Promoting Oral Therapy Adherence: Consensus Statements From the Faculty of the Melanoma Nursing Initiative on Oral Melanoma Therapies
Background: Inhibitors of BRAF and the downstream signaling protein MEK have improved outcomes for patients with BRAF-mutant advanced malignant melanoma. Despite their ease of administration, these oral therapies pose adherence challenges.
Objectives: This article aims to increase awareness of causes of nonadherence to oral targeted therapies in advanced malignant melanoma and to provide oncology nurses with strategies to address these nonadherence issues.
Methods: Members of the Melanoma Nursing Initiative explored issues related to adherence to targeted therapies in advanced malignant melanoma. The current literature and clinical experience were reviewed.
Findings: The authors present a care step pathway focused on increased patient engagement and rapid identification and optimal management of toxicities to avoid toxicity-related nonadherence. Other causes for nonadherence and employment of individualized strategies to support patient adherence are addressed.
Jump to a section
Therapies targeted to the mitogen-activated protein kinase (MAPK) pathway have improved the treatment options for patients with v-Raf murine sarcoma viral oncogene homolog B (BRAF)–mutant metastatic or unresectable advanced melanoma, increasing response rates and rates of overall and progression-free survival (Chapman et al., 2011; Flaherty et al., 2012; Larkin et al., 2014; McArthur et al., 2014; Robert et al., 2015; Welsh & Corrie, 2015). The targeted therapies approved by the U.S. Food and Drug Administration for use in melanoma are cobimetinib (Cotellic®), dabrafenib (Tafinlar®), trametinib (Mekinist®), and vemurafenib (Zelboraf®). Although generally well tolerated, these oral medications are associated with toxicity profiles unlike those of traditional cytotoxic chemotherapy. Clinical trials are examining their application in the adjuvant or neoadjuvant setting, and newer BRAF/mitogen-activated protein kinase kinase (MEK) agents are in development (Bendell et al., 2017; van Zeijl, van den Eertwegh, Haanen, & Wouters, 2017). Therefore, the important role of BRAF/MEK inhibitors in melanoma is likely to expand.
Researchers have categorized factors affecting adherence to long-term medications (Sabaté, 2003) and interventions to support adherence to oral anticancer medications in particular (Timmers et al., 2017). Oral delivery presents challenges for patient engagement and adherence that differ from those associated with IV administration (Weingart et al., 2008). Patients with cancer are viewed as particularly motivated to adhere to anticancer regimens (Weingart et al., 2008). In the authors’ clinical experience, although patients with advanced malignant melanoma are usually highly invested in their therapy, they still face obstacles to adherence (Schneider, Adams, & Gosselin, 2014), such as delays in accessing drugs from specialty pharmacies, high co-pays and coinsurance, difficulty following complex dosing regimens, reluctance to report adverse events, and challenges to management of adverse events.
As trusted members of the oncology care team, nurses are well equipped to deliver interventions promoting patient adherence to oral medication regimens (Schneider et al., 2014; Schneider, Hess, & Gosselin, 2011). For the purposes of development of this article, members of the Melanoma Nursing Initiative (MNI) focused specifically on identifying and addressing factors that affect adherence to oral targeted therapies introduced for metastatic/unresectable melanoma. The authors convened to discuss the current literature and their clinical experience regarding adherence to oral advanced malignant melanoma therapies and interventions to increase adherence, then drafted the care step pathway, which illustrates strategies to engage patients with BRAF-mutant advanced malignant melanoma and foster adherence to targeted oral therapy (see Figure 1). This article outlines interventions nurses can incorporate to improve adherence at each of the care steps in the patient treatment journey.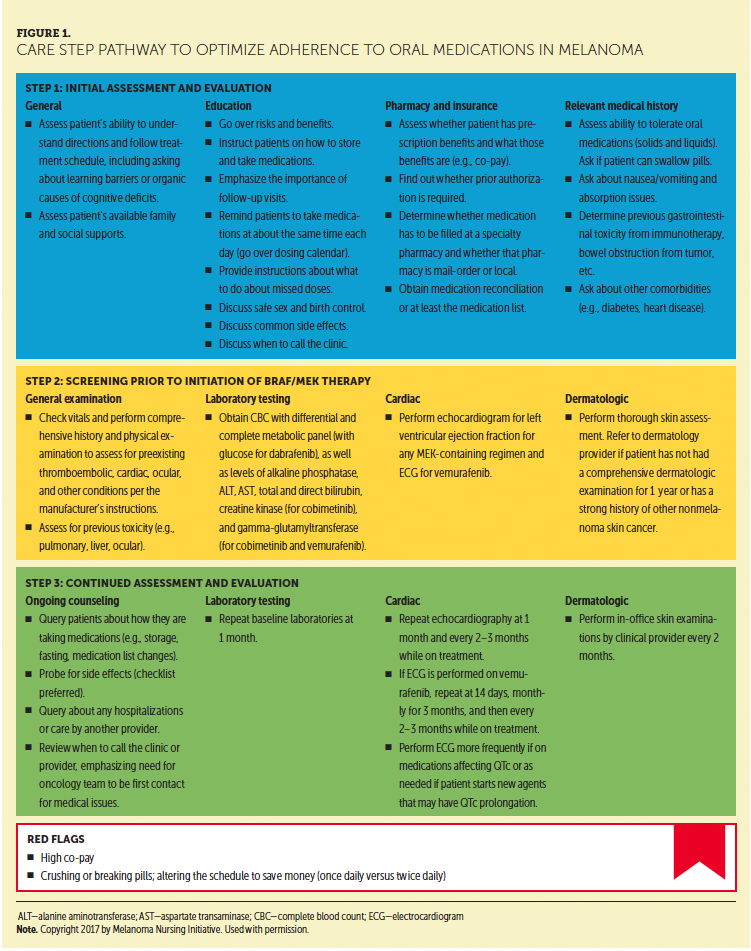
Findings and Recommendations to Improve Adherence
Pretreatment
Pretreatment assessment should include identification of patient- and disease-related factors that may affect the patient’s ability to adhere to the prescribed medication regimen. Cognitive ability, learning style, and family/social support should be evaluated (Schneider et al., 2011; Shinnick & Woo, 2015). Cognitive limitations owing to organic causes, such as brain metastases or dementia, or other learning barriers may hinder the patient’s ability to follow dosing instructions and/or to observe and report adverse events. Asking patients to verbally demonstrate their understanding by using the teach-back method can serve as a check on the patient’s comprehension (Agency for Healthcare Research and Quality, 2015).
Inadequate social support has been associated with nonadherence to oral medication (Partridge, Avorn, Wang, & Winer, 2002; Weingart et al., 2008). Family or friends can report adverse events, provide reminders to take medication, encourage patients to attend regular follow-up visits, and transport patients to those visits if necessary. Patient education is best delivered with a family member or caregiver present (Clay & Parsh, 2016). If the patient is not accompanied to a clinic visit by an involved caregiver, the nurse should ask the patient for a contact who can help with these issues. This question may prompt the patient to identify a supportive family member or friend. However, some patients are reluctant to ask family or friends for assistance. Others may not have individuals in their lives who can provide support and may be reluctant to share that information with the nurse or other members of the clinical team. A social worker, if one is available to the practice, should be engaged to connect such patients to local resources for services, such as transportation to medical appointments and assistance with medication cost.
Education
Patients’ knowledge, attitudes, beliefs, perceptions, and expectations about the course of the disease and their ability to manage their illness and treatment regimen affect adherence (Sabaté, 2003; Timmers et al., 2017). Patients may perceive oral medication as less important or effective or as safer and more forgiving of dosage and administration errors as compared to IV therapy (Weingart et al., 2008). Communicating key results from clinical trials, including risks, benefits, and expected treatment outcomes, may motivate patients to integrate the dosing schedule, follow-up appointments, and management of adverse events into their lives.
Adverse events affect adherence. In one study, one-third of those who discontinued oral anticancer medications reported side effects as a reason (Timmers et al., 2014). Prompt identification and management of adverse events are crucial for ensuring that patients are able to continue treatment (Welsh & Corrie, 2015). This point needs to be emphasized to patients before treatment initiation and throughout the course of therapy. Patients may not report side effects out of fear that therapy could be discontinued (Weingart et al., 2008). Some may choose to skip doses because of the discomfort or pain of particular symptoms (Schneider et al., 2014). Patients should be reminded about when to call the clinic or healthcare provider regarding adverse events.
Explaining that the clinical team shares the goals of continuing therapy and optimizing outcomes as long as the treatment is working and the patient is tolerating it is important. Those goals can be achieved safely. Prompt reporting of adverse events can allow for immediate intervention and possible dose modifications, as appropriate, in lieu of discontinuation of therapy. However, waiting until an adverse event has become severe may necessitate treatment cessation. Self-medicating to alleviate adverse events may lead to other problems, such as drug–drug interactions or worsening or development of additional adverse events. Of note, family members often provide valuable insight into the clinical situation and may report symptoms that patients minimize or fail to mention.
Patients receiving oral therapy instead of IV therapy may not adhere to the recommended schedule for follow-up appointments. Asking open-ended, nonjudgmental questions about the reasons for not keeping follow-up appointments and acknowledging the legitimacy of the issues the patient identifies are helpful communication techniques. Such dialogue may open a way to address those obstacles and serve as a teachable moment about the importance of periodic assessments (see Table 1). Stressing to patients that periodic assessments are crucial to evaluate toxicity and efficacy and maintain treatment safety is important. Establishing “no refills” on initial prescriptions can encourage patients to return to the office. 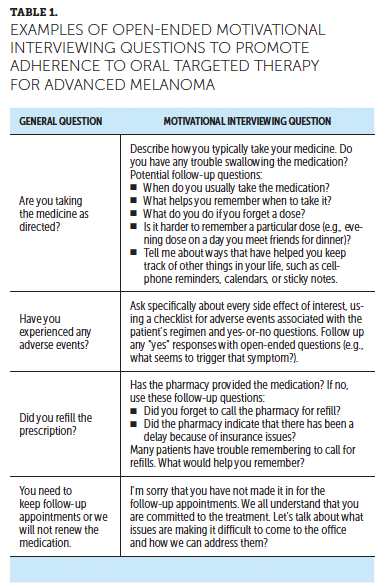
Following Complex Regimens
Greater regimen complexity has been associated with lower levels of adherence (Schneider et al., 2011; Spoelstra et al., 2013). In one analysis, noncontinuous oral regimens have been associated with lower adherence rates than continuous oral treatment schedules (88% for a continuous schedule versus 35% and 33% for two intermittent schedules) (Spoelstra et al., 2013). The combination of cobimetinib and vemurafenib is an example of two different dosing schedules within the same regimen. Vemurafenib is given continuously, and cobimetinib is only taken for three weeks out of a four-week cycle. In addition, vemurafenib is taken twice daily (roughly 12 hours apart), whereas cobimetinib is a once-daily medication (see Figure 2). In the dabrafenib/trametinib combination, dabrafenib is taken twice a day, whereas trametinib is taken once daily (see Figure 3). Given these complex dosing schedules, patient education by the nurse is paramount.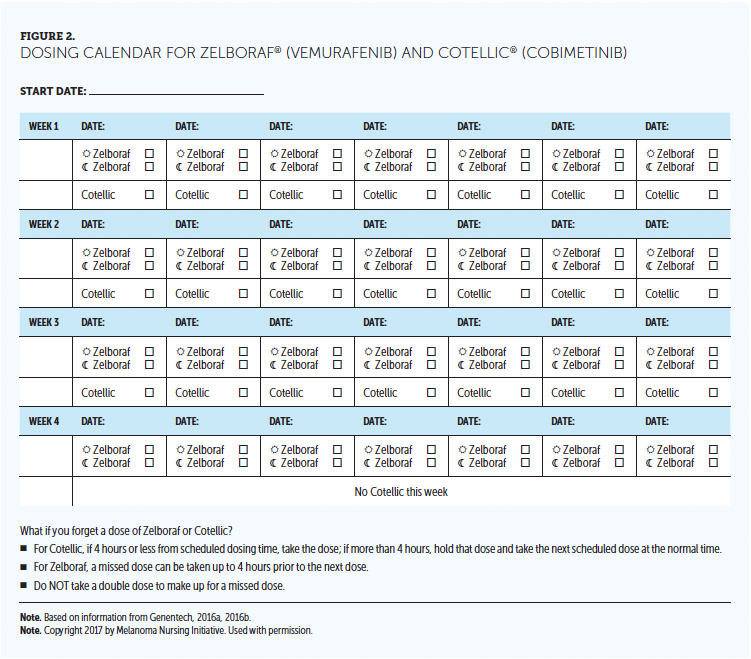
Anticipatory guidance to help patients identify strategies to manage these complex regimens is an important component of nursing care. Available downloadable resources to support adherence are listed in Figure 4. Dosing calendars; pillboxes; smartphone alarms; and reminders on mirrors, doors, or laptop computers may help, depending on the patient’s lifestyle and preference (Schneider et al., 2011). However, a study by Choudry et al. (2017) found that three types of pillboxes with different reminder devices did not increase the odds of optimal adherence as compared to a standard pillbox. These findings suggest that behavioral interventions are needed in addition to technological reminders. The nurse should assess which techniques are likely to work for the individual patient, possibly by querying him or her about strategies that currently work as reminder systems for the patient in other aspects of life.
[[{"type":"media","view_mode":"media_original","fid":"34071","field_deltas":{"1":{}},"link_text":null,"fields":{},"attributes":{"height":"662","width":"749","class":"media-image media-element file-media-original","data-delta":"1"}}]]
Patients with preexisting nausea and vomiting may find it challenging to swallow medications first thing in the morning after an overnight fast. In these situations, the nurse can advise patients to eat breakfast and then wait two hours to take dabrafenib or trametinib, providing an adequate fast period that is more tolerable than an overnight fast. Patients should also be instructed about what to do if they miss a scheduled dose. Dosing calendars can address this question for each regimen.
Medication Storage and Disposal
Trametinib must be kept refrigerated and stored in the original bottle with the lid closed tightly to protect the pills from heat, light, and moisture (Novartis, 2016). The nurse should instruct patients to keep all medications with them when traveling and not to place them in a stored baggage area, where temperatures are not controlled. In addition, trametinib bottles can be kept in refrigerated lunch packs. If the patient inadvertently leaves an opened bottle of trametinib out of the refrigerator, temperature excursion data have shown that the medication is not damaged by storage outside the refrigerator for as many as 30 days if it is maintained at a temperature below 86°F (30°C) (Novartis, 2017). Therefore, the nurse can typically advise the patient who has inadvertently left the medication out to simply keep the medication cool and get it back in the refrigerator as soon as possible.
Contraception and Family Planning
Before initiating any of the targeted therapies, patients must be counseled about pregnancy, fertility, and contraception. All four oral targeted agents approved for metastatic/unresectable melanoma can cause fetal harm. Some patients view themselves as sterile because of prior anticancer therapies or assume that they will be rendered sterile by targeted therapy. It is important to emphasize the need for effective birth control even in the face of these beliefs. Women of childbearing age must be advised to use effective birth control during treatment and for at least two weeks (for cobimetinib, dabrafenib, and vemurafenib) to four months (for trametinib) after treatment ends (Genentech, 2016a, 2016b; Novartis, 2015, 2016). Dabrafenib can interact with hormonal contraceptives (oral pills, injections, or patches), rendering them ineffective; therefore, women must be counseled to use nonhormonal contraception during and immediately after treatment with this drug (Novartis, 2016). Many practices advise patients to use two nonhormonal (barrier) methods of contraception to increase effectiveness.
Impact on fertility in humans is not known, but cobimetinib, dabrafenib, and trametinib have been shown to impair fertility in male and female animal models (Genentech, 2016a, 2016b; Novartis, 2015, 2016). Dabrafenib may irreversibly limit fertility in males (Novartis, 2016). Men should be counseled regarding decreased chances of impregnating a female partner while receiving treatment with any of the four targeted agents (OncoLink, 2015a, 2015b, 2016a, 2016b).
Beyond immediate fertility and pregnancy effects, patients may have questions surrounding future family planning. Nurses should be prepared to discuss options for sperm banking and egg freezing and offer supportive counseling regarding the prospect of family planning in the setting of advanced melanoma. Prognosis and life expectancy should be considered. Such discussions may become more commonplace if targeted therapies become available for use in the adjuvant setting.
Pharmacy and Insurance Issues
Cost and Access
Because most oral medications are covered under prescription insurance plans versus major medical plans (as with IV infusions), cost can be a significant barrier to medication initiation and refill (Schneider et al., 2014). To save money, patients with high out-of-pocket costs (i.e., co-pays or coinsurance) might delay treatment initiation or refills or dose less frequently than prescribed (Winn, Keating, & Dusetzina, 2016; Zafar et al., 2013). Patients covered by commercial, which must include prescription drug coverage, rather than government (e.g., Medicare, Medicaid) insurance have access to manufacturer co-pay cards that reduce patient financial responsibility. In addition, manufacturers of the targeted therapies for melanoma offer resources to reduce co-pays and connect patients on government insurance to foundations or other organizations that may offer assistance with the patient’s share of medication cost (see Figure 4).
[[{"type":"media","view_mode":"media_original","fid":"34076","field_deltas":{"2":{}},"link_text":null,"fields":{},"attributes":{"height":"644","width":"752","class":"media-image media-element file-media-original","data-delta":"2"}}]]
Health system barriers to obtaining and refilling medication in a timely manner may affect a substantial number of patients (Schneider et al., 2014). Obstacles can include (a) a requirement to use a specialty or mail-order pharmacy, (b) delays while processing a prior authorization, and (c) a requirement for a signature if delivered to home. Of particular concern is expediting access to the initial prescription.
Patients and pharmacies may not understand the urgency of promptly initiating treatment in the setting of a life-threatening disease. Nurses should impress on patients and their family members that immediately returning pharmacy calls is crucial to starting and maintaining medication supply. Manufacturers of targeted therapies offer support to facilitate prior authorizations and otherwise speed access to therapy. The nurse or other healthcare team members should provide assistance with clearing the barriers to drug access as much as possible.
Current Medications
Patients receiving medications to manage comorbid conditions, control cancer symptoms, or manage toxicities of previous anticancer therapies may be at high risk for drug–drug interactions. As with any drug therapy, a pharmacist should ideally review the patient’s medications, identify absolute and relative contraindications, and suggest possible dose adjustments or alternate medications to prevent or reduce the risk of drug–drug interactions. The specialty pharmacy contracted with the patient’s insurer may be able to offer this reconciliation service (Weingart et al., 2008). Pharmacy departments at academic medical centers and comprehensive cancer centers may provide a pharmacy consultation for patients even if the insurer will not cover prescriptions filled at those centers. In the absence of these pharmacy resources, the nurse should obtain a list of current medications, which includes any over-the-counter medications, herbals, supplements, and vitamins. It is important to explain to patients that some of these substances can lead to drug–drug interactions, which can increase the risk for toxicities or render the targeted melanoma therapy less efficacious (Carrington, 2015; Genentech, 2016a; Welsh & Corrie, 2015).
Relevant Medical History
Toxicities associated with targeted therapies can affect organ systems that already may be compromised by comorbid conditions and/or previous melanoma therapies. The average age at melanoma diagnosis is 63 years (American Cancer Society, 2017). Cardiovascular disease, diabetes, and certain visual impairments become more common in older adults, and adverse events can exacerbate these conditions. Patients with these conditions may take other medications, potentially raising the risk of drug–drug interactions (Spoelstra et al., 2013). In addition, patients with more comorbidities may report higher severity of adverse events to oral anticancer medications, as observed in a study by Spoelstra et al. (2015). Patients who have received prior anticancer regimens may have residual toxicities before starting targeted therapy. A baseline assessment is important to identify patients whose health is already compromised and to distinguish emerging adverse events from preexisting conditions.
Screening
Pretreatment screening should include assessment of vital signs, laboratory values, blood pressure, oxygen saturation, and cardiac function testing. Combination targeted therapies can cause fever, cardiomyopathy, arrhythmias, endocrinopathies, and hepatotoxicity (Genentech, 2016a; 2016b; Novartis, 2015, 2016; Welsh & Corrie, 2015). Creatine kinase is measured before and during cobimetinib administration because of risk of rhabdomyolysis (Genentech, 2016a). Gamma-glutamyltransferase is assessed for patients prescribed cobimetinib or vemurafenib because these drugs can cause elevation of this specific liver enzyme (Genentech, 2016a, 2016b). Liver function and kidney function should be checked on a periodic basis.
BRAF inhibitors are associated with the development of nonmelanoma skin cancers (keratinocyte carcinomas, such as basal cell and squamous cell carcinomas) and new primary melanoma malignancies. Patients should undergo a skin examination before treatment initiation and every two months during treatment and for as many as six months after treatment cessation (Genentech, 2016a, 2016b; Novartis, 2015, 2016). The MNI advises that individuals who have not had a comprehensive dermatologic examination within the past year or who have a history of basal cell or squamous cell carcinoma should receive a skin examination from a dermatologist with expertise in cutaneous malignancies prior to starting targeted therapy. Although the clinical trials using BRAF combination therapies require baseline ocular examinations by ophthalmology, oncology practices typically reserve these examinations for patients who develop symptoms of ocular toxicities. In addition, at baseline, evaluation of ejection fraction is needed when using an oral MEK inhibitor, and an electrocardiogram is recommended with use of vemurafenib (Genentech, 2016a, 2016b; Novartis, 2015, 2016).
Anticipatory Guidance During Treatment
Ongoing Patient Counseling
Toxicity education and monitoring are key roles of the oncology nurse. Monitoring between subsequent cycles promotes early detection of adverse events or other obstacles to adherence so that these issues can be addressed promptly. For example, pyrexia occurring with dabrafenib-containing regimens often requires active pharmacologic management and potential dose reduction. In addition, visits or telephone calls represent a chance to evaluate patient psychosocial status and offer emotional support (Schneider et al., 2011). In a study by Schneider et al. (2014), a nurse-coaching intervention delivered through telephone calls was associated with higher pharmacy refill rates compared with standard pretreatment education alone. During the calls, nurses discussed factors that could affect adherence, identified any issues interfering with the patient taking medication as prescribed, and brainstormed with the patient on strategies to address those issues. Such an approach is likely to be helpful to patients receiving targeted therapy. Every visit should include asking the patient about the routine employed for taking medication, any new symptoms or signs of adverse events, new disease symptoms, changes in other medications, and visits to other clinicians.
Clinical Examination and Testing During Follow-Up
Anticipatory guidance should include explaining to patients that vital signs must be measured at every follow-up visit. Regular monitoring promotes early detection of any adverse events and facilitates early management. Laboratory measures should be evaluated at follow-up visits and more frequently if clinically indicated (Genentech, 2016a, 2016b; Novartis, 2015, 2016). Echocardiography should be followed as recommended.
Because cutaneous toxicity is common with BRAF/MEK inhibitors, frequent skin examinations during treatment are important to screen for new melanomas and keratinocyte carcinomas, as well as to detect rashes, papillomas, and calluses. Dabrafenib and trametinib each carry a warning about palmar-plantar erythrodysesthesia, also called hyperkeratosis or hand-foot syndrome. This toxicity is also reported with vemurafenib (Anforth et al., 2012; Genentech, 2016a, 2016b; Novartis, 2015, 2016; Vanneste, Wolter, Van den Oord, Stas, & Garmyn, 2015). Patients may not recognize the redness, swelling, and pain on the palms of the hands and/or the soles of the feet as a treatment side effect. Hyperkeratotic lesions also can develop in other areas of the body (Livingstone, Zimmer, Vaubel, & Schadendorf, 2014). Palmar-plantar hyperkeratosis can be managed by counseling patients to minimize pressure and friction on the areas; wear soft, broad shoes; and use topical keratolytic medications containing urea, salicylic acid, or, rarely, steroids. Frequent paring by a podiatrist can relieve pressure and pain (Livingstone et al., 2014).
Eliciting Adverse Events and Nonadherence
When probing for adverse events of therapy, specific rather than general queries are more likely to elicit informative responses. Patients may respond “no” when asked if they have experienced any adverse events, but asking about every possible side effect may uncover new symptoms. Open-ended questions (e.g., what bothersome symptoms have you experienced since your last visit?) also may elicit information. Patients may not detect some changes or may view them as insignificant and, therefore, not mention them to the nurse or others on the clinical team. The authors recommend use of a structured checklist to probe for adverse events (see Figure 5).
If nonadherence is suspected, the nurse should continue to connect with the patient while showing acceptance, empathy, and support, which can facilitate trust and foster communication (Komatsu & Yagasaki, 2014). Motivational interviewing is one strategy for building a connection. A technique developed by Miller and Rollnick (2013) in addiction treatment, motivational interviewing has been defined as “a collaborative conversation style for strengthening a person’s own motivation and commitment to change” (p. 10). It builds on the person’s motivation for change rather than leaving the individual feeling defensive or shamed for not engaging in a certain behavior (Miller & Rollnick, 2013). Techniques drawing on the principles of motivational interviewing have improved patient self-management in multiple disease states. A study applying these techniques to promote adherence to oral anticancer therapies is in progress (Spoelstra, Burhenn, DeKoekkoek, & Schueller, 2016). 
Conclusion
Medication adherence issues associated with oral therapy for advanced melanoma differ from those observed with other treatment delivery routes. Responsibility for administration and self-management of potentially life-prolonging therapy shifts from the clinical team to the patient. The life-threatening nature of the disease, characteristics of the patient population, medication adverse events, insurance coverage, and access issues with oral medication create adherence obstacles in this setting. The assertive approach outlined in this article to elicit adverse events and address barriers to adherence should increase the likelihood of patients staying on these therapies and lead to improved outcomes.
The authors gratefully acknowledge Jill Maria Weberding, MPH, BSN, RN, OCN®, for reviewing the manuscript from the community oncology nursing perspective, and Lois J. Loescher, PhD, RN, FAAN, for reviewing the manuscript from a nursing education perspective.
About the Author(s)
Lisa A. Kottschade, APRN, MSN, CNP, is a nurse practitioner and assistant professor of oncology in the Department of Oncology at the Mayo Clinic in Rochester, MN; and Mollie Lehner Reed, MSN, RN, ACNP, is a nurse practitioner affiliated with Tennessee Oncology in Nashville. The authors take full responsibility for this content. This study was funded by the AIM at Melanoma Foundation, with support via unrestricted grants from Amgen, Array Biopharma, Bristol-Myers Squibb, Incyte Corporation, Merck and Co., and Novartis Pharmaceuticals. Writing and editorial support was provided by Eileen McCaffrey, MA, of the Content Firm, LLC; Lisa A. Tushla, PhD, H(ASCP), of Terranova Medica; and Marjorie Joyce, BA. Kottschade has previously consulted for Novartis Pharmaceuticals, Bristol-Myers Squibb, Genentech, and Merck and Co., and has received support from Bristol-Myers Squibb. Reed has previously consulted for or has served on research boards for Clovis Oncology, Novartis Pharmaceuticals, AstraZeneca, Taiho Oncology, Prometheus, and Ispen, and has served on speakers bureaus for Bristol-Myers Squibb, Genentech, Merck and Co., Pfizer, Novartis Pharmaceuticals, and Taiho Oncology. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Kottschade can be reached at kottschade.lisa@mayo.edu, with copy to CJONEditor@ons.org. (Submitted April 2017. Accepted June 4, 2017.)
References
Agency for Healthcare Research and Quality. (2015). Use the teach-back method: Tool #5. Rockville, MD: Author. Retrieved from http://bit.ly/2uNDbo2
American Cancer Society. (2017). Key statistics for melanoma skin cancer. Retrieved from https://www.cancer.org/cancer/melanoma-skin-cancer/about/key-statistics…
Anforth, R.M., Blumetti, T.C., Kefford, R.F., Sharma, R., Scolyer, R.A., Kossard, S., . . . Fernandez-Peñas, P. (2012). Cutaneous manifestations of dabrafenib (GSK2118436): A selective inhibitor of mutant BRAF in patients with metastatic melanoma. British Journal of Dermatology, 167, 1153–1160. doi:10.1111/j.1365-2133.2012.11155.x
Bendell, J.C., Javle, M., Bekaii-Saab, T.S., Finn, R.S., Wainberg, Z.A., Laheru, D.A., . . . Patnaik, A. (2017). A phase 1 dose-escalation and expansion study of binimetinib (MEK162), a potent and selective oral MEK1/2 inhibitor. British Journal of Cancer, 116, 575–583. doi:10.1038/bjc.2017.10
Carrington, C. (2015). Oral targeted therapy for cancer. Australian Prescriber, 38(5), 171–176. doi:10.18773/austprescr.2015.060
Chapman, P.B., Hauschild, A., Robert, C., Haanen, J.B., Ascierto, P., Larkin, J., . . . McArthur, G.A. (2011). Improved survival with vemurafenib in melanoma with BRAF V600E mutation. New England Journal of Medicine, 364, 2507–2516. doi:10.1056/NEJMoa1103782
Choudhry, N.K., Krumme, A.A., Ercole, P.M., Girdish, C., Tong, A.Y., Khan, N.F., . . . Franklin, J.M. (2017). Effect of reminder devices on medication adherence: The REMIND randomized clinical trial. JAMA Internal Medicine, 177, 624–631. doi:10.1001/jamainternmed.2016.9627
Clay, A.M., & Parsh, B. (2016). Patient- and family-centered care: It’s not just for pediatrics anymore. AMA Journal of Ethics, 18, 40–44. doi:10.1001/journalofethics.2016.18.1.medu3-1601
Flaherty, K.T., Robert, C., Hersey, P., Nathan, P., Garbe, C., Milhem, M., . . . Schadendorf, D. (2012). Improved survival with MEK inhibition in BRAF-mutated melanoma. New England Journal of Medicine, 367, 107–114. doi:10.1056/NEJMoa1203421
Genentech. (2016a). Cotellic® (cobimetinib) [Package insert]. Retrieved from http://bit.ly/2sM02ij
Genentech. (2016b). Zelboraf® (vemurafenib) [Package insert]. Retrieved from http://bit.ly/2t0C2fh
Komatsu, H., & Yagasaki, K. (2014). The power of nursing: Guiding patients through a journey of uncertainty. European Journal of Oncology Nursing, 18, 419–424. doi:10.1016/j.ejon.2014.03.006
Larkin, J., Ascierto, P.A., Dréno, B., Atkinson, V., Liszkay, G., Maio, M., . . . Ribas, A. (2014). Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. New England Journal of Medicine, 371, 1867–1876. doi:10.1056/NEJMoa1408868
Livingstone, E., Zimmer, L., Vaubel, J., & Schadendorf, D. (2014). BRAF, MEK and KIT inhibitors for melanoma: Adverse events and their management. Chinese Clinical Oncology, 3(3), 29. doi:10.3978/j.issn.2304-3865.2014.03.03
McArthur, G.A., Chapman, P.B., Robert, C., Larkin, J., Haanen, J.B., Dummer, R., . . . Hauschild, A. (2014). Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): Extended follow-up of a phase 3, randomised, open-label study. Lancet Oncology, 15, 323–332. doi:10.1016/s1470-2045(14)70012-9
Miller, W.R, & Rollnick, S. (Eds.). (2013). Conversations about change. In Motivational interviewing: Helping people change (3rd ed., pp. 3–13). New York, NY: Guilford Publications.
Novartis. (2015). Mekinist® (trametinib) [Package insert]. Retrieved from https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/fil…
Novartis. (2016). Tafinlar® (dabrafenib) [Package insert]. Retrieved from https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/fil…
Novartis. (2017, March 16). Storage, stability, and handling of Mekinist tablets. [Standard response letter to Valerie Guild, AIM at Melanoma Foundation].
OncoLink. (2015a). Dabrafenib (Tafinlar®). Retrieved from http://bit.ly/2ueD4Vo
OncoLink. (2015b). Vemurafenib (Zelboraf®). Retrieved from http://bit.ly/2t0PvUr
OncoLink. (2016a). Cobimetinib (Cotellic®). Retrieved from http://bit.ly/2tJERi9
OncoLink. (2016b). Trametinib (Mekinist®). Retrieved from http://bit.ly/2tEQ4SD
Partridge, A.H., Avorn, J., Wang, P.S., & Winer, E.P. (2002). Adherence to therapy with oral antineoplastic agents. Journal of the National Cancer Institute, 94, 652–661.
Robert, C., Karaszewska, B., Schachter, J., Rutkowski, P., Mackiewicz, A., Stroiakovski, D., . . . Schadendorf, D. (2015). Improved overall survival in melanoma with combined dabrafenib and trametinib. New England Journal of Medicine, 372, 30–39. doi:10.1056/NEJMoa1412690
Sabaté, E. (2003). Adherence to long-term therapies: Evidence for action. Geneva, Switzerland: World Health Organization. Retrieved from http://bit.ly/1dHSkqy
Schneider, S.M., Adams, D.B., & Gosselin, T. (2014). A tailored nurse coaching intervention for oral chemotherapy adherence. Journal of the Advanced Practitioner in Oncology, 5(3), 163–172.
Schneider, S.M., Hess, K., & Gosselin, T. (2011). Interventions to promote adherence with oral agents. Seminars in Oncology Nursing, 27, 133–141. doi:10.1016/j.soncn.2011.02.005
Shinnick, M.A., & Woo, M.A. (2015). Learning style impact on knowledge gains in human patient simulation. Nurse Education Today, 35, 63–67. doi:10.1016/j.nedt.2014.05.013
Spoelstra, S.L., Burhenn, P.S., DeKoekkoek, T., & Schueller, M. (2016). A trial examining an advanced practice nurse intervention to promote medication adherence and symptom management in adult cancer patients prescribed oral anti-cancer agents: Study protocol. Journal of Advanced Nursing, 72, 409–420. doi:10.1111/jan.12828
Spoelstra, S.L., Given, B.A., Given, C.W., Grant, M., Sikorskii, A., You, M., & Decker, V. (2013). An intervention to improve adherence and management of symptoms for patients prescribed oral chemotherapy agents: An exploratory study. Cancer Nursing, 36, 18–28. doi:10.1097/NCC.0b013e3182551587
Spoelstra, S.L., Given, C.W., Sikorskii, A., Majumder, A., Schueller, M., & Given, B.A. (2015). Treatment with oral anticancer agents: Symptom severity and attribution, and interference with comorbidity management. Oncology Nursing Forum, 42, 80–88. doi:10.1188/15.ONF.42-01p
Timmers, L., Boons, C.C., Kropff, F., van de Ven, P.M., Swart, E.L., Smit, E.F., . . . Hugtenburg, J.G. (2014). Adherence and patients’ experiences with the use of oral anticancer agents. Acta Oncologica, 53, 259–267. doi:10.3109/0284186x.2013.844353
Timmers, L., Boons, C.C., Verbrugghe, M., van den Bemt, B.J., Van Hecke, A., & Hugtenburg, J.G. (2017). Supporting adherence to oral anticancer agents: Clinical practice and clues to improve care provided by physicians, nurse practitioners, nurses and pharmacists. BMC Cancer, 17, 122. doi:10.1186/s12885-017-3110-2
van Zeijl, M.C., van den Eertwegh, A.J., Haanen, J.B., & Wouters, M.W. (2017). (Neo)adjuvant systemic therapy for melanoma. European Journal of Surgical Oncology, 43, 534–543. doi:10.1016/j.ejso.2016.07.001
Vanneste, L., Wolter, P., Van den Oord, J.J., Stas, M., & Garmyn, M. (2015). Cutaneous adverse effects of BRAF inhibitors in metastatic malignant melanoma, a prospective study in 20 patients. Journal of the European Academy of Dermatology and Venereology, 29, 61–68. doi:10.1111/jdv.12449
Weingart, S.N., Brown, E., Bach, P.B., Eng, K., Johnson, S.A., Kuzel, T.M., . . . Walters, R.S. (2008). NCCN Task Force Report: Oral chemotherapy. Journal of the National Comprehensive Cancer Network, 6(Suppl. 3), S1–S14.
Welsh, S.J., & Corrie, P.G. (2015). Management of BRAF and MEK inhibitor toxicities in patients with metastatic melanoma. Therapeutic Advances in Medical Oncology, 7, 122–136. doi:10.1177/1758834014566428
Winn, A.N., Keating, N.L., & Dusetzina, S.B. (2016). Factors associated with tyrosine kinase inhibitor initiation and adherence among medicare beneficiaries with chronic myeloid leukemia. Journal of Clinical Oncology, 34, 4323–4328. doi:10.1200/jco.2016.67.4184
Zafar, S.Y., Peppercorn, J.M., Schrag, D., Taylor, D.H., Goetzinger, A.M., Zhong, X., & Abernethy, A.P. (2013). The financial toxicity of cancer treatment: A pilot study assessing out-of-pocket expenses and the insured cancer patient’s experience. Oncologist, 18, 381–390. doi:10.1634/theoncologist.2012-0279




