms (Richardson et al., 2012). Concomitant prophylactic use of antiviral medication, such as acyclovir, with proteasome inhibitor therapy will prevent activation of VZV and postherpetic neuralgia.
Level of Evidence III
• In general, the studies of CAM therapies are small and not randomized, controlled trials, which makes it difficult to determine the efficacy of specific CAM interventions. However, data suggest that providing CAM therapies with concurrent chemotherapy improves QOL in patients with cancer (Bar-Sela, Danos, Visel, Mashiach, & Mitnik, 2015).
• In a published study by Han et al. (2017), patients with MM received methylcobalamin therapy or methylcobalamin and acupuncture to alleviate symptoms of PN. The study reported a decrease in PN pain scores that was greater in the acupuncture than the non-acupuncture group. Using a visual analog scale, the pain scores decreased in 86% of participants in the methylcobalamin plus acupuncture group compared to 78% in the methylcobalamin-only group. In addition, the magnitude of decrease was 5.6 to 3.2 versus 5.5 to 4.3. A decrease of 2 points on an 11-point pain visual analog scale is considered clinically relevant (Farrar, Young, LaMoreaux, Werth, & Poole, 2001; Han et al., 2017).
Level of Evidence IV
• For patients experiencing PN or PHN, coanalgesics often are used in conjunction with opioids to treat neuropathic pain (Fallon, 2013). Coanalgesics, or adjuvant analgesics, have pain-relieving properties in specific clinical situations; however, the primary indications of coanalgesics are not for the treatment of pain. Common coanalgesic medications include antidepressants (nortriptyline, duloxetine, or venlafaxine), anticonvulsants (gabapentin and pregabalin), corticosteroids, and topical agents (capsaicin or local anesthetics) (Fallon, 2013; Moulin et al., 2014; NCCN, 2017a).
• Tricyclic antidepressants are effective and inexpensive medications used to treat neuropathic pain; however, patients should be monitored for cardiotoxicity, urinary retention, orthostatic hypotension, constipation, and drowsiness. Secondary amine tricyclic antidepressants, such as nortriptyline and desipramine, are preferred over tertiary amine tricyclic antidepressants (e.g., amitriptyline, imipramine) because they are better tolerated by older adults (Fallon, 2013; Moulin et al., 2014; NCCN, 2017a).
• Anticonvulsants and antidepressants used for neuropathic pain management should be increased slowly to avoid toxicities (Moulin et al., 2014; NCCN, 2017a).
Mobility and Safety in Multiple Myeloma Survivors
Mobility and safety practice recommendations were provided by Rome, Jenkins, and Lilleby (2011), which incorporated aspects of health maintenance with promotion of QOL. As previously noted, mobility challenges vary among patients with MM and may change over time. An individual plan of care is based on physical activity, the overall level of functioning, frailty, and fall risk.
Exercise is an effective behavioral intervention with the potential to mitigate multiple side effects and improve physical function in patients with cancer and survivors. Exercise can improve side effects, such as fatigue, cognitive impairment, sleep problems, depression, pain, anxiety, and physical dysfunction (Mustian, Sprod, Janelsins, Peppone, & Mohile, 2012), and symptom clusters (Mustian et al., 2016). Many cancer survivors are highly motivated to seek information about nutrition, physical activity, dietary supplement use, and complementary therapies (Rock et al., 2012). Most patients with cancer report that they do not discuss initiating or continuing an exercise program, yet they want healthcare providers to initiate discussions about exercise (Mustian et al., 2012). Clinicians should feel encouraged that lifestyle behaviors are one area survivors can control if they are prompted to change and are provided with resource information. Interventions may include telephone-based health behavior coaching and positive feedback, as well as motivational counseling (Denlinger et al., 2014). Survivors often have heightened motivation, particularly close to the time of diagnosis, and this is perhaps the ideal time to employ an exercise program or other behavioral intervention in the treatment plan (Mustian et al., 2012). Tip sheets for patients and for healthcare providers can be found in Figures 5 and 6.
Evidence-Based Recommendations to Improve Mobility and Safety
Level of Evidence I
• The American Cancer Society, through a group of experts, developed nutritional and physical activity guidelines for cancer survivors (Rock et al., 2012). These guidelines offer detailed information in the following categories: (a) nutrition and physical activity across the cancer continuum; (b) weight management, physical activity, food choices, alcohol intake, and food safety; (c) selected cancer sites; and (d) answers to common questions many survivors have (Rock et al., 2012). These guidelines may be implemented in any clinical practice.
• A Cochrane systematic review by Bergenthal et al. (2014) was conducted on the quality of trials on aerobic physical exercise for adult patients with hematologic malignancies, including MM. Based on the findings from this systematic review, the IMF NLB recommends that physical exercise should be incorporated with standard care to improve patient QOL, particularly aspects of physical functioning, depression, and fatigue. Three trials investigated serious adverse events of physical exercise, and found no evidence of an increase in serious adverse events, such as bleeding, fever, thrombosis, pneumonia, and other infections, with exercise (Bergenthal et al., 2014).
Level of Evidence II
• The NCCN guidelines on survivorship propose that structured aerobic and resistance training programs, when initiated after treatment, can improve cardiovascular fitness and strength and can have positive effects on balance, body composition, and QOL (Denlinger et al., 2014; NCCN, 2017b).
Level of Evidence III
• Several studies demonstrated that physical activity can be safely performed by MM survivors, and there is evidence suggesting physical activity may aid in various treatments and alleviate some comorbidities (Coleman et al., 2003; Coon & Coleman, 2004a, 2004b; Groeneveldt et al., 2013; Jones et al., 2004).
Level of Evidence VII
• Care should be provided to frail patients by a range of healthcare providers, including geriatricians, psychiatrists, pharmacists, physiatrists, social workers, and dietitians (Tuchman et al., 2014). The goal for these patients is to reduce stressors that can enhance frailty and potentially limit effective treatment. Because of the increased toxicities that may occur in older adult patients with cancer, careful selection of treatment and adjuncts is critical to reduce harm; for example, the risks and benefits of anticoagulation for prevention of venous thromboembolism should be carefully considered in patients already at risk for falls and bleeding (Tuchman et al., 2014).
Functional and Frailty Assessment
About one-third of patients with MM are older than age 75 years at diagnosis. However, the population is heterogeneous, and tolerability of treatment is not defined by age alone, but rather by a combination of age, physical function, cognitive function, and comorbidities (Pawlyn, Gay, Larocca, Roy, & Ailawadhi, 2016). In addition to a general assessment, patient-specific screening of functioning, fall risk, frailty, and comorbidity indices should be employed (Bila et al., 2015). A variety of tools are available to assess fall risk and were previously described by Rome et al. (2011). The NCCN guidelines on survivorship (Denlinger et al., 2014; NCCN, 2017b) recommend that a risk assessment for exercise-induced adverse events be used based on disease and treatment history, late and long-term effects, and comorbidities. The NCCN guidelines recommend that the Physical Activity Readiness Questionnaire for Everyone (PAR-Q+) be considered as an assessment tool (Bredin, Gledhill, Jamnik, & Warburton, 2013).
Patients with MM are at moderate risk for exercise-induced adverse events because of PN, bone metastases, poor bone health, arthritis, and musculoskeletal issues. Stability, balance, and gait should be assessed in these patients, and exercise plans should be tailored to the condition of each patient (e.g., stationary bike and water aerobics for patients with poor balance). Patients with MM should also be assessed for fracture and spinal cord compression risk. These moderate-risk survivors can often follow general recommendations, but with specific medical clearance and referrals to trained personnel for their exercise programs (Denlinger et al., 2014).
A comprehensive geriatric assessment (CGA) for those aged 65 years or older should be routinely conducted. CGA appraises objective health, including comorbidities and functional status, that can impact prognosis and treatment (NCCN, 2017c). Functional status includes activities of daily living and instrumental activities of daily living. With MM treatment and adjunct therapies, functional status can improve or worsen. Therefore, functional status assessment should be ongoing and include fall and gait assessment; medication review; and psychological, physical, and cognitive function (NCCN, 2017c).
The IMWG conducted a pooled analysis of 869 newly diagnosed older adult patients, all of whom had undergone a CGA (Palumbo et al., 2015). The CGA consisted of three tools: the Katz activities of daily living (Katz & Akpom, 1976), the Lawton instrumental activities of daily living (Lawton & Brody, 1969), and the Charlson Comorbidity Index (Charlson, Pompei, Ales, & MacKenzie, 1987). The analysis revealed that the frailty score predicted mortality and risk of toxicity in older adult patients with myeloma. A patient’s chronologic age, performance status, and physician’s clinical judgment were not sufficient to classify patient frailty. Instead, CGA is a more sensitive predictor of clinical outcomes and, therefore, performing CGA can optimize treatment.
Evidence-Based Recommendations to Manage Frailty
Level of Evidence VII
• Larocca and Palumbo (2015) published a narrative review of the literature and proposed an approach to MM assessment and treatment with a focus on patient frailty. They recommend the use of CGA to assess patients with MM in everyday clinical practice. Ideally, CGA results can be used to direct clinical decision making, tailor therapy, and ultimately to reduce risk of toxicity and improve treatment tolerance (Larocca & Palumbo, 2015).
• The IMF NLB recommends incorporating a CGA score to measure patient frailty in future clinical trials (Palumbo et al., 2015).
Implementing an Activity Plan of Care
Behavioral support interventions can assist cancer survivors in making behavior changes required for the implementation of a successful exercise program. Supervised exercise, support groups, telephone counseling, motivational interviewing, and cancer survivor–specific print materials may help overcome personal barriers (Rock et al., 2012; Smith et al., 2015). Barriers to physical activity in patients with MM may include side effects of therapy, pain, fatigue, fear of infection, and low self-motivation (Craike, Hose, Courneya, Harrison, & Livingston, 2013). Other factors that may be associated with noncompliance in patients with MM include history of fracture, spinal cord compression, and radiation. These individuals may require additional supervision to implement a safe and successful exercise program (Shallwani et al., 2015). When implementing a plan, it is important to consider personal interests and goals. Ask questions such as, “What activities do you enjoy, and are currently able to do?”; “What benefits will you get from activity—physical benefits, social benefits, keeping busy, etc.?”; and “What is your goal—weight loss, pain management, improved mobility and flexibility?” Addressing perceived benefits, barriers, and personal needs and goals will enhance compliance and lead to a more successful program (Craike et al., 2013).
A structured, individualized program that includes supervision and in-home follow-up can promote durable participation for patients with MM (Groenveldt et al., 2013). MM survivors, particularly those with bone disease, may require specialty consultation to advise them on restrictions for weight lifting or impact sports before implementing a plan. The American College of Sports Medicine (ACSM) provides minimum activity guidelines (Schmitz et al., 2010), but implementing these guidelines for individuals within their community may present challenges, including lack of appropriate services. To overcome challenges in connecting survivors to appropriate programs in their community, healthcare professionals should anticipate questions from exercise professionals and collaborate closely with them. In addition, healthcare professionals should feel comfortable following the ACSM guidelines and prescribing exercise to cancer survivors (Rome et al., 2011; Schmitz et al., 2010; Wolin, Schwartz, Matthews, Courneya, & Schmitz, 2012).
Evidence-Based Recommendations to Implement an Activity Plan of Care
Level of Evidence I
• The IMF NLB recommends that oncology nurses help maximize patients’ QOL by ensuring that mobility and safety is assessed and that the plan of care includes appropriate interventions and resources for survivorship. Every survivorship care plan should include recommendations for a healthy lifestyle, such as regular health screening, sun safety, immunizations, physical activity, dietary habits, and weight management (Denlinger et al., 2014).
• To reach the goal of improved patient outcomes with QOL, the IMF NLB recommends that nurses continuously assess and tailor patient interventions to manage pain, protect bone health, and maximize functional abilities (Denlinger et al., 2014).
Conclusion
Patients with MM are affected in many ways by physical changes, comorbidities, and treatment effects. In particular, patient QOL can be negatively affected by bone disease, pain, and limited mobility. Ongoing research and outcomes from that research provide healthcare providers with evidence to guide practice and patient care. Oncology nurses can help improve outcomes for patients with MM by conducting assessments at every encounter, regardless of care setting; playing a role in symptom prevention; managing medication side effects; and implementing plans of care that promote safety, mobility, and QOL in conjunction with the healthcare team.
The authors gratefully acknowledge Rafat Abonour, MD, Brian G.M. Durie, MD, and Diane P. Moran, RN, MA, EdM, at the International Myeloma Foundation, for their review of this manuscript.
About the Author(s)
Sandra Rome, RN, MN, AOCN®, CNS, is a hematology-oncology clinical nurse specialist at the Cedars-Sinai Medical Center in Los Angeles, CA; Kimberly Noonan, RN, MS, CNP, AOCN®, is a nurse practitioner at the Dana-Farber Cancer Institute in Boston, MA; Page Bertolotti, RN, BSN, OCN®, is a clinical nurse III in the Samuel Oschin Cancer Center at Cedars-Sinai Medical Center in Los Angeles, CA; Joseph D. Tariman, PhD, RN, ANP-BC, FAAN, is an assistant professor in the School of Nursing at DePaul University in Chicago, IL; and Teresa Miceli, BSN, RN, OCN®, is a BMT RN care coordinator at the Mayo Clinic in Minneapolis, MN. The authors take full responsibility for this content. This supplement was supported by the International Myeloma Foundation, with funding from Celgene Corporation, Karyopharm Therapeutics, and Takeda Oncology. Writing and editorial support was provided by Eubio Medical Communications. Bertolotti serves on speakers bureaus for Celgene Corporation and Takeda Pharmaceuticals. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Rome can be reached at sandra.rome@cshs.org, with copy to CJONEditor@ons.org. (Submitted June 2017. Accepted July 17, 2017.)
References
Anderson, J.G., & Taylor, A.G. (2012). Use of complementary therapies for cancer symptom management. Journal of Alternative and Complementary Medicine, 18, 235–241.
Andreae, M.H., Carter, G.M., Shaparin, N., Suslov, K., Ellis, R.J., Ware, M.A., . . . Sacks, H.S. (2015). Inhaled cannabis for chronic neuropathic pain: A meta-analysis of individual patient data. Journal of Pain, 16, 1221–1232. https://doi.org/10.1016/j.jpain.2015.07.009
Bardia, A., Barton, D.L., Prokop, L.J., Bauer, B.A., & Moynihan, T.J. (2006). Efficacy of complementary and alternative medicine therapies in relieving cancer pain: A systematic review. Journal of Clinical Oncology, 24, 5457–5464. https://doi.org/10.1200/JCO.2006.08.3725
Bar-Sela, G., Danos, S., Visel, B., Mashiach, T., & Mitnik, I. (2015). The effect of complementary and alternative medicine on quality of life, depression, anxiety, and fatigue levels among cancer patients during active oncology treatment: Phase II study. Supportive Care in Cancer, 23, 1979–1985. https://doi.org/10.1007/s00520-014-2560-1
Bergenthal, N., Will, A., Streckmann, F., Wolkewitz, K.D., Monsef, I., Engert, A., . . . Skoetz, N. (2014). Aerobic physical exercise for adult patients with haematological malignancies. Cochrane Database of Systematic Reviews, 11, CD009075.
Bila, J., Jelicic, J., Djurasinovic, V., Vukovic, V., Sretenovic, A., Andjelic, B., . . . Mihaljevic, B. (2015). Prognostic effect of comorbidity indices in elderly patients with multiple myeloma. Clinical Lymphoma, Myeloma and Leukemia, 15, 416–419.
Bredin, S.S., Gledhill, N., Jamnik, V.K., & Warburton, D.E. (2013). PAR-Q+ and ePARmed-X+: New risk stratification and physical activity clearance strategy for physicians and patients alike. Canadian Family Physician, 59, 273–277.
Centers for Disease Control and Prevention. (2016). HRQOL concepts. Retrieved from www.cdc.gov/hrqol/concept.htm
Charlson, M.E., Pompei, P., Ales, K.L., & MacKenzie, C.R. (1987). A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. Journal of Chronic Diseases, 40, 373–383.
Coleman, E.A., Coon, S., Hall-Barrow, J., Richards, K., Gaylor, D., & Stewart, B. (2003). Feasibility of exercise during treatment for multiple myeloma. Cancer Nursing, 26, 410–419.
Coon, S.K., & Coleman, E.A. (2004a). Exercise decisions within the context of multiple myeloma, transplant, and fatigue. Cancer Nursing, 27, 108–118.
Coon, S.K., & Coleman, E.A. (2004b). Keep moving: Patients with myeloma talk about exercise and fatigue. Oncology Nursing Forum, 31, 1127–1135.
Craike, M.J., Hose, K., Courneya, K.S., Harrison, S.J., & Livingston, P.M. (2013). Perceived benefits and barriers to exercise for recently treated patients with multiple myeloma: A qualitative study. BMC Cancer, 13, 319. https://doi.org/10.1186/1471-2407-13-319
Delgado-Calle, J., Bellido, T., & Roodman, G.D. (2014). Role of osteocytes in multiple myeloma bone disease. Current Opinion in Supportive and Palliative Care, 8, 407–413.
Denlinger, C.S., Ligibel, J.A., Are, M., Baker, K.S., Demark-Wahnefried, W., Dizon, D., . . . Freedman-Cass, D.A. (2014). Survivorship: Healthy lifestyles, version 2.2014. Journal of the National Comprehensive Cancer Network, 12, 1222–1237.
Dimopoulos, M.A., Hillengass, J., Usmani, S., Zamagni, E., Lentzsch, S., Davies, F.E., . . . Terpos, E. (2015). Role of magnetic resonance imaging in the management of patients with multiple myeloma: A consensus statement. Journal of Clinical Oncology, 33, 657–664. https://doi .org/10.1200/JCO.2014.57.9961
Dudeney, S., Lieberman, I.H., Reinhardt, M.K., & Hussein, M. (2002). Kyphoplasty in the treatment of osteolytic vertebral compression fractures as a result of multiple myeloma. Journal of Clinical Oncology, 20, 2382–2387. https://doi.org/10.1200/JCO.2002.09.097
Durie, B.G., Kyle, R.A., Belch, A., Bensinger, W., Blade, J., Boccadoro, M., . . . Van Ness, B. (2003). Myeloma management guidelines: A consensus report from the scientific advisors of the International Myeloma Foundation. Hematology Journal, 4, 379–398.
Dworkin, R.H., O’Connor, A.B., Audette, J., Baron, R., Gourlay, G.K., Haanpää, M.L., . . . Wells, C.D. (2010). Recommendations for the pharmacological management of neuropathic pain: An overview and literature update. Mayo Clinic Proceedings, 85(Suppl. 3), S3–S14. https://doi.org/10.4065/mcp.2009.0649
Faiman, B. (2016). Pain management for patients with cancer. In Practice. Retrieved from https://www.inpractice.com/Textbooks/Oncology-Nursing/Symptom-Managemen…
Fallon, M.T. (2013). Neuropathic pain in cancer. British Journal of Anaesthesia, 111, 105–111. https://doi.org/10.1093/bja/aet208
Farrar, J.T., Young, J.P., Jr., LaMoreaux, L., Werth, J.L., & Poole, R.M. (2001). Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain, 94, 149–158.
Felbel, S., Meerpohl, J.J., Monsef, I., Engert, A., & Skoetz, N. (2014). Yoga in addition to standard care for patients with haematological malignancies. Cochrane Database of Systematic Reviews, 6, CD010146. https://doi.org/10.1002/14651858.CD010146.pub2
Greco, M.T., Corli, O., Montanari, M., Deandrea, S., Zagonel, V., & Apolone, G. (2011). Epidemiology and pattern of care of breakthrough cancer pain in a longitudinal sample of cancer patients: Results from the Cancer Pain Outcome Research Study Group. Clinical Journal of Pain, 27, 9–18. https://doi.org/10.1097/AJP.0b013e3181edc250
Greenberg, A.J., Rajkumar, S.V., Therneau, T.M., Singh, P.P., Dispenzieri, A., & Kumar, S.K. (2014). Relationship between initial clinical presentation and the molecular cytogenetic classification of myeloma. Leukemia, 28, 398–403. https://doi.org/10.1038/leu.2013.258
Groeneveldt, L., Mein, G., Garrod, R., Jewell, A.P., Van Someren, K., Stephens, R., . . . Yong, K.L. (2013). A mixed exercise training programme is feasible and safe and may improve quality of life and muscle strength in multiple myeloma survivors. BMC Cancer, 13, 31. https://doi .org/10.1186/1471-2407-13-31
Hadjipavlou, A.G., Tzermiadianos, M.N., Katonis, P.G., & Szpalski, M. (2005). Percutaneous vertebroplasty and balloon kyphoplasty for the treatment of osteoporotic vertebral compression fractures and osteolytic tumours. Journal of Bone and Joint Surgery, 87, 1595–1604. https://doi.org/10.1302/0301-620X.87B12.16074
Han, X., Wang, L., Shi, H., Zheng, G., He, J., Wu, W., . . . Cai, Z. (2017). Acupuncture combined with methylcobalamin for the treatment of chemotherapy-induced peripheral neuropathy in patients with multiple myeloma. BMC Cancer, 17, 40. https://doi.org/10.1186/s12885 -016-3037-z
Hjortholm, N., Jaddini, E., Halaburda, K., & Snarski, E. (2013). Strategies of pain reduction during the bone marrow biopsy. Annals of Hematology, 92, 145–149. https://doi.org/10.1007/s00277-012-1641-9
Johnson, D.C., Corthals, S.L., Walker, B.A., Ross, F.M., Gregory, W.M., Dickens, N.J., . . . Morgan, G.J. (2011). Genetic factors underlying the risk of thalidomide-related neuropathy in patients with multiple myeloma. Journal of Clinical Oncology, 29, 797–804. https://doi .org/10.1200/JCO.2010.28.0792
Jones, L.W., Courneya, K.S., Vallance, J.K., Ladha, A.B., Mant, M.J., Belch, A.R., . . . Reiman, T. (2004). Association between exercise and quality of life in multiple myeloma cancer survivors. Supportive Care in Cancer, 12, 780–788. https://doi.org/10.1007/s00520-004-0668-4
Katz, S., & Akpom, C.A. (1976). A measure of primary sociobiological functions. International Journal of Health Services, 6, 493–508. https://doi.org/10.2190/UURL-2RYU-WRYD-EY3K
Kumar, S.K., Dispenzieri, A., Lacy, M.Q., Gertz, M.A., Buadi, F.K., Pandey, S., . . . Rajkumar, S.V. (2014). Continued improvement in survival in multiple myeloma: Changes in early mortality and outcomes in older patients. Leukemia, 28, 1122–1128.
Kyle, R.A., & Rajkumar, S.V. (2009). Criteria for diagnosis, staging, risk stratification and response assessment of multiple myeloma. Leukemia, 23, 3–9. https://doi.org/10.1038/leu.2008.291
Lane, J.M., Hong, R., Koob, J., Kiechle, T., Niesvizky, R., Pearse, R., . . . Poynton, A.R. (2004). Kyphoplasty enhances function and structural alignment in multiple myeloma. Clinical Orthopaedics and Related Research, 426, 49–53.
Larocca, A., & Palumbo, A. (2015). How I treat fragile myeloma patients. Blood, 126, 2179–2185. https://doi.org/10.1182/blood-2015-05-612960
Lawton, M.P., & Brody, E.M. (1969). Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist, 9, 179–186.
Maida, V., & Daeninck, P.J. (2016). A user’s guide to cannabinoid therapies in oncology. Current Oncology, 23, 398–406. https://doi.org/10.3747/co.23.3487
McMenamin, E. (2011). Cancer pain management. In C.H. Yarbro, D. Wujcik, & B.H. Gobel (Eds.), Cancer nursing: Principles and practice (7th ed., pp. 685–712). Burlington, MA: Jones and Bartlett Learning.
Melnyk, B.M., & Fineout-Overholt, E. (2011). Evidence-based practice in nursing and healthcare: A guide to best practice (2nd ed., p. 13). Philadelphia, PA: Lippincott Williams and Wilkins.
Mesgarpour, B., Griebler, U., Glechner, A., Kien, C., Strobelberger, M., Van Noord, M.G., & Michalek-Sauberer, A. (2014). Extended-release opioids in the management of cancer pain: A systematic review of efficacy and safety. European Journal of Pain, 18, 605–616. https://doi.org/10.1002/j.1532-2149.2013.00401.x
Miaskowski, C. (2010). Cancer pain. In C.G. Brown (Ed.), A guide to oncology symptom management (pp. 369–403). Pittsburgh, PA: Oncology Nursing Society.
Miceli, T.S., Colson, K., Faiman, B.M., Miller, K., & Tariman, J.D. (2011). Maintaining bone health in patients with multiple myeloma: Survivorship care plan of the International Myeloma Foundation Nurse Leadership Board. Clinical Journal of Oncology Nursing, 15(Suppl.), S9–S23. https://doi.org/10.1188/11.S1.CJON.9-23
Mohty, B., El-Cheikh, J., Yakoub-Agha, I., Moreau, P., Harousseau, J.L., & Mohty, M. (2010). Peripheral neuropathy and new treatments for multiple myeloma: Background and practical recommendations. Haematologica, 95, 311–319.
Moreau, P., Pylypenko, H., Grosicki, S., Karamanesht, I., Leleu, X., Grishunina, M., . . . Harousseau, J.L. (2011). Subcutaneous versus intravenous administration of bortezomib in patients with relapsed multiple myeloma: A randomised, phase 3, non-inferiority study. Lancet Oncology, 12, 431–440. https://doi.org/10.1016/S1470-2045(11)70081-X
Moulin, D., Boulanger, A., Clark, A.J., Clarke, H., Dao, T., Finley, G.A., . . . Williamson, O.D. (2014). Pharmacological management of chronic neuropathic pain: Revised consensus statement from the Canadian Pain Society. Pain Research and Management, 19, 328–335.
Mustian, K.M., Cole, C.L., Lin, P.J., Asare, M., Fung, C., Janelsins, M.C., . . . Magnuson, A. (2016). Exercise recommendations for the management of symptoms clusters resulting from cancer and cancer treatments. Seminars in Oncology Nursing, 32, 383–393.
Mustian, K.M., Sprod, L.K., Janelsins, M., Peppone, L.J., & Mohile, S. (2012). Exercise recommendations for cancer-related fatigue, cognitive impairment, sleep problems, depression, pain, anxiety, and physical dysfunction: A review. Oncology and Hematology Review, 8, 81–88.
National Comprehensive Cancer Network. (2017a). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Adult cancer pain [v.2.2017]. Retrieved from http:www.nccn .org/professionals/physician_gls/pdf/pain.pdf
National Comprehensive Cancer Network. (2017b). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Cancer-related fatigue [v.2.2017]. Retrieved from http:www.nccn.org/professionals/physician_gls/pdf/fatigue.pdf
National Comprehensive Cancer Network. (2017c). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Multiple myeloma [v.3.2017]. Retrieved from http:www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf
National Comprehensive Cancer Network. (2017d). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Older adult oncology [v.2.2017]. Retrieved from http:www .nccn.org/professionals/physician_gls/pdf/senior.pdf
National Comprehensive Cancer Network. (2017e). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Survivorship [v.1.2017]. Retrieved from http:www.nccn.org/professionals/physician_gls/pdf/survivorship.pdf
National Osteoporosis Foundation. (2017). Get the facts on calcium and vitamin D. Retrieved from https://www.nof.org/patients/treatment/calciumvitamin-d/get-the-facts-o…
Palumbo, A., Bringhen, S., Mateos, M.V., Larocca, A., Facon, T., Kumar, S.K., . . . Rajkumar, S.V. (2015). Geriatric assessment predicts survival and toxicities in the elderly myeloma patients: An International Myeloma Working Group report. Blood, 125, 2068–2074.
Pawlyn, C., Gay, F., Larocca, A., Roy, V., & Ailawadhi, S. (2016). Nuances in the management of older people with multiple myeloma. Current Hematologic Malignancy Reports, 11, 241–251. https://doi.org/10.1007/s11899-016-0323-4
Portenoy, R.K. (2011). Treatment of cancer pain. Lancet, 377, 2236–2247. https://doi.org/10.1016/S0140-6736(11)60236-5
Pozzi, S., & Raje, N. (2011). The role of bisphosphonates in multiple myeloma: Mechanisms, side effects, and the future. Oncologist, 16, 651–662.
Rajkumar, S.V. (2009). Multiple myeloma. Current Problems in Cancer, 33, 7–64. https://doi.org/10.1016/j.currproblcancer.2009.01.001
Rajkumar, S.V. (2011a). Multiple myeloma: 2011 update on diagnosis, risk-stratification, and management. American Journal of Hematology, 86, 57–65. https://doi.org/10.1002/ajh.21913
Rajkumar, S.V. (2011b). Treatment of multiple myeloma. Nature Reviews Clinical Oncology, 8, 479–491. https://doi.org/10.1038/nrclinonc.2011.63
Rajkumar, S.V., Dimopoulos, M.A., Palumbo, A., Blade, J., Merlini, G., Mateos, M.V., . . . Miguel, J.F. (2014). International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncology, 15, e538–e548. https://doi.org/10.1016/S1470-2045(14)70442-5
Rajkumar, S.V., Larson, D., & Kyle, R.A. (2011). Diagnosis of smoldering multiple myeloma. New England Journal of Medicine, 365, 474–475. https://doi.org/10.1056/NEJMc1106428
Regelink, J.C., Minnema, M.C., Terpos, E., Kamphuis, M.H., Raijmakers, P.G., Pieters-van den Bos, I.C., . . . Zweegman, S. (2013). Comparison of modern and conventional imaging techniques in establishing multiple myeloma-related bone disease: A systematic review. British Journal of Haematology, 162, 50–61. https://doi.org/10.1111/bjh.12346
Richards, T., & Brigle, K. (2016). Palliative care in multiple myeloma. Journal of the Advanced Practitioners in Oncology, 7, 31–43. https://doi.org/10.6004/jadpro.2016.7.2.12
Richardson, P.G., Delforge, M., Beksac, M., Wen, P., Jongen, J.L., Sezer, O., . . . Sonneveld, P. (2012). Management of treatment-emergent peripheral neuropathy in multiple myeloma. Leukemia, 26, 595–608. https://doi.org/10.1038/leu.2011.346
Ripamonti, C.I., Santini, D., Maranzano, E., Berti, M., & Roila, F. (2012). Management of cancer pain: ESMO Clinical Practice Guidelines. Annals of Oncology, 23(Suppl. 7), vii139–vii154. https://doi.org/10.1093/annonc/mds233
Rock, C.L., Doyle, C., Demark-Wahnefried, W., Meyerhardt, J., Courneya, K.S., Schwartz, A.L., . . . Gansler, T. (2012). Nutrition and physical activity guidelines for cancer survivors. CA: A Cancer Journal for Clinincians, 62(4), 243–274. https://doi.org/10.3322/caac.21142
Rome, S.I., Jenkins, B.S., & Lilleby, K.E. (2011). Mobility and safety in the multiple myeloma survivor: Survivorship care plan of the International Myeloma Foundation Nurse Leadership Board. Clinical Journal of Oncology Nursing, 15(Suppl.), S41–S52. https://doi.org/10.1188/11.S1.CJON.41-52
Salhofer, I., Will, A., Monsef, I., & Skoetz, N. (2016). Meditation for adults with haematological malignancies. Cochrane Database of Systematic Reviews, 2, CD011157.
Schmitz, K.H., Courneya, K.S., Matthews, C., Demark-Wahnefried, W., Galvão, D.A., Pinto, B.M., . . . Schwartz, A.L. (2010). American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Medicine and Science in Sports and Exercise, 42, 1409–1426. https://doi.org/10.1249/MSS.0b013e3181e0c112
Shallwani, S., Dalzell, M.A., Sateren, W., & O’Brien, S. (2015). Exercise compliance among patients with multiple myeloma undergoing chemotherapy: A retrospective study. Supportive Care in Cancer, 23, 3081–3088. https://doi.org/10.1007/s00520-015-2680-2
Siontis, B., Kumar, S., Dispenzieri, A., Drake, M.T., Lacy, M.Q., Buadi, F., . . . Rajkumar, S.V. (2015). Positron emission tomography-computed tomography in the diagnostic evaluation of smoldering multiple myeloma: Identification of patients needing therapy. Blood Cancer Journal, 5, e364. https://doi.org/10.1038/bcj.2015.87
Smith, L., McCourt, O., Henrich, M., Paton, B., Yong, K., Wardle, J., & Fisher, A. (2015). Multiple myeloma and physical activity: A scoping review. BMJ Open, 5(11), e009576.
Tariman, J.D., Love, G., McCullagh, E., & Sandifer, S. (2008). Peripheral neuropathy associated with novel therapies in patients with multiple myeloma: Consensus statement of the IMF Nurse Leadership Board. Clinical Journal of Oncology Nursing, 12(Suppl. 3), S29–S36. https://doi.org/10.1188/08.CJON.S1.29-35
Tateo, S. (2017). State of the evidence: Cannabinoids and cancer pain—A systematic review. Journal of the American Association of Nurse Practitioners, 29, 94–103.
Terpos, E., Kleber, M., Engelhardt, M., Zweegman, S., Gay, F., Kastritis, E., . . . Palumbo, A. (2015). European Myeloma Network guidelines for the management of multiple myeloma-related complications. Haematologica, 100, 1254–1266.
Terpos, E., Morgan, G., Dimopoulos, M.A., Drake, M.T., Lentzsch, S., Raje, N., . . . Roodman, G.D. (2013). International Myeloma Working Group recommendations for the treatment of multiple myeloma-related bone disease. Journal of Clinical Oncology, 31, 2347–2357. https://doi.org/10.1200/JCO.2012.47.7901
Tetrault, J.M., Crothers, K., Moore, B.A., Mehra, R., Concato, J., & Fiellin, D.A. (2007). Effects of marijuana smoking on pulmonary function and respiratory complications: A systematic review. Archives of Internal Medicine, 167, 221–228. https://doi.org/10.1001/archinte.167.3.221
Tuchman, S.A., Shapiro, G.R., Ershler, W.B., Badros, A., Cohen, H.J., Dispenzieri, A., . . . Yates, J.W. (2014). Multiple myeloma in the very old: An IASIA conference report. Journal of the National Cancer Institute, 106, dju067. https://doi.org/10.1093/jnci/dju067
Wolin, K.Y., Schwartz, A.L., Matthews, C.E., Courneya, K.S., & Schmitz, K.H. (2012). Implementing the exercise guidelines for cancer survivors. Journal of Supportive Oncology, 10(5), 171–177. https://doi.org/10.1016/j.suponc.2012.02.001
World Health Organization. (1990). Cancer pain relief and palliative care: Report of a WHO expert committee. Retrieved from http://apps.who.int/iris/bitstream/10665/39524/1/WHO_TRS_804.pdf
World Health Organization. (2017). WHO’s cancer pain ladder for adults. Retrieved from http://www.who.int/cancer/palliative/painladder/en/




