PD-1 Inhibitor Therapy: Consensus Statement From the Faculty of the Melanoma Nursing Initiative on Managing Adverse Events
Background: Programmed cell death protein 1 (PD-1) inhibitor therapies are now a standard treatment for advanced melanoma and other tumor types. The immune-related adverse events (irAEs) associated with PD-1 inhibitor therapy are drastically different from the AEs associated with chemotherapy. Because these irAEs reflect immune system activation rather than side effects of therapy, nurses should be cognizant of the range of organ systems potentially affected as well as likely clinical presentations.
Objectives: This article presents consensus statements to guide nurses in the recognition and management of irAEs associated with PD-1 inhibitor monotherapy for advanced melanoma.
Methods: Members of the Melanoma Nursing Initiative discussed the current literature and clinical experience regarding nursing interventions related to irAEs associated with PD-1 inhibitor therapy.
Findings: The care step pathways provided for select irAEs represent a proactive, comprehensive nursing care plan to support optimal outcomes for patients receiving PD-1 inhibitor therapy.
Jump to a section
Since their recent introduction in oncology, programmed cell death protein 1 (PD-1) inhibitors have become a common treatment modality for unresectable or metastatic (advanced) melanoma as well as other malignancies, including non-small cell lung cancer, squamous cell carcinoma of the head and neck, Hodgkin lymphoma, renal cell carcinoma, and urothelial carcinoma (Franklin, Livingstone, Roesch, Schilling, & Schadendorf, 2017; Redman, Gibney, & Atkins, 2016). Two PD-1 inhibitor therapies have received approval from the U.S. Food and Drug Administration (FDA) as monotherapies for the treatment of patients with advanced melanoma: pembrolizumab (Keytruda®) and nivolumab (Opdivo®) (Bristol-Myers Squibb, 2017; Franklin et al., 2017; Merck, 2017; Redman et al., 2016). In phase 3 trials conducted in patients with melanoma, both agents significantly improved one-year survival and progression-free survival: nivolumab when compared with dacarbazine and pembrolizumab when compared with ipilimumab (Robert, Long, et al., 2015; Robert, Schachter, et al., 2015). Nivolumab was subsequently approved in the United States in combination therapy with ipilimumab for use in advanced melanoma. Because PD-1 inhibitor therapies were first approved in the melanoma setting, nurses with melanoma expertise have developed a wealth of experience in the identification and management of the unique toxicity profile of these agents. Two PD-L1 inhibitors have been approved by the FDA for the treatment of other cancers: atezolizumab (Tecentriq®) and avelumab (Bavencio®). These will not be discussed in this consensus statement because they are not currently in use for metastatic melanoma (Genentech, 2017; Pfizer, 2017).
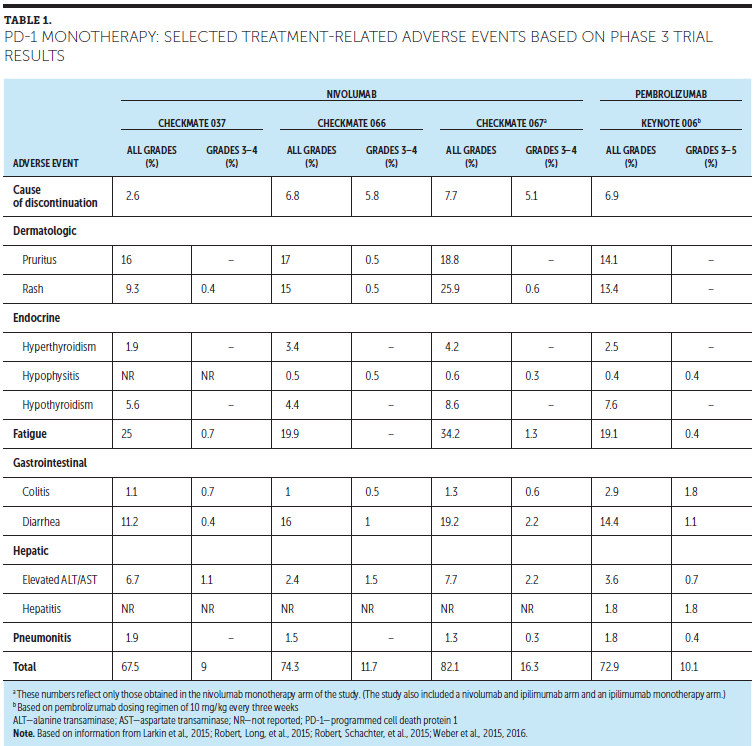
An examination of the mechanism of action of PD-1 inhibitors provides insight into the efficacy and immune-related adverse events (irAEs) associated with this immune checkpoint inhibitor (ICI) class. Several irAEs for PD-1 inhibitor therapies are shown in Table 1. Checkpoints are one mechanism by which the immune system regulates itself to prevent too robust of an immune response, which could lead to tissue destruction and autoimmunity, among other consequences (Buchbinder & Desai, 2016; Shimanovsky, Jethava, & Dasanu, 2013; Tentori, Lacal, & Graziani, 2013). Simply put, they are major components of a complex system that functions as a provider of checks and balances to control immune response and prevent autoimmunity. One checkpoint, cytotoxic T lymphocyte-associated antigen 4 (CTLA4), regulates T-cell proliferation early in an immune response (priming phase), primarily in lymph nodes, whereas PD-1 suppresses T cells later in an immune response (effector phase), primarily in peripheral tissue (Buchbinder & Desai, 2016; Rubin, 2012). Activated T cells perform immune surveillance. They are tasked with recognizing foreign cells, as well as host cells that are functioning improperly, such as cancer cells. PD-1 is a protein that is expressed on activated T cells. When PD-1 binds with its ligands (PD-L1 and PD-L2), T-cell function is inhibited or turned off. These ligands can be expressed by other cells in the immune system, including dendritic cells, or by the tumor itself; this is one mechanism by which cancer cells are able to avoid destruction by a patient’s immune system (Buchbinder & Desai, 2016; Rubin, 2012). PD-1 inhibitor therapies bind to PD-1 receptors on T cells, preventing the binding to PD-L1 and, thereby, unleashing the immune system.
Understanding the mechanism of action of ICIs lays the foundation for understanding the associated toxicity profiles. An uninhibited T cell will remain fully and persistently active. CTLA4 acts earlier in T-cell activation, whereas PD-1 acts later. This important distinction underscores the differences in toxicity between CTLA4 inhibitor therapy and PD-1 inhibitor therapy (i.e., PD-1 inhibitor therapy tends to produce fewer and less severe irAEs than ipilimumab). Figure 1 shows helpful analogies for explaining the relationship between the mechanism of action and efficacy and safety profiles of ICIs to patients.

Nurses must understand that the irAEs associated with PD-1 inhibitor therapies result from a dramatically different mechanism than AEs stemming from cytotoxic chemotherapy. Although the presenting symptoms may be identical, the difference in the mechanism of development necessitates different strategies to reverse or otherwise minimize their negative impact. These strategies require that nurses be aware of how a manipulated, overly active immune system can affect various organ systems, sometimes in subtle or unanticipated ways. Nurses who anticipate these varied effects and know what to do when they occur will have a critical role in the early recognition and management of these irAEs.
Faculty of the Melanoma Nursing Initiative (MNI) convened to define management challenges associated with the use of PD-1 inhibitor therapies in advanced melanoma. Based on a review of the current literature (Boutros et al., 2016; Dadu, Zobniw, & Diab, 2016; Day & Hansen, 2016; Friedman, Proverbs-Singh, & Postow, 2016; Kähler et al., 2016; Spain, Diem, & Larkin, 2016; Villadolid & Amin, 2015; Weber, Postow, Lao, & Schadendorf, 2016) and clinical experience, they developed consensus recommendations for nursing care of this population. Recommendations are made for the following:
• Administration and dosing, with a focus on dosage holds and schedule adjustments
• irAE recognition and management, with care step pathways (CSPs) developed for high-priority irAEs associated with ICIs for which nursing assessment and care are essential to producing successful outcomes
The text of this article focuses on irAE recognition and management principles specific to PD-1 inhibitor monotherapy.
Testing and Patient Selection
PD-1 inhibitor therapies are used in a range of tumor types. Given the mechanism of action of these agents, selection of patients based on tumor PD-L1 expression would be a reasonable consideration. However, PD-L1 expression status in melanoma remains investigational, given the lack of reliable assays (National Comprehensive Cancer Network, 2016). PD-L1 status or elevated expression is not a prerequisite for PD-1 inhibitor treatment of advanced melanoma, as it is in lung cancer (Bristol-Myers Squibb, 2017; Merck, 2017).
Another aspect of patient selection with PD-1 inhibitor therapy relates to the use of single-agent nivolumab or pembrolizumab versus combination therapy using anti-CTLA4 and PD-1 inhibitors (ipilimumab and nivolumab). Grade 3 or 4 toxicities are typically more common with combination therapy than with either nivolumab or pembrolizumab monotherapies, and should be considered when recommending treatment. Nurses play fundamental roles in assessing patient knowledge, understanding treatment options, and providing support as patients consider these options. Such support includes assessing each patient’s fitness for different therapeutic options.
Dosing and Administration
Pembrolizumab is given as an IV infusion every three weeks; the dosage has been modified to a fixed dosage of 200 mg administered over 30 minutes versus a weight-based dosing regimen (Merck, 2017). Nivolumab is given as an IV infusion every two weeks; the dosage has been modified to a fixed dose of 240 mg administered over 60 minutes rather than a weight-based dose (Bristol-Myers Squibb, 2017). Specific holds and discontinuation instructions for irAEs are discussed in the CSPs. No definitive standardized treatment duration exists for PD-1 inhibitor therapy. Although the labeling indicates continuation of PD-1 inhibitor monotherapy to disease progression or unacceptable toxicity, institutions (and even different clinicians within a given institution) often vary in how long therapy will continue if a patient achieves a state of no evidence of disease and continues to tolerate treatment well.
Management
Each CSP for the management of notable irAEs associated with PD-1 inhibitor therapy incorporates essential components of the nursing assessment specific to that AE. Look, listen, and recognize categories within the nursing assessment section direct the nurse to a specific set of symptom-related queries to ask the patient and/or caregiver as well as highlight additional information to be considered and reviewed as part of the nursing assessment. Data obtained from this assessment will guide appropriate management strategies. Wherever possible, grading for the specific AE is provided within the pathway based on the National Cancer Institute’s (2010) Common Terminology Criteria for Adverse Events (CTCAE). However, for the CSPs for thyroiditis and type 1 diabetes mellitus (DM), the CTCAE guidance was not used. Instead, Melanoma Nursing Initiative members agreed that, for these irAEs, diagnosis and management were better informed by laboratory value criteria from an individual’s institutional norms (such as thyroid stimulating hormone, free T3, and T4 for thyroiditis and serum glucose levels for DM).
Each CSP describes overall management strategies as well as nursing-specific interventions. Where applicable, strategies specific to each AE grade are listed, including dose holds and discontinuations. Patient counseling, along with recommendations for additional care and referral to specialty or ancillary care providers, are included in the management section, as appropriate.
General Education
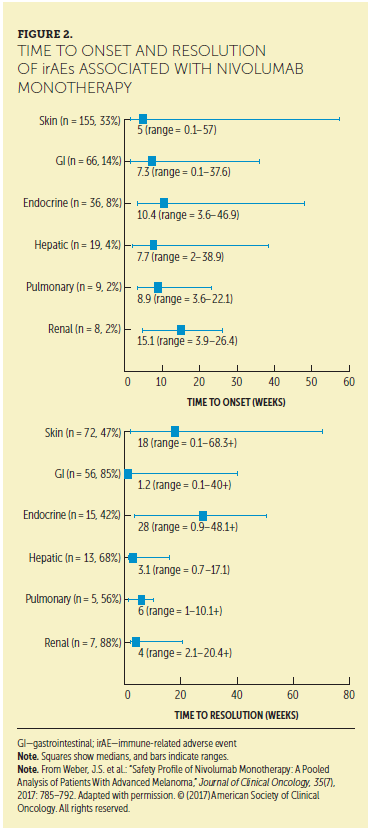
Patients should be educated as a first step in early recognition and management of irAEs. A key point to emphasize is that any change from baseline health, no matter how subtle or seemingly insignificant, may be a sign of an irAE and should be reported immediately to the oncology healthcare provider (HCP). This is an important communication point for patients who may be hesitant to report a change for fear that treatment may be discontinued. Such patients may feel the need to persist with treatment, which may result in more severe toxicity. Forming a strong partnership with patients and their caregivers, encouraging open and honest communication, and reassuring patients that reporting symptoms will increase the likelihood of staying on therapy are strategies that should ensure early reporting of symptoms. In addition, patients and their family members must be counseled that irAEs may occur at any time after initiating treatment, even months later (see Figure 2) (Champiat et al., 2016). Within this framework, the importance and rationale for obtaining baseline and follow-up testing to aid in irAE detection and diagnosis should be emphasized (Champiat et al., 2016). Counseling women with childbearing potential to avoid pregnancy is important because of the potential embryo-fetal toxicity (Friedman et al., 2016).
Oncology nurses can help to ensure that patients are aware of educational and financial resources for their therapies and how to access such resources (see Figure 3). Particularly useful are drug-specific patient wallet cards that identify signs and symptoms that should be immediately reported to the treating oncologist or other members of the healthcare team. Patients should be encouraged to carry the card with them at all times. One strategy discussed by the MNI was a recommendation to patients topaperclip their insurance cards to their wallet card. Consequently, whenever they need to show their insurance card, they can share information about their immunotherapy regimen. Wallet cards not only serve as ready guides for notable signs and symptoms potentially reflective of an irAE, but also provide relevant information to other HCPs (such as emergency department personnel) to improve patient care (Chae, Chae, Isaacson, & James, 2009; Institute for Healthcare Improvement, 2011).
[[{"type":"media","view_mode":"media_original","fid":"34306","field_deltas":{"1":{}},"link_text":null,"fields":{},"attributes":{"height":"718","width":"750","class":"media-image media-element file-media-original","data-delta":"1"}}]]
Overall Approach
Some general principles inform management strategies across all the CSPs. Many of the irAEs with PD-1 therapies are either grade 1 (mild; asymptomatic or mildly symptomatic) or grade 2 (moderate; moderately symptomatic, with some impact on activities of daily living). For grade 2 irAEs, ICIs are generally withheld and not resumed until toxicity is reduced to grade 1 or less. Corticosteroids are usually initiated if symptoms do not resolve within a week. For some grade 3 (severely symptomatic or having a large impact on activities of daily living) or grade 4 (life-threatening) irAEs, the ICI is often permanently discontinued, and high doses of corticosteroids are given. These high dosages are often split into two daily doses. Corticosteroids are slowly tapered (over at least a month) to avoid rebound symptoms. Methylprednisolone dosepaks are used by some providers, but are often not sufficient to reduce the inflammatory reactions resulting from the ICIs. Gastric prophylaxis with a proton pump inhibitor or H2 blocker should be considered when initiating high-dose corticosteroids. Patients should be educated to take their corticosteroid dose with food. In addition, antimicrobial prophylaxis is a consideration for patients prescribed high-dose corticosteroids, particularly those patients requiring extended therapy (more than 20 mg per day for more than one month) (Limper et al., 2011).
Dermatologic Toxicities
Appendix A provides a detailed CSP for cutaneous toxicities. Maculopapular rash and pruritus are the most common irAEs associated with PD-1 inhibitor therapy and are typically the first to develop (at 9 weeks to 2.8 months in trials) (Dadu et al., 2016; Friedman et al., 2016). These toxicities can have a detrimental impact on quality of life. Pruritus can be particularly distressing and distracting to patients. The nurse plays a crucial role in counseling and in providing individualized interventions, particularly in high-risk patients, as well as in assessing skin integrity and symptom distress. Mild to moderate cases are typically well managed with skin care, oral antihistamines, and, if necessary, topical or oral corticosteroids. Severe rash cases require dermatology consultation, hospitalization, treatment with systemic corticosteroids, and discontinuation of PD-1 inhibitor therapy.
[[{"type":"media","view_mode":"media_original","fid":"34311","field_deltas":{"2":{}},"link_text":null,"fields":{},"attributes":{"height":"940","width":"752","class":"media-image media-element file-media-original","data-delta":"2"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34316","field_deltas":{"3":{}},"link_text":null,"fields":{},"attributes":{"height":"938","width":"764","class":"media-image media-element file-media-original","data-delta":"3"}}]]
Gastrointestinal Toxicities
Appendixes B and C provide detailed CSPs for gastrointestinal toxicities. Diarrhea and colitis are the second most frequently encountered irAEs observed with PD-1 inhibitor therapy (Larkin et al., 2015; Robert, Long, et al., 2015; Robert, Schachter, et al., 2015; Weber et al., 2015). The time of onset for diarrhea and colitis in clinical trials was 3.4 months for pembrolizumab and 5.6 months for nivolumab (Dadu et al., 2016). Nurses play an essential role in the early recognition and management of diarrhea. Key to management is establishing a patient’s baseline bowel pattern, including frequency and consistency of stools. Determining severity of symptoms is based on change from patient’s baseline. For example, CTCAE grading of diarrhea is determined based on the increase of bowel movements above baseline, not the absolute number of stools. Therefore, depending on the baseline stool output, one patient with five stools a day may have mild diarrhea, whereas another with the same number of stools per day could have moderate diarrhea. Colitis can occur in the absence of diarrhea and may lead to serious sequelae, including death, if not identified promptly and managed appropriately.
[[{"type":"media","view_mode":"media_original","fid":"34321","field_deltas":{"4":{}},"link_text":null,"fields":{},"attributes":{"height":"771","width":"751","class":"media-image media-element file-media-original","data-delta":"4"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34326","field_deltas":{"5":{}},"link_text":null,"fields":{},"attributes":{"height":"936","width":"753","class":"media-image media-element file-media-original","data-delta":"5"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34331","field_deltas":{"6":{}},"link_text":null,"fields":{},"attributes":{"height":"640","width":"750","class":"media-image media-element file-media-original","data-delta":"6"}}]]
Upper gastrointestinal–related irAEs, such as mucositis or xerostomia, have also been noted with these regimens. Although not common or medically significant, they can have a major negative impact on quality of life. Little to no data support best practices or interventions for oral irAEs. Interventions and management approaches were developed based on the clinical experience of the members of the MNI. Esophagitis and gastritis have also been reported with PD-1 inhibitor therapies.


Hepatotoxicity (Hepatitis)
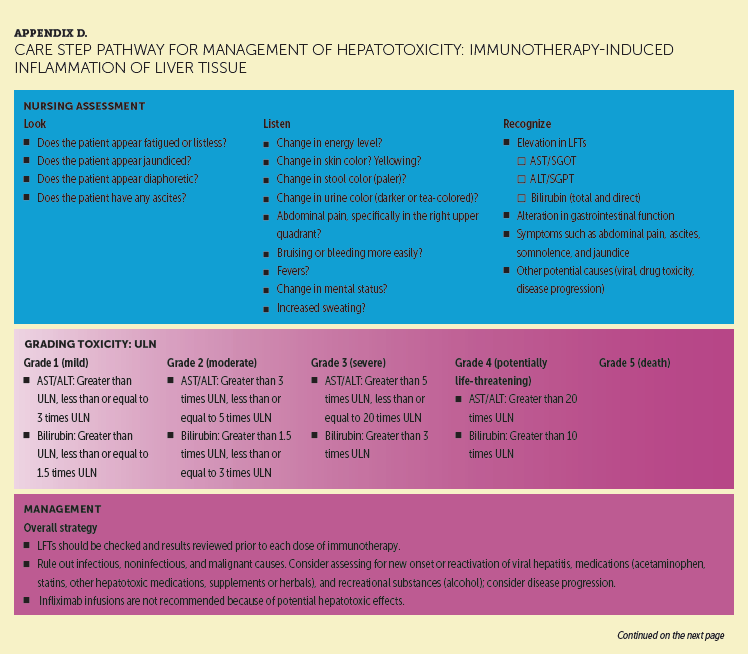
Appendix D provides a CSP for hepatotoxicity. PD-1 inhibitor therapies may cause inflammation of liver tissue (autoimmune hepatitis) that commonly presents as an asymptomatic elevation in alanine transaminase (ALT) and/or aspartate transaminase (AST) and, less frequently, elevated bilirubin (Dadu et al., 2016; Friedman et al., 2016). Although ALT and AST elevations occur less often with PD-1 inhibitor monotherapy than do skin and gastrointestinal toxicities (Larkin et al., 2015; Robert, Long, et al., 2015; Robert, Schachter, et al., 2015; Weber et al., 2015, 2016), hepatic irAEs can be severe and even life-threatening if not recognized early and managed effectively (Dadu et al., 2016). Onset of hepatic irAEs has been reported at 1.2 months and at 3.7 months with pembrolizumab and nivolumab monotherapy, respectively (Dadu et al., 2016). Because liver function tests (LFTs) play an essential role in the detection and monitoring of changes in ALT and AST levels during treatments, oncology nurses should ensure that LFTs are obtained prior to each infusion and as directed (Dadu et al., 2016; Friedman et al., 2016). If LFTs are elevated, other possible causes of elevations (e.g., drug-induced toxicities, tumor progression, infectious causes) should be investigated (Dadu et al., 2016; Friedman et al., 2016; Weber et al., 2016).
[[{"type":"media","view_mode":"media_original","fid":"34336","field_deltas":{"7":{}},"link_text":null,"fields":{},"attributes":{"height":"827","width":"750","class":"media-image media-element file-media-original","data-delta":"7"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34341","field_deltas":{"8":{}},"link_text":null,"fields":{},"attributes":{"height":"545","width":"750","class":"media-image media-element file-media-original","data-delta":"8"}}]]
Endocrinopathies
Various endocrinopathies have been associated with PD-1 inhibitor therapy, including hypophysitis (inflammation of the pituitary gland), thyroid dysfunction (including hypothyroidism, hyperthyroidism, and, rarely, acute thyroiditis), adrenal insufficiency, and type 1 DM (Dadu et al., 2016; Friedman et al., 2016; Weber et al., 2016). Diagnosing endocrinopathies can be difficult because they often present with fatigue or other nonspecific symptoms. Because endocrinopathy is outside of the typical side-effect profile of chemotherapies, nurses should be particularly diligent in monitoring for these toxicities when assessing patients receiving immunotherapy. Using the look, listen, and recognize categories of the CSPs for thyroiditis and hypophysitis will assist oncology nurses in homing in on specific questions to ask at each patient visit and during triage calls, as necessary. Unique from other irAEs, endocrinopathies do not usually resolve because the function of the gland rarely recovers. In those cases, appropriate hormone replacement will be lifelong, but the irAE does not typically require treatment discontinuation. Oncology nurses should educate patients about the possibility of the requirement for lifelong hormone replacement before starting therapy, as well as reassure patients about the ability to live high-quality lives with replacement therapy. In general, management of endocrinopathies related to PD-1 inhibitors warrants endocrinology consultation for initial diagnosis and ongoing treatment (Dadu et al., 2016; Friedman et al., 2016; Weber et al., 2016).
[[{"type":"media","view_mode":"media_original","fid":"34346","field_deltas":{"9":{}},"link_text":null,"fields":{},"attributes":{"height":"700","width":"744","class":"media-image media-element file-media-original","data-delta":"9"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34351","field_deltas":{"10":{}},"link_text":null,"fields":{},"attributes":{"height":"639","width":"752","class":"media-image media-element file-media-original","data-delta":"10"}}]]
Endocrinopathies associated with PD-1 inhibitor therapy are relatively common, with as many as 20% of patients developing an endocrine-related irAE in clinical trials. Hypothyroidism is the most prevalent irAE, followed by hyperthyroidism, then thyroiditis. In most instances, severity is rarely higher than grade 2. Thyroid disorders are more frequent in women, which is akin to the higher incidence observed in the general population (González-Rodríguez & Rodríguez-Abreu, 2016). Inflammation of the thyroid gland (thyroiditis) may also produce hypothyroidism or, less commonly, hyperthyroidism. Hyperthyroidism commonly precedes hypogonadism (Dadu et al., 2016; Weber et al., 2016). The most severe endocrinopathy related to PD-1 inhibitors is acute adrenal insufficiency, a life-threatening toxicity that often requires hospitalization for monitoring and the administration of stress-dose corticosteroids. PD-1 inhibitor therapy should be held and may have to be discontinued, depending on the severity of symptoms (Friedman et al., 2016; Weber et al., 2016).
[[{"type":"media","view_mode":"media_original","fid":"34356","field_deltas":{"11":{}},"link_text":null,"fields":{},"attributes":{"height":"484","width":"749","class":"media-image media-element file-media-original","data-delta":"11"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34361","field_deltas":{"12":{}},"link_text":null,"fields":{},"attributes":{"height":"861","width":"752","class":"media-image media-element file-media-original","data-delta":"12"}}]]
Appendixes E and F provide detailed CSPs for PD-1 inhibitor–emergent hypophysitis and thyroiditis, respectively. High-dose systemic corticosteroid therapy is sometimes used during the acute phase of hypophysitis to reverse the inflammatory process, restore function, and prevent the need for permanent hormone replacement (Dadu et al., 2016; Friedman et al., 2016). However, most patients with hypophysitis and hypothyroidism will require long-term supplementation of affected hormones. Rare but notable cases of type 1 DM have been reported (Chae et al., 2017; Friedman et al., 2016). This irAE is characterized by an absolute insulin deficiency caused by autoimmune destruction of pancreatic beta cells, implying dependence on insulin (González-Rodríguez & Rodríguez-Abreu, 2016). The CSP for type 1 DM is shown in Appendix G.

Pneumonitis
Appendix H provides a detailed CSP for pneumonitis, which is a rare but serious and potentially life-threatening irAE associated with PD-1 inhibitor therapy, typically emerging several months after treatment initiation (although it can occur at any time) (Dadu et al., 2016; Friedman et al., 2016; Weber et al., 2016). Signs and symptoms can be innocuous. Patients who have received prior radiation therapy to the lung and/or have large or multiple lung metastases may be at greater risk. Clinical presentations can vary, making accurate diagnosis difficult. Patients may be asymptomatic, with lung infiltrates seen on routine restaging scans. Imaging findings can also be variable, and no characteristic radiographic findings of ICI-related pneumonitis exist (Teply & Lipson, 2014). Symptomatic patients may report new or worsening respiratory symptoms, including dyspnea, shortness of breath, cough, pleuritic chest pain, and hypoxia. Lack of prompt recognition and proper treatment may result in increased morbidity or possibly mortality. Nurses play a significant role in the early identification of pneumonitis by checking patients’ pulse oxygen saturation at rest and on exertion at every visit, as well as assessing for a decrease, possibly indicating an early finding of pneumonitis.


Performing directed and focused assessments to pick up seemingly insignificant patient reports may also uncover additional symptoms suggestive of early toxicity. For example, during a prior visit, a patient may have reported that he or she could climb a set of stairs without difficulty. However, during the current visit, he or she may have reported becoming slightly “winded” with the same activity. A nurse with knowledge of the spectrum of toxicities with PD-1 inhibitor therapy would recognize these subtle signs, further evaluate the symptoms as potential red flags for pneumonitis, and intervene accordingly. These simple nursing interventions are paramount to early recognition of a possible irAE and may make the difference in a patient’s ability to continue on therapy.
Specialty care from a pulmonologist is often needed for management of immune-mediated pneumonitis. Moderate pneumonitis is managed with corticosteroids while withholding PD-1 inhibitor therapy. Severe pneumonitis is managed with prompt initiation of high-dose corticosteroids and PD-1 inhibitor discontinuation. Mycophenolate mofetil, cyclophosphamide, or infliximab may be indicated for corticosteroid-resistant symptoms. Because of the possibility of respiratory compromise, patients may require oxygen (grade 3) or urgent intubation and ventilatory support (grade 4).
Other Immune-Related Adverse Events
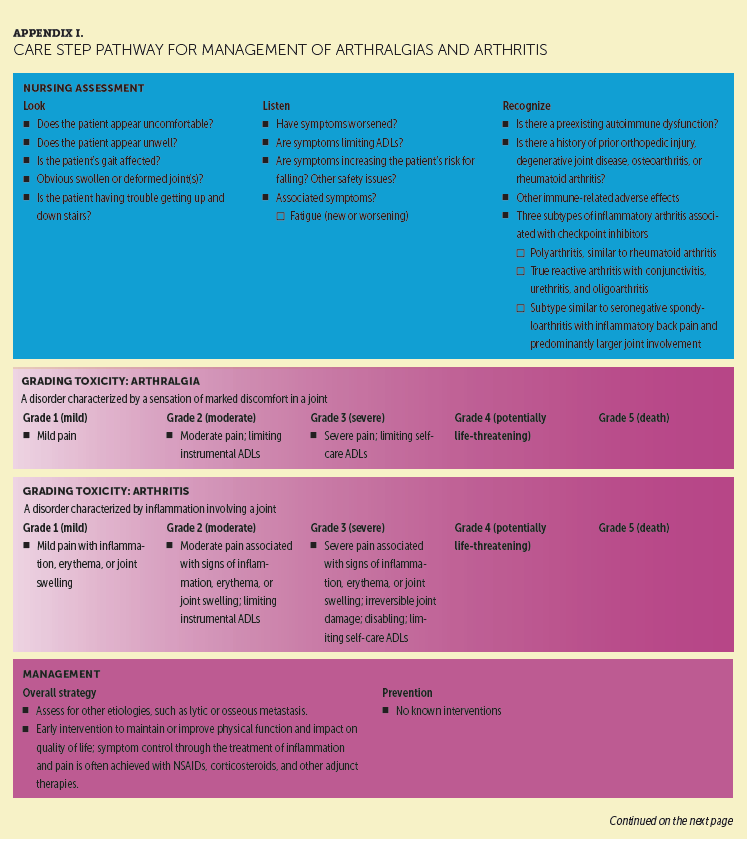
Appendix I presents a CSP for arthralgias and arthritis, less-common irAEs that often remain underrecognized or untreated, negatively affecting patient quality of life. The clinical presentations of these irAEs vary, and they typically require higher doses of corticosteroids for management than is used in the general population. Appendix J presents a CSP for neuropathy, sensory and motor. A wide variety of neurologic conditions can result from ICIs, including Guillain-Barré syndrome, myasthenia gravis, posterior reversible encephalopathy syndrome, and other forms of neuropathy. These conditions require neurology or neuromuscular consultation and, ideally, comanagement. Appendix K presents a CSP for nephritis. Nephritis is another rare complication associated with PD-1 inhibitor therapy; however, if unrecognized or misdiagnosed, it can lead to serious sequelae. Early recognition and aggressive management, ideally provided collaboratively with a nephrologist, is the best approach.
[[{"type":"media","view_mode":"media_original","fid":"34366","field_deltas":{"13":{}},"link_text":null,"fields":{},"attributes":{"height":"920","width":"752","class":"media-image media-element file-media-original","data-delta":"13"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34371","field_deltas":{"14":{}},"link_text":null,"fields":{},"attributes":{"height":"347","width":"747","class":"media-image media-element file-media-original","data-delta":"14"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34376","field_deltas":{"15":{}},"link_text":null,"fields":{},"attributes":{"height":"865","width":"755","class":"media-image media-element file-media-original","data-delta":"15"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34381","field_deltas":{"16":{}},"link_text":null,"fields":{},"attributes":{"height":"428","width":"750","class":"media-image media-element file-media-original","data-delta":"16"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34386","field_deltas":{"17":{}},"link_text":null,"fields":{},"attributes":{"height":"936","width":"754","class":"media-image media-element file-media-original","data-delta":"17"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34391","field_deltas":{"18":{}},"link_text":null,"fields":{},"attributes":{"height":"934","width":"760","class":"media-image media-element file-media-original","data-delta":"18"}}]]
PD-1 inhibitor therapy has been associated with several other AEs. Table 2 lists these, along with recommended management strategies. With the exception of the infusion reaction, these AEs generally reflect the widespread impact of the immune system on various body systems, although some may have a different etiology. For example, low-grade fatigue and other constitutional symptoms are the most common of all AEs associated with PD-1 inhibitor therapy, but whether these are irAEs is unclear. In some cases, they may be caused by nonspecific cytokine release or undiagnosed endocrinopathies related to PD-1 inhibitor therapy use (Day & Hansen, 2016).
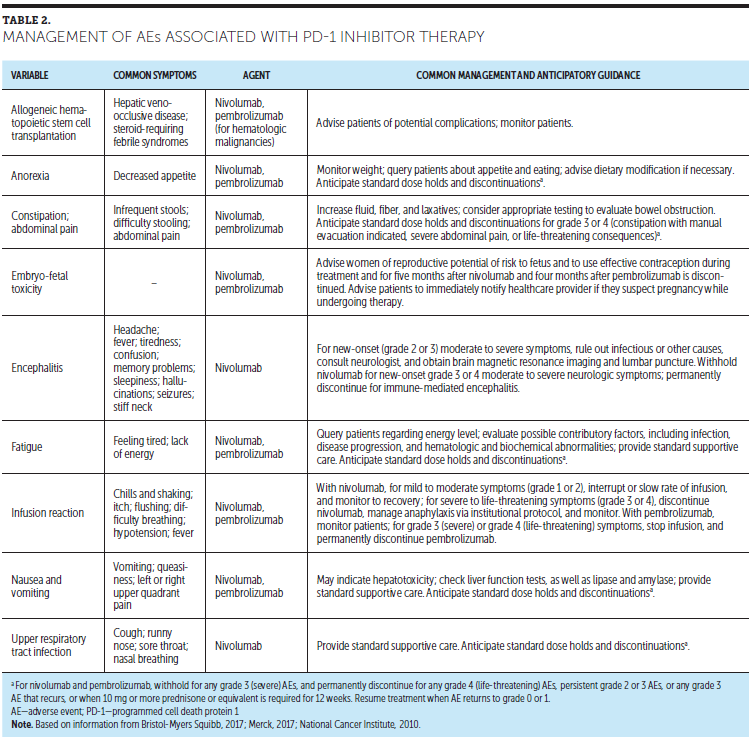
Implications for Nursing
The incidence of irAEs is generally lower with PD-1 inhibitor therapy than with ipilimumab, with no notable differences between nivolumab and pembrolizumab (Day & Hansen, 2016; Friedman et al., 2016; Weber et al., 2016). Also, the onset of grade 3 or 4 irAEs is typically later in the treatment course with PD-1 inhibitor therapy versus ipilimumab-based therapy. For oncology nurses familiar with ipilimumab, some irAEs do tend to occur more frequently with PD-1 antibodies than with ipilimumab, including thyroid dysfunction, some dermatologic toxicities (rash, vitiligo), arthralgias, myalgias, and pneumonitis (Day & Hansen, 2016; Spain et al., 2016).

Conclusion
PD-1 inhibitor therapy has become a standard approach for the treatment of advanced melanoma. The unique mechanism of action affords improved outcomes in terms of higher response rates and more durable responses, but accompanying that is the vast array of immune toxicities, including dermatitis, colitis, hepatotoxicity, hypophysitis, thyroiditis, diabetes, pneumonitis, arthralgia, nephritis, and neuropathy. Oncology nurses familiar with irAEs are ideally positioned to optimize care via patient education about the importance of early detection and reporting of new or worsening signs or symptoms, regular and ongoing patient assessments, and prompt intervention when toxicity is identified. Nurses can also help patients access additional sources of support and financial assistance. By becoming familiar with the CSPs highlighted in this article, nurses will be better prepared to provide the comprehensive nursing care so critical to improving irAE management and outcomes with PD-1 inhibitor therapy.
The authors gratefully acknowledge Jill Maria Weberding, MPH, BSN, RN, OCN®, for reviewing the manuscript from the community oncology nursing perspective.
About the Author(s)
Suzanne McGettigan, MSN, CRNP, ANP-BC, AOCN®, is a clinical nurse practitioner in the Division of Hematology-Oncology at the Abramson Cancer Center at the University of Pennsylvania in Philadelphia; and Krista M. Rubin, RN, MS, FNP-BC, is an advanced nurse practitioner in the Center for Melanoma at the Massachusetts General Hospital Cancer Center in Boston. The authors take full responsibility for this content. This supplement was funded by the AIM at Melanoma Foundation, with support via unrestricted grants from Amgen, Array Biopharma, Bristol-Myers Squibb, Incyte Corporation, Merck and Co., and Novartis Pharmaceuticals. Writing and editorial support was provided by Michael L. Coco, PhD, of Coco Communications, Inc., Lisa A. Tushla, PhD, H(ASCP), of Terranova Medica, and Marjorie Joyce, BA. McGettigan serves on speakers bureaus for Bristol-Myers Squibb, Genentech, Merck and Co., and Novartis Pharmaceuticals. Rubin has previously consulted for Merck and Co., and has received support from EMD Serono, Merck and Co., and Novartis Pharmaceuticals. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. McGettigan can be reached at suzanne.mcgettigan@uphs.upenn.edu, with copy to CJONEditor@ons.org. (Submitted April 2017. Accepted June 2, 2017.)
References
Boutros, C., Tarhini, A., Routier, E., Lambotte, O., Ladurie, F.L., Carbonnel, F., . . . Robert, C. (2016). Safety profiles of anti-CTLA-4 and anti-PD-1 antibodies alone and in combination. Nature Reviews Clinical Oncology, 13, 473–486. doi:10.1038/nrclinonc.2016.58
Bristol-Myers Squibb. (2017). Opdivo® (nivolumab) [Package insert]. Retrieved from http://www.opdivoyervoyhcp.com
Buchbinder, E.I., & Desai, A. (2016). CTLA-4 and PD-1 pathways: Similarities, differences, and implications of their inhibition. American Journal of Clinical Oncology, 39, 98–106. doi:10.1097/COC.0000000000000239
Chae, S.Y., Chae, M.H., Isaacson, N., & James, T.S. (2009). The patient medication list: Can we get patients more involved in their medical care? Journal of the American Board of Family Medicine, 22, 677–685. doi:10.3122/jabfm.2009.06.090059
Chae, Y.K., Chiec, L., Mohindra, N., Gentzler, R., Patel, J., & Giles, F. (2017). A case of pembrolizumab-induced type-1 diabetes mellitus and discussion of immune checkpoint inhibitor-induced type 1 diabetes. Cancer Immunology, Immunotherapy, 66, 25–32. doi:10.1007/s00262-016-1913-7
Champiat, S., Lambotte, O., Barreau, E., Belkhir, R., Berdelou, A., Carbonnel, F., . . . Marabelle, A. (2016). Management of immune checkpoint blockade dysimmune toxicities: A collaborative position paper. Annals of Oncology, 27, 559–574. doi:10.1093/annonc/mdv623
Dadu, R., Zobniw, C., & Diab, A. (2016). Managing adverse events with immune checkpoint agents. Cancer Journal, 22, 121–129. doi:10.1097/PPO.0000000000000186
Day, D., & Hansen, A.R. (2016). Immune-related adverse events associated with immune checkpoint inhibitors. BioDrugs, 30, 571–584. doi:10.1007/s40259-016-0204-3
Franklin, C., Livingstone, E., Roesch, A., Schilling, B., & Schadendorf, D. (2017). Immunotherapy in melanoma: Recent advances and future directions. European Journal of Surgical Oncology, 43, 604–611. doi:10.1016/j.ejso.2016.07.145
Friedman, C.F., Proverbs-Singh, T.A., & Postow, M.A. (2016). Treatment of the immune-related adverse effects of immune checkpoint inhibitors: A review. JAMA Oncology, 2, 1346–1353. doi:10.1001/jamaoncol.2016.1051
Genentech. (2017). Tecentriq® (atezolizumab) [Package insert]. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761034s001lbl…
González-Rodríguez, E., & Rodríguez-Abreu, D. (2016). Immune checkpoint inhibitors: Review and management of endocrine adverse events. Oncologist, 21, 804–816. doi:10.1634/theoncologist.2015-0509
Institute for Healthcare Improvement. (2011). How-to guide: Prevent adverse drug events (Medication reconcilitation). Retrieved from http://www.ihi.org/resources/Pages/Tools/HowtoGuidePreventAdverseDrugEv…
Kähler, K.C., Hassel, J.C., Heinzerling, L., Loquai, C., Mössner, R., Ugurel, S., . . . Gutzmer, R. (2016). Management of side effects of immune checkpoint blockade by anti-CTLA-4 and anti-PD-1 antibodies in metastatic melanoma. Journal of the German Society of Dermatology, 14, 662–681. doi:10.1111/ddg.13047
Larkin, J., Chiarion-Sileni, V., Gonzalez, R., Grob, J.J., Cowey, C.L., Lao, C.D., . . . Wolchok, J.D. (2015). Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. New England Journal of Medicine, 373, 23–34. doi:10.1056/NEJMoa1504030
Limper, A.H., Knox, K.S., Sarosi, G.A., Ampel, N.M., Bennett, J.E., Catanzaro, A., . . . Stevens, D.A. (2011). An official American Thoracic Society statement: Treatment of fungal infections in adult pulmonary and critical care patients. American Journal of Respiratory and Critical Care Medicine, 183, 96–128. doi:10.1164/rccm.2008-740ST
Merck. (2017). Keytruda® (pembrolizumab) [Package insert]. Retrieved from http://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf
National Cancer Institute. (2010). Common Terminology Criteria for Adverse Events [v.4.03]. Retrieved from https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference…
National Comprehensive Cancer Network. (2016). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Melanoma [v.1.2017]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/melanoma.pdf
Pfizer. (2017). Bavencio® (avelumab) [Package insert]. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761078s000lbl…
Redman, J.M., Gibney, G.T., & Atkins, M.B. (2016). Advances in immunotherapy for melanoma. BMC Medicine, 14, 20. doi:10.1186/s12916-016-0571-0
Robert, C., Long, G.V., Brady, B., Dutriaux, C., Maio, M., Mortier, L., . . . Ascierto, P.A. (2015). Nivolumab in previously untreated melanoma without BRAF mutation. New England Journal of Medicine, 372, 320–330. doi:10.1056/NEJMoa1412082
Robert, C., Schachter, J., Long, G.V., Arance, A., Grob, J.J., Mortier, L., . . . Ribas, A. (2015). Pembrolizumab versus ipilimumab in advanced melanoma. New England Journal of Medicine, 372, 2521–2532. doi:10.1056/NEJMoa1503093
Rubin, K.M. (2012). Managing immune-related adverse events to ipilimumab: A nurse’s guide [Online exclusive]. Clinical Journal of Oncology Nursing, 16, E69–E75. doi:10.1188/12.CJON.E69-E75
Shimanovsky, A., Jethava, A., & Dasanu, C.A. (2013). Immune alterations in malignant melanoma and current immunotherapy concepts. Expert Opinion on Biological Therapy, 13, 1413–1427. doi:10.1517/14712598.2013.827658
Spain, L., Diem, S., & Larkin, J. (2016). Management of toxicities of immune checkpoint inhibitors. Cancer Treatment Reviews, 44, 51–60. doi:10.1016/j.ctrv.2016.02.001
Tentori, L., Lacal, P.M., & Graziani, G. (2013). Challenging resistance mechanisms to therapies for metastatic melanoma. Trends in Pharmacological Sciences, 34, 656–666. doi:10.1016/j.tips.2013.10.003
Teply, B.A., & Lipson, E.J. (2014). Identification and management of toxicities from immune checkpoint-blocking drugs. Oncology, 28(Suppl. 3), 30–38.
Villadolid, J., & Amin, A. (2015). Immune checkpoint inhibitors in clinical practice: Update on management of immune-related toxicities. Translational Lung Cancer Research, 4, 560–575. doi:10.3978/j.issn.2218-6751.2015.06.06
Weber, J.S., D’Angelo, S.P., Minor, D., Hodi, F.S., Gutzmer, R., Neyns, B., . . . Larkin, J. (2015). Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): A randomised, controlled, open-label, phase 3 trial. Lancet Oncology, 16, 375–384. doi:10.1016/S1470-2045(15)70076-8
Weber, J.S., Postow, M., Lao, C.D., & Schadendorf, D. (2016). Management of adverse events following treatment with anti-programmed death-1 agents. Oncologist, 21, 1230–1240. doi:10.1634/theoncologist.2016-0055
References for Appendixes A-K
Bristol-Myers Squibb. (2017a). Opdivo® (nivolumab) [Package insert]. Retrieved from http://www.opdivoyervoyhcp.com
Bristol-Myers Squibb. (2017b, February 21). Use of vaccines in patients receiving nivolumab [Standard response letter to Lisa Tushla, Terranova Medica, LLC].
Bristol-Myers Squibb. (2017c). Yervoy® (ipilimumab) [Package insert]. Retrieved from http://packageinserts.bms.com/pi/pi_yervoy.pdf
Byun, D.J., Wolchok, J.D., Rosenberg, L.M., & Girotra, M. (2017). Cancer immunotherapy—Immune checkpoint blockade and associated endocrinopathies. Nature Reviews Endocrinology, 13, 195–207. doi:10.1038/nrendo.2016.205
Cappelli, L.C., Naidoo, J., Bingham, C.O., III, & Shah, A.A. (2017). Inflammatory arthritis due to immune checkpoint inhibitors: Challenges in diagnosis and treatment. Immunotherapy, 9, 5–8. doi:10.2217/imt-2016-0117
Cappelli, L.C., Shah, A.A., & Bingham, C.O., III. (2017). Immune-related adverse effects of cancer immunotherapy—Implications for rheumatology. Rheumatic Diseases Clinics of North America, 43, 65–78. doi:10.1016/j.rdc.2016.09.007
Champiat, S., Lambotte, O., Barreau, E., Belkhir, R., Berdelou, A., Carbonnel, F., . . . Marabelle, A. (2016). Management of immune checkpoint blockade dysimmune toxicities: A collaborative position paper. Annals of Oncology, 27, 559–574. doi:10.1093/annonc/mdv623
Dadu, R., Zobniw, C., & Diab, A. (2016). Managing adverse events with immune checkpoint agents. Cancer Journal, 22, 121–129. doi:10.1097/PPO.0000000000000186
Durham, C.O., Fowler, T., Donato, A., Smith, W., & Jensen, E. (2015). Pain management in patients with rheumatoid arthritis. Nurse Practitioner, 40(5), 38–45. doi:10.1097/01.NPR.0000463784.36883.23
Fecher, L.A., Agarwala, S.S., Hodi, F.S., & Weber, J.S. (2013). Ipilimumab and its toxicities: A multidisciplinary approach. Oncologist, 18, 733–743. doi:10.1634/theoncologist.2012-0483
Friedman, C.F., Proverbs-Singh, T.A., & Postow, M.A. (2016). Treatment of the immune-related adverse effects of immune checkpoint inhibitors: A review. JAMA Oncology, 2, 1346–1353. doi:10.1001/jamaoncol.2016.1051
Kähler, K.C., Hassel, J.C., Heinzerling, L., Loquai, C., Mössner, R., Ugurel, S., . . . Gutzmer, R. (2016). Management of side effects of immune checkpoint blockade by anti-CTLA-4 and anti-PD-1 antibodies in metastatic melanoma. Journal of the German Society of Dermatology, 14, 662–681. doi:10.1111/ddg.13047
Kumar, V., Chaudhary, N., Garg, M., Floudas, C.S., Soni, P., & Chandra, A.B. (2017). Current diagnosis and management of immune related adverse events (irAEs) induced by immune checkpoint inhibitor therapy. Frontiers in Pharmacology, 8, 49. doi:10.3389/fphar.2017.00049
Lalla, R.V., Bowen, J., Barasch, A., Elting, L., Epstein, J., Keefe, D.M., . . . Elad, S. (2014). MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer, 120, 1453–1461. doi:10.1002/cncr.28592
Merck. (2017, February 17). Use with live attenuated vaccines [Standard response letter to Kathleen Marie Madden, NP].
Naidoo, J., Page, D.B., Li, B.T., Connell, L.C., Schindler, K., Lacouture, M.E., . . . Wolchok, J.D. (2015). Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Annals of Oncology, 26, 2375–2391. doi:10.1093/annonc/mdv383
National Cancer Institute. (2010). Common Terminology Criteria for Adverse Events [v.4.03]. Retrieved from https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference…
Rassy, E.E., Kourie, H.R., Rizkallah, J., El Karak, F., Hanna, C., Chelala, D.N., & Ghosn, M. (2016). Immune checkpoint inhibitors renal side effects and management. Immunotherapy, 8, 1417–1425. doi:10.2217/imt-2016-0099
Spain, L., Diem, S., & Larkin, J. (2016). Management of toxicities of immune checkpoint inhibitors. Cancer Treatment Reviews, 44, 51–60. doi:10.1016/j.ctrv.2016.02.001
U.S. Food and Drug Administration. (2012). Risk evaluation and mitigation strategy (REMS). Retrieved from https://www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafetyInfo…
Van Sebille, Y.Z., Stansborough, R., Wardill, H.R., Bateman, E., Gibson, R.J., & Keefe, D.M. (2015). Management of mucositis during chemotherapy: From pathophysiology to pragmatic therapeutics. Current Oncology Reports, 17, 50. doi:10.1007/s11912-015-0474-9
Weber, J.S., Postow, M., Lao, C.D., & Schadendorf, D. (2016). Management of adverse events following treatment with anti-programmed death-1 agents. Oncologist, 21, 1230–1240. doi:10.1634/theoncologist.2016-0055




