Intralesional Therapy: Consensus Statements for Best Practices in Administration From the Melanoma Nursing Initiative
Background: Talimogene laherparepvec (T-VEC) is the first intralesional therapy for melanoma approved by the U.S. Food and Drug Administration. This oncolytic viral immunotherapy has improved outcomes for patients with locoregional recurrent melanoma and is showing promise in combination with systemic therapies.
Objectives: This article aims to provide oncology nurses with expert guidance on best practices in incorporating intralesional therapy for patients diagnosed with melanoma in practice.
Methods: Members of the Melanoma Nursing Initiative explored issues related to administration of T-VEC in melanoma. The current literature and clinical experiences were reviewed.
Findings: The author offers a care step pathway (CSP) and commentary detailing best practices in infection control, drug storage, pharmacy interface, patient flow, space/staff allocation, patient education, and adverse event management with T-VEC. The CSP will help nurses improve patient outcomes and streamline the workflow with this novel therapeutic approach.
Jump to a section
Talimogene laherparepvec (T-VEC) (Imlygic®) received approval from the U.S. Food and Drug Administration (FDA) in 2015 for the local treatment of unresectable cutaneous, subcutaneous, and nodal lesions in patients with melanoma that recurs after initial surgery (Amgen, 2017b). This is the first intralesional treatment for melanoma and the first-in-class oncolytic viral immunotherapy approved by the FDA (Harrington et al., 2016).
Injecting an agent directly into the tumor is an attractive strategy for melanoma because cutaneous, subcutaneous, and nodal lesions are accessible for treatment (Agarwala, 2015). Interest in intralesional immunotherapy has been longstanding, but early attempts with intralesional Bacillus Calmette-Guérin (BCG) resulted in fatal cases of anaphylaxis and disseminated disease (de la Monte & Hutchins, 1986; Mastrangelo, Bellet, Berkelhammer, & Clark, 1975). Other intralesional approaches, such as plasmid interleukin-12 and PV-10 (rose bengal disodium 10%), have shown promise (Algazi et al., 2016; Foote et al., 2017; Thompson et al., 2015). Preliminary data suggest a synergistic effect of combining T-VEC with systemic immunotherapy in melanoma (Puzanov et al., 2016). T-VEC is also being studied for use with systemic therapy and in other cancer types, including nonmelanoma skin cancers, breast cancer, lymphoma, and sarcoma (see Table 1). Because the role of intralesional therapy in oncology is likely to expand, nurses can expect a greater demand for its clinical use.
Intralesional oncolytic viral immunotherapy is new to clinical practice and poses unique challenges for integration into the clinical workflow. To date, oncology nurses have played a central role in the setup and day-to-day administration of T-VEC. Members of the Melanoma Nursing Initiative (MNI) identified a lack of knowledge about intralesional therapy as one of several barriers to optimal melanoma care. The author developed the care step pathway (CSP) and the outline for this article based on a literature review, multiple online conferences with the MNI, and clinical experience at the first institution to offer T-VEC in Boston, Massachusetts. This article reviews the MNI consensus regarding best practices for addressing the nursing challenges in delivering intralesional therapy in melanoma.
Background
T-VEC is composed of a herpes simplex virus (HSV)-1 modified by the insertion of a gene encoding for the immunostimulatory cytokine human granulocyte-macrophage colony–stimulating factor (GM-CSF) and removal of two nonessential genes to reduce neurovirulence and tumor suppression (Orloff, 2016). In simple terms, T-VEC has been modified to replicate specifically within tumor cells, resulting in cell destruction or lysis. Tumor cell lysis leads to release of tumor cell antigens, which stimulates the body’s antitumor immune response. This resulting immune response can extend beyond the injected lesion (Agarwala, 2016; Amgen, 2017b; Orloff, 2016). Investigators have observed regression of uninjected and injected lesions with T-VEC treatment (Agarwala, 2016).
Efficacy and Safety
In a phase 3 trial, T-VEC provided significantly better response rates versus subcutaneously administered GM-CSF in patients with unresectable stage IIIB or IV melanoma (N = 436). Durable response rate (i.e., continuous complete or partial response for six months or longer as a primary outcome) was 16.3% for T-VEC (95% confidence interval [CI] [12.1, 20.5]) versus 2.1% (95% CI [0, 4.5], p < 0.0001) for GM-CSF. T-VEC also was associated with a significantly greater overall response rate (26.4%, 95% CI [21.4, 31.5]) versus GM-CSF (5.7%, 95% CI [1.9, 9.5]) (Andtbacka et al., 2015). In that study, the estimated probability of response lasting at least a year was 65% (95% CI [51, 76]) with T-VEC. Although Andtbacka et al. (2015) found that the median overall survival was longer with T-VEC versus GM-CSF (23.3 versus 18.9 months), this did not reach significance (p = 0.051). The median time to response was 4.1 months. Patients with earlier stages of disease (stages IIIB, IIIC, and IVM1a) with no bone, brain, lung, or other visceral disease derived greater benefit than the overall population in terms of durable and overall response and the risk of developing visceral or bone metastases (Andtbacka et al., 2015; Andtbacka, Ross, et al., 2016; Harrington et al., 2016). T-VEC was well tolerated (see Table 2), and no deaths were considered related to treatment (Andtbacka et al., 2015). 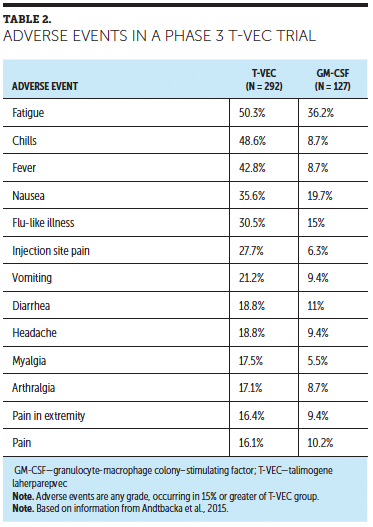
Findings and Steps for Incorporation Into Practice
Introducing a live, attenuated viral therapy, such as T-VEC, requires implementation of infection control procedures for staff members who handle the medication and for anyone exposed to surfaces in contact with the medication. The nurse or other healthcare provider responsible for office setup needs to address staff concerns about possible exposure to the herpes virus. Universal biohazard precautions are required for handling T-VEC (Amgen, 2017b) (see Figure 1). In addition, incorporation requires practice changes, and resources are available to support these infrastructure changes (see Figure 2).
Exposure and Transmission Risk
Reports of healthcare provider and caregiver exposure to or infection with T-VEC are limited. An accidental needlestick to the finger of a healthcare provider during the phase 3 trial led to a herpetic lesion at the injury site. A quantitative polymerase chain reaction test was positive for T-VEC. The case resolved with acyclovir therapy (FDA, 2015). Another accidental needlestick exposure in an animal handler and a splashback to the eye were asymptomatic. In addition, of 177 patients in a family and caregiver surveillance program, 15 reported living with others or having a caregiver or close contact who reported signs and symptoms possibly related to the patients’ participation in a T-VEC clinical trial (Amgen, 2015).
[[{"type":"media","view_mode":"media_original","fid":"34151","field_deltas":{"1":{}},"link_text":null,"fields":{},"attributes":{"height":"952","width":"368","class":"media-image media-element file-media-original","data-delta":"1"}}]]
An interim report of a phase 2 study evaluated the shedding of T-VEC on skin and body fluids and transmission of T-VEC from patients (n = 30) to others. Preliminary findings from the study of biodistribution of T-VEC are shown in Table 3 (Andtbacka, Amatruda, et al., 2016; Gangi & Zager, 2017). The authors noted that transient, low levels of T-VEC were present in blood and urine during treatment, with low risk of transmission to clinicians or close contacts. Harrington et al. (2016) found that, in the event of accidental infection or exposure, T-VEC is susceptible to antiviral agents, such as acyclovir and famciclovir, and the authors further reiterated the low likelihood of household transmission.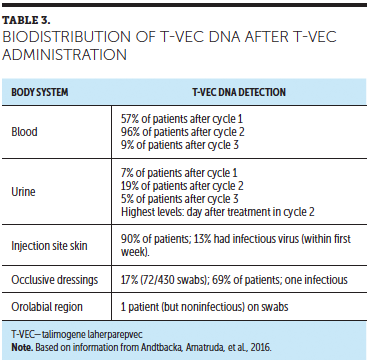
Screening the Clinician and Patient
Healthcare providers who are pregnant or immunocompromised (e.g., those taking corticosteroids or other immunosuppressive therapy, or with conditions such as HIV, leukemia, or lymphoma) should not prepare or administer T-VEC or touch patient injection sites, dressings, or body fluids (Amgen, 2017b). Clinical staff must be trained in infectious control precautions before administering T-VEC. Likewise, patients who are pregnant or immunocompromised should not receive T-VEC (Amgen, 2017b) and, therefore, should be screened for those conditions. In the author’s practice, patients may be on physiologic doses of corticosteroids (i.e., prednisone 10 mg per day or less) and receive T-VEC safely. Patients also should be asked about the health status of close contacts and caregivers (Amgen, 2017b). Clinical judgment can be used in evaluating risk for these individuals. Risk relates directly to contact with drainage from injected lesions and/or bodily fluids. General close contact would not be expected to create a risk. Because T-VEC may result in immune-mediated events, the clinician should consider the risk and benefits in patients with underlying autoimmune disease (Amgen, 2017b).
Initial Patient Evaluation and Education
Pretreatment evaluation includes assessment of lesions to be injected and the patient’s functional status for the ability to travel to treatments, change injection site dressings, and manage adverse events (AEs). Patients with lesions in hard-to-reach locations (e.g., the back) may need someone to change their dressings. Enlisting the help of home healthcare services to change dressings is an option, but those agencies may have concerns about infection control, and such services can be difficult to arrange, depending on the patient’s insurance. If neither family nor home healthcare services are available, then the nurse may have the patient return to the clinic for dressing changes and wound assessments.
Prior to initiating T-VEC therapy, patients and their caregivers should be counseled about benefits and risks of T-VEC, universal precautions, and dressing change procedures. Nurses must be prepared to answer questions about herpes exposure and transmission risk and infection control precautions (see Table 4). They should inform the patient about the time commitment for the administration sessions for planning purposes (e.g., expect about two hours from start to finish; first session lasts the longest). The nurse should review the potential AEs and the possibility of premedicating with ibuprofen or acetaminophen per the doctor or nurse practitioner (if not contraindicated) on the day of treatment (see Figure 3). Patients should be provided with written instructions and other resources, and this education should be repeated on the first day of treatment.
As observed in other immunotherapy studies (Wolchok et al., 2009), some patients receiving T-VEC display pseudoprogression before achieving response. More than half of those who responded to T-VEC first showed at least a 25% increase in lesion size or developed new lesions before improving. Patients should be educated about the phenomenon of pseudoprogression so that they do not become discouraged if it occurs. Nurses should reassure patients that pseudoprogression will not necessarily lead to treatment cessation (Andtbacka et al., 2015).
[[{"type":"media","view_mode":"media_original","fid":"34156","field_deltas":{"2":{}},"link_text":null,"fields":{},"attributes":{"height":"938","width":"753","class":"media-image media-element file-media-original","data-delta":"2"}}]]
T-VEC patient education should include the need for contraception in women of childbearing potential to prevent pregnancy during treatment (Amgen, 2017b). If a pregnant woman is infected with wild-type HSV-1, there is a risk of viral shedding and transmission during birth or through placental transfer, which could lead to serious AEs if the fetus contracts the infection. Although no clinical data on T-VEC infections in pregnant women exist, there could be a risk to the fetus/neonate if T-VEC acted in a similar manner (Amgen, 2017b). The effect of T-VEC on fertility for women and men is not known, as this has not been studied (Amgen, 2017b). However, men are advised to use condoms during sexual activity to prevent inadvertent drug transfer (Amgen, 2017b). For all patients who are sexually active, the use of condoms is generally recommended to prevent potential T-VEC transfer via body fluids (Amgen, 2017b).
Setup
Ideally, two clinicians should be involved in the delivery of T-VEC: one to perform the injection and the other to assist with applying the occlusive dressing. This practice reduces the risk of contaminating the bandages with T-VEC. However, injections can be performed by one person if necessary. If possible, it is advantageous to perform all injections in a treatment room designated for that purpose.
The room should be cleaned between patients. Work surfaces and spills should be contained and treated with a virucidal agent (e.g., 1% sodium hypochlorite, 70% isopropyl alcohol) and absorbent materials. Common disinfectants have reduced T-VEC infectivity by more than 99.99% within one minute. In addition, infectivity was reduced on plastic surfaces that were not cleaned by about 99% following exposure to room temperature for four hours (Amgen, 2015, 2016, 2017b). At the author’s facility, the bed sheets are changed and all surfaces wiped down with an antibacterial solution between T-VEC patients. After all T-VEC treatments for the day are completed, the room is terminally cleaned (i.e., detachable objects are removed, surfaces are cleaned with disinfectant down to the floor, and detachable items are returned after sanitization). The practice may consider scheduling all T-VEC injections on the same day each week to facilitate workflow.
Patients with draining lesions are considered potentially infectious (Amgen, 2017b); their time in the waiting area should be minimized to avoid contact with immunosuppressed individuals. This risk should be low if the draining lesion is covered with an occlusive bandage. Open or ulcerated lesions that are not draining also should be covered with an occlusive bandage, and time in the waiting room should be minimized. Most patients do not have draining lesions and can follow the same intake procedures as other patients. The healthcare provider should inform waiting room staff about which patients should be sent to the clinical area immediately upon arrival rather than be asked to wait in the reception room.
Preinjection Drug Preparation
To evaluate the amount of drug required for injections, it is helpful to review the manufacturer’s recommended dosing. T-VEC is dosed at a lower concentration for the first visit (i.e., 106 [1 million] plaque-forming units [PFU] per milliliter) with the intention of seroconverting patients not previously exposed to HSV (i.e., HSV-negative patients). The second, more concentrated therapeutic dose (i.e., 108 [100 million] PFU/ml) is given three weeks later. Subsequent treatments at the 108 PFU/ml dose occur every two weeks, pending treatment response, tolerability, and patient condition (Amgen, 2017b; Rehman, Silk, Kane, & Kaufman, 2016).
The dose per lesion depends on lesion size, measured as the longest dimension of each lesion (see Table 5). Patients can receive a maximum of 4 ml per treatment session, but that may be an insufficient amount to inject every lesion (Amgen, 2017b). At the first treatment session, lesions are injected in size order from largest to smallest or until the maximum dose is reached. Thereafter, the healthcare provider should treat any new lesions first, then inject in size order, starting with the largest lesion. 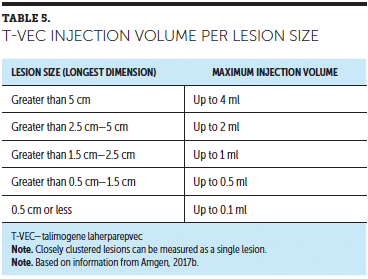
Marking the location and size of each lesion on a lesion tracking sheet provided by the manufacturer is helpful (see Figure 4). A record should be kept for every patient and updated at each treatment session, noting lesion size, change in size, and which lesions were injected. Ideally, all lesions should be measured, even if they will not be injected at a given session, to track any changes in size. Alternatively, photos of the lesions could also be used for tracking. If multiple lesions are present, the clinician should avoid injecting painful lesions. Lesions that recur after response can be reinjected, with at least two weeks between treatments. Treatment should continue for at least six months or until all lesions have resolved or other therapy is needed (Amgen, 2017b).
T-VEC is stored at –90°C to –70°C (–130°F to –94°F) in a freezer, which the manufacturer will provide upon request. Once thawed, it must be administered immediately or refrigerated for a limited time period: 12 hours or less for the lower dose (106 PFU/ml) and 48 hours or less for the higher dose (108 PFU/ml). T-VEC cannot be refrozen after it has thawed (Amgen, 2017b). Therefore, accurate dose measurement is important to avoid wasting medication. Syringes should be drawn up immediately before administration (Amgen, 2017b). If T-VEC is intended for use within 96 hours, a just-in-time delivery ordering system is also available, which requires coordination of scheduling of the patient visit with T-VEC administration (Amgen, 2017a).
The drug order should be verified per each institution’s policy for chemotherapy or biotherapy prior to sending it to the pharmacy. A pharmacy staff member should take the requested quantity of drug from the freezer upon receipt of the order. Both concentrations come in 1-ml vials. Thawing requires about 30–45 minutes, which should be taken into consideration when scheduling the patient’s appointment. This waiting period offers an opportunity to assess the patient further, invite any questions, and apply topical anesthetic (e.g., lidocaine) to areas for planned injection. Patients who have not taken ibuprofen or acetaminophen to reduce risk and severity of fever and chills should be offered such medication per doctor or nurse practitioner order, if not contraindicated.
Each institution typically devises its own medication preparation protocol based on manufacturer guidelines. At the author’s institution, a pharmacist or other pharmacy staff member draws up a separate syringe for each lesion with the requested dosage for that lesion (Rehman et al., 2016). Preparing a syringe with exactly the appropriate amount for each lesion reduces the risk of over- or underinjecting. Small-unit syringes allow for better injection control (Amgen, 2017b). Syringes are filled under a hood at the pharmacy to capture any aerosolized material, with the hood and work space cleaned prior to any other use. The syringes are capped and placed in a chemotherapy bag for transportation to the treatment room.
While wearing gown, gloves, goggles, and mask (or eye shield with mask), the nurse should attach a sterile 22- to 26-gauge needle to each syringe prior to injection (Amgen, 2017b). The needle must be attached securely to prevent drug leakage. Prior to palpating the lesion, the nurse should clean the fingertip of the glove and the injection site with alcohol, allow the areas to dry, then palpate the lesion prior to injecting. Lesions detectable by ultrasound guidance also can be injected (Amgen, 2017b).
Administration Technique
Figure 5 depicts the administration technique from start to finish. A single insertion point is used for each lesion (Amgen, 2017b). The injection should be made on multiple tracks, pulling the needle back after injecting a portion of the drug without withdrawing it from the lesion, then rotating it radially on another track (see Figure 6). This should be repeated until the entire lesion is treated. T-VEC should be injected evenly throughout the lesion. If patients report pain with injection after topical anesthetic, the nurse can add ice packs for 5–10 minutes (Rehman et al., 2016). If pain persists, the healthcare provider should consider injecting 1% lidocaine around the lesion periphery. Local anesthetic should not be injected into the lesion itself, because it may alter the pH and affect T-VEC stability (Hoffner, Iodice, & Gasal, 2016; Rehman et al., 2016). Care should be taken when injecting lesions near major airways, because obstructive airway disorder has been reported following T-VEC treatment (Amgen, 2017b).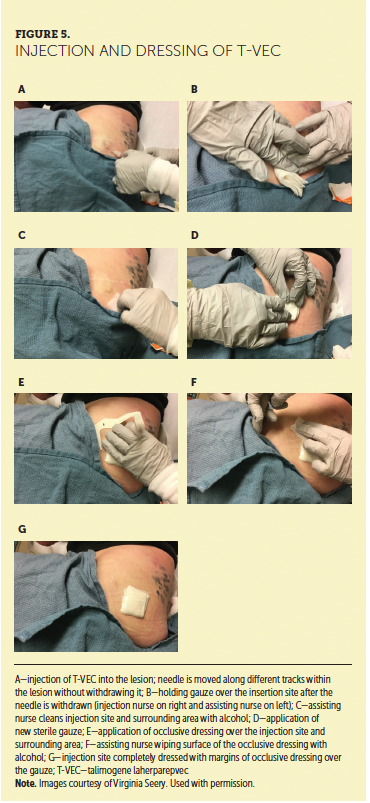
Two injections can be performed in the same lesion if the radial reach of needle does not cover the entire lesion, but this should be avoided if possible. Multiple insertion points in the same lesion offer portals for possible drug leakage, reducing the amount of drug delivered to the patient and increasing exposure risk (Sloot, Rashid, Sarnaik, & Zager, 2016). If it is necessary to inject one lesion at multiple insertion points, then a new needle and syringe should be used for each separate injection (Amgen, 2017b). A temporary dressing should be placed over the first insertion point and taped in place prior to initiating the next injection into the same lesion. It is advisable to avoid having fingers near the lesion being injected to prevent needlesticks.
[[{"type":"media","view_mode":"media_original","fid":"34161","field_deltas":{"3":{}},"link_text":null,"fields":{},"attributes":{"height":"356","width":"750","class":"media-image media-element file-media-original","data-delta":"3"}}]]
Some lesions accept the drug more easily than others, and leakage may occur if the lesion is resistant to drug injection. In such cases, the nurse should apply slow, steady pressure, moving the needle slightly if necessary. Once the drug is fully injected, the needle should be withdrawn slowly to reduce the risk of T-VEC leakage. Once the needle is withdrawn, the area should be immediately covered with sterile gauze for at least 30 seconds. Then, the injection site and surrounding area should be cleaned with an alcohol pad. Next, a new sterile gauze and occlusive dressing are applied. After this dressing is applied, the assisting clinician should wipe the occlusive dressing completely with alcohol as a final step. This occlusive dressing should remain in place for seven days. During the procedure, the nurse should talk with the patient during administration, offering support and inquiring about pain, discomfort, or other AEs.
Postinjection Treatment and Patient Counseling
As part of the postinjection nursing assessment, the patient should again be asked about discomfort, pain, or other symptoms. The patient should be given instructions on postprocedure care. The nurse should advise the patient that if the dressing comes off within the first week or is draining a large amount, then it should be replaced. The occlusive dressing may be covered to allow a patient to shower. Any draining injection sites must remain covered until the drainage stops. Gloves must be worn for all dressing changes. Patients should be advised to avoid touching or scratching the injection sites, even if covered, to prevent inadvertent transfer of T-VEC (Amgen, 2017b). Patients should be provided with gloves, alcohol pads, gauze, and occlusive dressing for home use and instructed about when to call the office if issues arise. The nurse should assess the patient’s and family’s understanding of these instructions and answer any questions.
Follow-Up
The nurse should call the patient one to two days after the first treatment to assess treatment tolerability and offer counsel about managing any AEs that may have occurred. Ideally, patients should be contacted the day before the second treatment with a reminder to take premedication prescribed for them (e.g., ibuprofen or acetaminophen for fever or chills, as well as antiemetic medication). In follow-up telephone calls/visits the nurse should perform a look, listen, and recognize assessment of potential AEs, including nausea and vomiting, flu-like symptoms, herpetic lesions, injection-site pain or swelling, and cellulitis. Of note, the flu-like symptoms typically resolve within 24–72 hours (Amgen, 2017b). As mentioned previously, because immune-related events have been reported in patients treated with T-VEC, the benefits and risks of T-VEC should be discussed in patients who experience such events (Amgen, 2017b).
Another important consideration is distinguishing expected injection site pain or swelling from cellulitis (which occurs rarely, in about 2% of cases). Swelling and drainage from the injection site may occur in some patients, but persistent fever (longer than 48 hours), swelling, erythema, or warmth that expands rapidly should be evaluated for cellulitis. Herpetic cellulitis usually lasts no more than 24–48 hours (Rehman et al., 2016). Because T-VEC–related inflammation usually clears quickly, persistent or worsening symptoms of erythema, fever (up to 102°F or 38.9°C, longer than 48 hours), or leukocytosis may signal bacterial cellulitis (Rehman et al., 2016).
Amgen offers testing of any suspected herpetic lesions that develop during T-VEC therapy for patients and caregivers. The herpes virus in T-VEC remains susceptible to acyclovir therapy, but acyclovir therapy can interfere with the effect of T-VEC on melanoma lesions (Amgen, 2017b). Therefore, the healthcare team needs to carefully balance the risk and benefit of acyclovir therapy in patients receiving T-VEC if a herpetic lesion develops.
Conclusion
T-VEC, the first-in-class intralesional oncolytic viral immunotherapy approved for melanoma, offers patients with local or regional metastatic disease the possibility of improved response rates with a favorable toxicity profile. Its viral composition may raise concerns about exposure and transmission among healthcare staff, patients, and family members. Storage and handling requirements necessitate close coordination with the pharmacy. Nurses familiar with infection control recommendations, drug-handling requirements, and patient education topics detailed in this article are well equipped to contribute to their practice’s adoption of intralesional therapy and overall patient outcomes and satisfaction.
The author gratefully acknowledges Jill Maria Weberding, MPH, BSN, RN, OCN®, for reviewing the manuscript from the community oncology nursing perspective, and Lois J. Loescher, PhD, RN, FAAN, for reviewing the content from a melanoma nursing educational perspective.
About the Author(s)
Virginia Seery, MSN, RN, ANP-BC, is a nurse practitioner in the melanoma/skin cancer program at the Beth Israel Deaconess Medical Center in Boston, MA. The author takes full responsibility for this content. This initiative was funded by the AIM at Melanoma Foundation, with support via unrestricted grants from Amgen, Array Biopharma, Bristol-Myers Squibb, Incyte Corporation, Merck and Co., and Novartis Pharmaceuticals. Writing and editorial support was provided by Eileen McCaffrey, MA, of the Content Firm, LLC, Lisa A. Tushla, PhD, H(ASCP), of Terranova Medica, and Marjorie Joyce, BA. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Seery can be reached at vseery@bidmc.harvard.edu, with copy to CJONEditor@ons.org. (Submitted April 2017. Accepted June 6, 2017.)
References
Agarwala, S.S. (2015). Intralesional therapy for advanced melanoma: Promise and limitation. Current Opinion in Oncology, 27, 151–156. doi:10.1097/cco.0000000000000158
Agarwala, S.S. (2016). The role of intralesional therapies in melanoma. Oncology (Williston Park), 30, 436–441.
Algazi, A., Tsai, K.K., Takamura, K.T., Chen, L., Twitty, C., Dwyer, M., . . . Daud, A. (2016). Intratumoral electroporation of plasmid IL-12 can prime response to anti-PD1/PD-L1 blockade in patients with stage III/IV-M1a melanoma [Abstract CT134]. Cancer Research, 76(Suppl. 14), CT134. doi:10.1158/1538-7445.am2016-ct134
Amgen. (2015). Background information for joint meeting of the Cellular, Tissue, and Gene Therapy, and Oncologic Drugs Advisory Committees. Thousand Oaks, CA: Author. Retrieved from https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMater…
Amgen. (2016). Imlygic™ safety data sheet. Retrieved from http://msds.amgen.com/imlygic-safety-datasheet
Amgen. (2017a). Imlygic® (talimogene laherparepvec). Retrieved from http://www.imlygic.com
Amgen. (2017b). Imlygic® (talimogene laherparepvec) [Package insert]. Retrieved from http://pi.amgen.com/~/media/amgen/repositorysites/pi-amgen-com/imlygic/…
Andtbacka, R.H., Amatruda, T., Mehnert, J., Walker, J., Zager, J.S., Nemunaitis, J, . . . Shilkrut, M. (2016, October). Interim analysis of phase 2 trial to evaluate biodistribution and shedding of talimogene laherparepvec in unresected stage IIIB-IV melanoma patients. Poster presented at the International Meeting on Replicating Virus Therapeutics, Vancouver, BC.
Andtbacka, R.H., Kaufman, H.L., Collichio, F., Amatruda, T., Senzer, N., Chesney, J., . . . Coffin, R.S. (2015). Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. Journal of Clinical Oncology, 33, 2780–2788. doi:10.1200/jco.2014.58.3377
Andtbacka, R.H., Ross, M., Puzanov, I., Milhem, M., Collichio, F., Delman, K.A., . . . Kaufman, H.L. (2016). Patterns of clinical response with talimogene laherparepvec (T-VEC) in patients with melanoma treated in the OPTiM phase III clinical trial. Annals of Surgical Oncology, 23, 4169–4177. doi:10.1245/s10434-016-5286-0
de la Monte, S.M., & Hutchins, G.M. (1986). Fatal disseminated bacillus Calmette-Guerin infection and arrested growth of cutaneous malignant melanoma following intralesional immunotherapy. American Journal of Dermatopathology, 8, 331–335.
Foote, M., Read, T., Thomas, J., Wagels, M., Burmeister, B., & Smithers, B.M. (2017). Results of a phase II, open-label, non-comparative study of intralesional PV-10 followed by radiotherapy for the treatment of in-transit or metastatic melanoma. Journal of Surgical Oncology. Advance online publication. doi:10.1002/jso.24580
Gangi, A., & Zager, J.S. (2017). The safety of talimogene laherparepvec for the treatment of advanced melanoma. Expert Opinion on Drug Safety, 16, 265–269. doi:10.1080/14740338.2017.1274729
Harrington, K.J., Andtbacka, R.H., Collichio, F., Downey, G., Chen, L., Szabo, Z., & Kaufman, H.L. (2016). Efficacy and safety of talimogene laherparepvec versus granulocyte-macrophage colony-stimulating factor in patients with stage IIIB/C and IVM1a melanoma: Subanalysis of the phase III OPTiM trial. OncoTargets and Therapy, 9, 7081–7093. doi:10.2147/ott.s115245
Hoffner, B., Iodice, G.M., & Gasal, E. (2016). Administration and handling of talimogene laherparepvec: An intralesional oncolytic immunotherapy for melanoma. Oncology Nursing Forum, 43, 219–226. doi:10.1188/16.ONF.219-226
Mastrangelo, M.J., Bellet, R.E., Berkelhammer, J., & Clark, W.H., Jr. (1975). Regression of pulmonary metastatic disease associated with intralesional BCG therapy of intracutaneous melanoma metastases. Cancer, 36, 1305–1308.
Orloff, M. (2016). Spotlight on talimogene laherparepvec for the treatment of melanoma lesions in the skin and lymph nodes. Oncolytic Virotherapy, 5, 91–98. doi:10.2147/ov.s99532
Puzanov, I., Milhem, M.M., Minor, D., Hamid, O., Li, A., Chen, L., . . . Andtbacka, R.H. (2016). Talimogene laherparepvec in combination with ipilimumab in previously untreated, unresectable stage IIIB-IV melanoma. Journal of Clinical Oncology, 34, 2619–2626. doi:10.1200/jco.2016.67.1529
Rehman, H., Silk, A.W., Kane, M.P., & Kaufman, H.L. (2016). Into the clinic: Talimogene laherparepvec (T-VEC), a first-in-class intratumoral oncolytic viral therapy. Journal for Immunotherapy of Cancer, 4, 53. doi:10.1186/s40425-016-0158-5
Sloot, S., Rashid, O.M., Sarnaik, A.A., & Zager, J.S. (2016). Developments in intralesional therapy for metastatic melanoma. Cancer Control, 23, 12–20.
Thompson, J.F., Agarwala, S.S., Smithers, B.M., Ross, M.I., Scoggins, C.R., Coventry, B.J., . . . Wachter, E.A. (2015). Phase 2 study of intralesional PV-10 in refractory metastatic melanoma. Annals of Surgical Oncology, 22, 2135–2142. doi:10.1245/s10434-014-4169-5
U.S. Food and Drug Administration. (2015). Cellular, Tissue, and Gene Therapies Advisory Committee and Oncologic Drugs Advisory Committee meeting. BLA 125518 talimogene laherparepvec (Amgen). Retrieved from https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMater…
Wolchok, J.D., Hoos, A., O’Day, S., Weber, J.S., Hamid, O., Lebbé, C., . . . Hodi, F.S. (2009). Guidelines for the evaluation of immune therapy activity in solid tumors: Immune-related response criteria. Clinical Cancer Research, 15, 7412–7420. doi:10.1158/1078-0432.ccr-09-1624




