BRAF/MEK Inhibitor Therapy: Consensus Statement From the Faculty of the Melanoma Nursing Initiative on Managing Adverse Events and Potential Drug Interactions
Background: BRAF/MEK inhibitor therapy improves outcomes in BRAF V600E- and V600K-mutated unresectable or metastatic melanoma. However, these regimens are associated with adverse events (AEs) that may lead to unnecessary drug modifications and discontinuations or potentially serious sequelae. In addition, drug–drug interactions (DDIs) may result in AEs or altered therapeutic efficacy.
Objectives: This article presents consensus statements to guide nurses in the prevention, recognition, and management of AEs and potential DDIs associated with BRAF/MEK inhibitor therapy.
Methods: Members of the Melanoma Nurse Initiative reviewed the current literature and clinical experience related to AEs and DDIs associated with BRAF/MEK inhibitor therapy.
Findings: The care step pathways provided for select AEs represent a proactive, comprehensive nursing care plan to support optimal patient outcomes. Recommendations are also offered for preventing and managing DDIs.
Jump to a section
In the past several years, novel therapeutic options have been developed for patients with v-Raf murine sarcoma viral oncogene homolog B (BRAF)–mutant unresectable, high-risk, or metastatic melanoma (Eroglu & Ribas, 2016). About 50% of all advanced melanomas have an activating mutation in the BRAF proto-oncogene, which encodes a kinase in the mitogen-activated protein kinase (MAPK) pathway that helps regulate cell growth. Mutations in the BRAF gene can cause uncontrolled activation of the BRAF protein, thereby initiating a series of intracellular phosphorylation events that promote oncogenesis. Two of the most common BRAF-activating mutations are found in codon 600 of this protein and are designated V600E and V600K (Medina & Lewis, 2016). Vemurafenib (Zelboraf®) and dabrafenib (Tafinlar®) are inhibitors of BRAF that are approved by the U.S. Food and Drug Administration (FDA) as single agents for patients with metastatic melanoma with the BRAF V600E mutation (Genentech, 2017; Novartis, 2016).
Single-agent BRAF inhibitor therapy is associated with high rates of resistance and the development of secondary cutaneous malignancies, owing to paradoxical MAPK pathway activation in skin cells that do not have a BRAF mutation (wild-type cells) (Medina & Lewis, 2016). Mitogen-activated protein kinase kinase (MEK), a downstream kinase in the MAPK pathway, provides another therapeutic target for regulating cellular proliferation (Eroglu & Ribas, 2016). Drugs that inhibit MEK include trametinib (Mekinist®) and cobimetinib (Cotellic®) (Genentech, 2016; Novartis, 2017a). These agents inhibit hyperactive signaling in the MAPK pathway but have relatively low response rates as single agents compared with BRAF inhibitors (Medina & Lewis, 2016). Combination BRAF/MEK inhibitor therapy is associated with superior overall and progression-free survival, overall response rate, and duration of response versus single-agent BRAF inhibitor therapy (Larkin et al., 2014; Long et al., 2014; Robert et al., 2015). In addition, in these studies, the incidence of new primary melanomas, cutaneous squamous cell carcinoma, basal cell carcinoma, and keratoacanthoma decreased with the combination therapy versus single-agent BRAF inhibitor therapy. Two FDA-approved BRAF/MEK inhibitor combination regimens are available: (a) dabrafenib and trametinib and (b) vemurafenib and cobimetinib (Genentech, 2016, 2017; Novartis, 2016, 2017a). A third BRAF/MEK inhibitor combination therapy, encorafenib and binimetinib, has demonstrated similarly favorable results (in efficacy and safety profiles) in a phase 3 clinical trial (Dummer et al., 2016) and is undergoing FDA review. To date, no controlled clinical trials have directly compared BRAF/MEK inhibitor combination therapy regimens. These drugs are referred to as targeted therapies because they act on specific protein targets in the MAPK pathway.
BRAF/MEK inhibitor drugs are orally administered and have enhanced convenience compared with injectable therapies, but some barriers hamper appropriate use. Adverse events (AEs) associated with BRAF/MEK inhibitors differ from those seen with chemotherapy or immunotherapy and can be challenging to recognize and manage (Dy & Adjei, 2013). Faculty of the Melanoma Nursing Initiative (MNI) convened to define supportive care challenges associated with the use of BRAF/MEK inhibitor therapy. The MNI evaluated the literature and clinical experience to recommend nursing interventions to improve patient and therapeutic outcomes. The authors made recommendations in the following areas:
• Patient counseling and education about BRAF testing
• Administration and dosing, with a focus on dosage adjustments related to AEs
• AE management, including strategies to educate and assess patients’ understanding, as well as specific care step pathways (CSPs) to guide nursing interventions regarding prompt recognition and management of AEs of particular concern
• Drug–drug interactions (DDIs), with a focus on identifying concomitant medications for which potential exists for an interaction that would decrease drug effectiveness or exacerbate toxicities
Testing
Both FDA-approved BRAF/MEK inhibitor combination therapies require positive identification of the BRAF V600E or V600K mutation as detected by an FDA-approved test. Patients with wild-type BRAF melanoma are not suitable candidates for these treatments (Genentech, 2017; Novartis, 2017b) because BRAF inhibitors may promote tumor growth in cells with wild-type BRAF (Medina & Lewis, 2016). Two tests are approved by the FDA for the detection of BRAF V600 mutations: the THxID™ BRAF kit (for dabrafenib and trametinib) and the cobas 4800 BRAF V600 Mutation Test (for vemurafenib and cobimetinib) (FDA, 2017). Both use polymerase chain reaction technology to evaluate melanoma tissue for V600 mutations. The THxID test detects either V600E or V600K mutations, whereas the cobas test detects only V600E. Other assays based on sequencing methods are being evaluated by the FDA and are used at some centers because of their improved sensitivity and ability to analyze multiple genes (Ma et al., 2016); however, insurance coverage may vary.
A link to the FDA website that lists approved companion diagnostic tests, along with links to other nursing and patient resources, can be found in Figure 1. Oncology nurses are in a key position to field questions related to testing. Patients should be informed about the need for specific BRAF testing that is tailored to the treatment planned and that is likely to be reimbursed. If testing has previously been performed, but not via an approved method, clearly explaining the rationale for repeat testing is necessary, given the potential reimbursement issues. Anticipating the need for possible repeat analysis and expediting necessary arrangements will not only minimize delays in commencing therapy but will improve patient satisfaction. 
Drug Administration and Dosages
BRAF and MEK inhibitors are self-administered oral agents. Approved doses and recommended dose modifications are shown in Table 1. To reiterate, dabrafenib dosed at 150 mg twice a day is combined with trametinib at 2 mg daily. Vemurafenib dosed at 960 mg twice a day is combined with cobimetinib at 60 mg daily. Dabrafenib (at the 150 mg dose, twice a day) and vemurafenib (at the 960 mg dose, twice a day) are also approved as monotherapy. Some global comments can be made about dosing. Of note, dabrafenib and trametinib should be taken on an empty stomach. All the agents are stored at room temperature, except for trametinib, which requires refrigeration. Dabrafenib, trametinib, and vemurafenib are given on a continuous daily basis (Genentech, 2017; Novartis, 2016, 2017a), although some clinics use drug holidays (brief treatment breaks) during the treatment course when patients are having AEs that affect their activities of daily living. Cobimetinib is given for the first 21 days of a 28-day cycle, followed by seven days off of the drug (Genentech, 2016). A generalized dose-reduction scheme for toxicity is shown, and dose-reduction schemes for specific AEs are outlined in more detail in the CSPs. 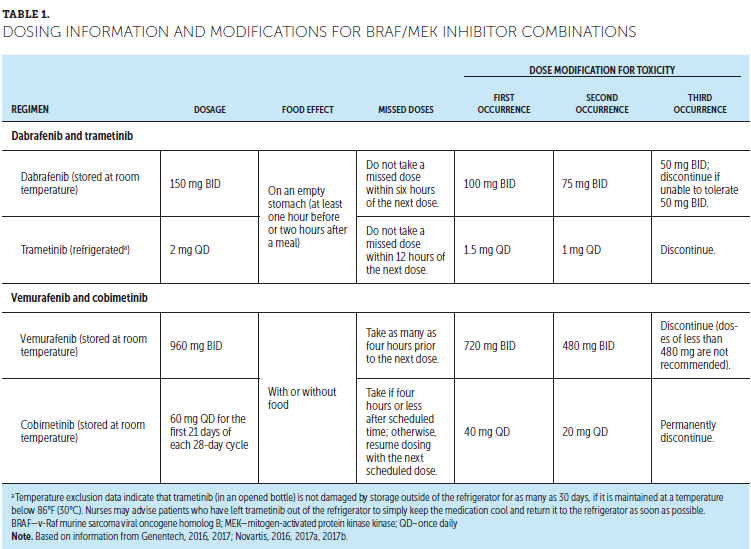
Adverse Events and Care Step Pathways
Table 2 lists some common AEs associated with BRAF/MEK inhibitors. Although some similarities can be found in the AE profiles across the targeted therapy combinations, some individual differences exist. In particular, photosensitivity is more frequently associated with vemurafenib-containing regimens, whereas pyrexia is more frequently associated with dabrafenib-containing regimens. MEK inhibitor drugs are associated with cardiomyopathy, while BRAF inhibitor drugs are associated with QT interval prolongation.
The available AE data were obtained from trials of BRAF/MEK inhibitor combination therapies in patients with previously untreated melanoma. As the therapeutic options available for patients with BRAF-mutated melanoma expand, whether the toxicity profile will differ is unknown. Members of the MNI agreed that patients receiving targeted therapies who were previously treated with immunotherapy should be monitored carefully for overlapping toxicities, such as rash, fatigue, joint pain, diarrhea, and altered liver function, because these AEs are also observed with immune checkpoint inhibitors and may result in cumulative toxicities (Welsh & Corrie, 2015).
Each of the four CSPs presented in this article represents a notable AE associated with targeted therapy and incorporates essential components of the nursing assessment specific to that AE. Look, listen, and recognize categories within the nursing assessment section direct the nurse to a specific set of symptom-related queries to ask the patient and/or caregiver and highlights additional information important to optimal management. Wherever possible, the grading in the CSPs is based on the National Cancer Institute’s (2010) Common Terminology Criteria for Adverse Events (CTCAE), which is, in some cases, supplemented with information from the package inserts of the drugs (Genentech, 2016, 2017; Novartis, 2016, 2017a). Each CSP describes overall management strategies and nursing-specific interventions. Where applicable, prevention strategies, as well as strategies specific to each AE grade, are listed, including dose reductions or modifications. Patient counseling, recommendations for additional care, and referral to specialty or ancillary care providers are included in the management section, as appropriate.
General Education
Patient education regarding a recommended treatment regimen is a key component of the oncology nursing role. The general components of pretreatment education include reviewing treatment expectations (of provider, patient, and family members), addressing logistic and financial considerations, and discussing potential toxicities. Education should be comprehensive and individualized for patients and their plan of care. If possible, that education should be given in the presence of a patient caregiver or support person, with validation of patient comprehension and compliance. Foundational to any AE discussion is educating each patient to promptly recognize and report any new or worsening symptoms, whether or not the patient thinks they are related (Welsh & Corrie, 2015). Providing clear instruction on when, why, and how to contact the patient’s oncology provider is critical and should be reiterated at every visit. New or worsening symptoms, and any symptoms indicative of a serious AE, such as vision change, bleeding, or cardiotoxicity, are critical to report (Welsh & Corrie, 2015).
General education also includes counseling concerning the use of nonhormonal methods of contraception to avoid embryo-fetal toxicity associated with these agents. Barrier (nonhormonal) methods are preferred because BRAF inhibitors may render hormonal contraceptives ineffective (attributable to DDIs). Given the importance of avoiding fetal toxicity and the potential impact on fertility, the oncology team should have open, candid discussions about family planning with patients of childbearing age and note any potential barriers or contraindications to future goals. A possible recommendation may be a referral to a fertility specialist to discuss potential sperm or egg banking as an option. Nurses continue to support the process by reinforcing conversations and addressing patient concerns. Patients may also benefit from other educational materials and online resources to support them on their treatment journey, including resources from the manufacturers, as well as advocacy organizations.
Common Adverse Events
Pyrexia
One of the most common AEs associated with BRAF/MEK inhibitor combination therapy, particularly dabrafenib and trametinib, is pyrexia, which is elevated body temperature in the absence of clinical or microbiologic evidence of infection (Lee et al., 2014). In clinical trials of dabrafenib and trametinib, pyrexia usually appeared within one to two months and plateaued about six months after initiation of treatment (Long et al., 2014; Menzies et al., 2015). The etiology of pyrexia is not well understood but is hypothesized to be an off-target effect (i.e., related to interactions between the drug and proteins other than BRAF/MEK) (Atkinson et al., 2016; Lee et al., 2014; Menzies et al., 2015).
Figure 2 shows the MNI CSP for management of pyrexia. Current CTCAE grading criteria do not include grading categories for this novel effect; the fever criteria in the CTCAE were designed for the management of febrile neutropenia, which has a different etiology, and the temperature cut points in the CTCAE are different from those used for drug holds with targeted therapy. In addition to temperature elevation, management of pyrexia is also driven by symptoms (e.g., rigors, hypotension, dehydration, renal failure) or recurrent episodes. In general, management of pyrexia involves maintaining hydration, providing supportive care for symptoms, and ensuring that patients are adhering to treatment recommendations, particularly those concerning treatment holds or dose adjustments. Based on MNI members’ experience, premedication with acetaminophen or nonsteroidal anti-inflammatory drugs is not useful in preventing pyrexia. This practice has been adopted by some clinicians in the community but is not evidence based.
[[{"type":"media","view_mode":"media_original","fid":"34186","field_deltas":{"1":{}},"link_text":null,"fields":{},"attributes":{"height":"773","width":"755","class":"media-image media-element file-media-original","data-delta":"1"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34191","field_deltas":{"2":{}},"link_text":null,"fields":{},"attributes":{"height":"866","width":"755","class":"media-image media-element file-media-original","data-delta":"2"}}]]
Management strategies for recurrent pyrexia are also discussed in the CSP. Corticosteroids (e.g., prednisone 10 mg daily) for at least five days are recommended for a second or subsequent pyrexia if temperature does not return to baseline within three days of onset, or if pyrexia is associated with complications, including dehydration, hypotension, renal failure, and severe chills or rigors, in the absence of evidence of active infection (Novartis, 2017a). For patients with steroid-refractory pyrexia, an increased dosage of prednisone with a tapering course (for instance, beginning at 25 mg per day and titrating downward) in addition to withholding therapy until symptoms return to baseline or grade 1 may help to control pyrexia (Atkinson et al., 2016). Targeted therapy can then be restarted at a lower dose and gradually titrated upward. MNI members recommend that steroids be taken with food, preferably in the morning, to minimize problems with insomnia. They should not be taken at the same time as the BRAF/MEK inhibitors. If high temperatures continue to recur, even with antipyretics, prednisone, dose reductions, or drug holidays, severely affecting a patient’s quality of life, a change in targeted therapy to a different combination regimen could be considered.
Skin Toxicities
Also associated with BRAF/MEK inhibitor combination therapy are skin toxicities, which may have a tremendous impact on quality of life. Rashes may have a variety of presentations, including maculopapular, verrucous, hyperkeratotic, keratosis pilaris-like, and acneiform (Lacouture et al., 2013; Livingstone, Zimmer, Vaubel, & Schadendorf, 2014; Segaert, 2008; Segaert & Van Cutsem, 2005). Pruritus (itching) and xerosis (dry skin) can occur in the absence of rash or after the rash resolves (Bryce & Boers-Doets, 2014; Valentine et al., 2015). Photosensitivity is a unique toxicity that is characterized by a burning sensation with marked erythema or edema on sun-exposed skin; it is most frequently observed with vemurafenib-based regimens and is attributed to exposure to ultraviolet A radiation (Bryce & Boers-Doets, 2014; Mavropoulos & Wang, 2014). If persistent photosensitive reactions do not respond to dose reductions or drug holidays, a change from vemurafenib to another combination therapy (such as dabrafenib and trametinib) should be considered.
As highlighted in Figure 3, nurses are in a key position to educate patients to institute appropriate self-care and prevention strategies to minimize skin toxicities. Because of photosensitivity issues, direct and indirect sunlight should be avoided whenever possible. Patients should be counseled to practice sun avoidance by using sunscreen and lip balm with an SPF of at least 30 and wearing sun-protective clothing, including sunglasses, when outside. Patients who work outdoors should be counseled about the possibility of modifying their jobs while on this therapy because of high risk for photosensitivity. Gentle skin care should be encouraged, including avoidance of harsh soaps (alcohol-based or with fragrances) and regular use of emollient-based, non-irritant moisturizers at least daily. Some skin toxicities are typically observed within days of initiation of therapy, so educating patients and their families about these preventive measures while emphasizing when to contact the oncology team is essential.
[[{"type":"media","view_mode":"media_original","fid":"34196","field_deltas":{"3":{}},"link_text":null,"fields":{},"attributes":{"height":"938","width":"751","class":"media-image media-element file-media-original","data-delta":"3"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34201","field_deltas":{"4":{}},"link_text":null,"fields":{},"attributes":{"height":"934","width":"750","class":"media-image media-element file-media-original","data-delta":"4"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34111","field_deltas":{"5":{}},"link_text":null,"fields":{},"attributes":{"height":"258","width":"748","class":"media-image media-element file-media-original","data-delta":"5"}}]]
Another novel cutaneous toxicity related to these regimens is the development of benign, premalignant, or malignant secondary skin neoplasms, including actinic keratosis, keratoacanthoma, basal cell carcinoma, and squamous cell carcinoma (Flaherty et al., 2012; Larkin et al., 2014; Long et al., 2014). This toxicity is thought to be caused by activation of the MAPK pathway signaling in cells that have not yet acquired a mutation in the BRAF protein. For these reasons, a full skin examination (including oral and genital areas) by a dermatologist with expertise in skin cancer should be performed before beginning treatment, every two months during treatment, and as many as seven months after treatment discontinuation (Genentech, 2016; Novartis, 2016, 2017a). Suspicious lesions should be examined by a dermatologist; given the associated risk of secondary skin malignancy, a biopsy is recommended. Secondary malignancies are typically managed surgically without dose interruption or modification (Rubin, in press).
Other cutaneous disorders may occur. Benign growths (e.g., squamous papillomas, warts) may be a cosmetic or quality-of-life concern for some patients and can be treated with topical agents, such as fluorouracil, or by cryotherapy or curettage (Mandalà, Massi, & De Giorgi, 2013; Welsh & Corrie, 2015). Palmoplantar hyperkeratosis, which often presents as thickened yellow plaques over friction sites (e.g., on the soles of the feet) (Macdonald, Macdonald, Golitz, LoRusso, & Sekulic, 2015), can occur rapidly after the initiation of BRAF inhibitor therapy (Anforth et al., 2012; Livingstone et al., 2014). Preventive measures include avoidance of tight-fitting shoes and clothing. Hyperkeratotic lesions can be pared down mechanically by a podiatrist or treated with keratolytic medications or topical steroids (Livingstone et al., 2014; Macdonald et al., 2015). For severe cutaneous symptoms, dose reductions or treatment holidays may be necessary until symptoms resolve or are reduced to grade 1. Alopecia has also been noted with BRAF/MEK inhibitor therapy (Livingstone et al., 2014; Sinha et al., 2012), although the authors have not observed this frequently. Hair changes (such as in curliness or graying) have also been noted (Mavropoulos & Wang, 2014). No medical management is necessary for these changes; however, if patients are bothered by these effects, topical minoxidil may be used for alopecia, and cosmetic techniques, such as hair coloring, may be employed (Rubin, in press).
Rare But Serious Adverse Events
Ocular Toxicities
Eye-related AEs are rare, but may be serious if not recognized and promptly treated. The potential for patients to become blind should not be overlooked. These AEs appear to be related to an inflammatory response or breakdown of the blood–retina barrier (Huang et al., 2009). Uveitis appears to be associated with BRAF inhibitor therapy, whereas retinal disorders are associated with MEK inhibitor therapy (Schoenberger & Kim, 2013). In many instances, ocular AEs are transient and will resolve, but persistent symptoms necessitate dose interruption with dose reduction on improvement or, in severe cases, require permanent drug discontinuation (see Figure 4). Patients should be advised to immediately report any visual disturbances, and an ophthalmic examination should be completed at baseline and whenever patients report eye symptoms.
[[{"type":"media","view_mode":"media_original","fid":"34206","field_deltas":{"6":{}},"link_text":null,"fields":{},"attributes":{"height":"940","width":"751","class":"media-image media-element file-media-original","data-delta":"6"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34211","field_deltas":{"7":{}},"link_text":null,"fields":{},"attributes":{"height":"652","width":"747","class":"media-image media-element file-media-original","data-delta":"7"}}]]
Cardiac Toxicities
Cardiotoxicity, including cardiomyopathy and QT prolongation, is a potentially serious consequence of treatment with BRAF/MEK inhibitor therapy. Figure 5 provides a CSP for the evaluation and management of cardiotoxicity. A thorough history can reveal preexisting cardiac conditions that may preclude targeted therapy. Vemurafenib treatment should not be initiated in patients with uncorrectable electrolyte abnormalities, QTc of greater than 500 milliseconds, or long QT syndrome, or in patients who are taking drugs known to prolong the QT interval (e.g., anti-arrhythmics, azole antifungals, fluoroquinolones) (Nachimuthu, Assar, & Schussler, 2012). Thorough cardiac testing should be completed, with left ventricular ejection fraction assessed before initiation of therapy and at regular intervals during therapy (one month after initiation and every two to three months thereafter) (Genentech, 2016, 2017; National Cancer Institute, 2010; Novartis, 2016, 2017a). Both BRAF/MEK inhibitor combinations are associated with hypertension, so careful monitoring is required, particularly for patients with preexisting hypertension.
[[{"type":"media","view_mode":"media_original","fid":"34216","field_deltas":{"8":{}},"link_text":null,"fields":{},"attributes":{"height":"761","width":"753","class":"media-image media-element file-media-original","data-delta":"8"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34221","field_deltas":{"9":{}},"link_text":null,"fields":{},"attributes":{"height":"715","width":"754","class":"media-image media-element file-media-original","data-delta":"9"}}]]
Other Adverse Events
Table 3 lists additional AEs and laboratory abnormalities associated with BRAF/MEK inhibitors, along with information on appropriate questions to determine the AE and recommendations for drug holds, dose reductions, and discontinuations specific to the AE.
[[{"type":"media","view_mode":"media_original","fid":"34136","field_deltas":{"10":{}},"link_text":null,"fields":{},"attributes":{"height":"938","width":"751","class":"media-image media-element file-media-original","data-delta":"10"}}]]
[[{"type":"media","view_mode":"media_original","fid":"34141","field_deltas":{"11":{}},"link_text":null,"fields":{},"attributes":{"height":"770","width":"747","class":"media-image media-element file-media-original","data-delta":"11"}}]]
Drug-Drug Interactions
Dabrafenib, vemurafenib, and cobimetinib are metabolized by the cytochrome P450 (CYP450) system (Genentech, 2016, 2017; Novartis, 2016), which includes more than 50 CYP enzymes. This hepatic system metabolizes many other drugs; therefore, targeted therapies have significant potential for DDIs. Trametinib is metabolized by deacetylation and glucuronidation biotransformation pathways (Novartis, 2017a) and is, therefore, less likely to be involved in DDIs.
Because DDIs can diminish efficacy and promote AEs, nurses should have a working knowledge of these potential interactions, ensuring that all members of the medical team, including primary care providers and other specialists, communicate to address any potential DDIs. Table 4 shows important DDIs that may affect the use of BRAF/MEK inhibitors. In particular, dabrafenib induces CYP3A4, which can result in decreased concentrations and loss of efficacy for hormonal contraceptive substrates and proton pump inhibitors (Flockhart, 2007; Novartis, 2016). 
Targeted therapies can also prolong the QT interval. Therefore, identifying concomitant medications that may also have this effect is important (Welsh & Corrie, 2015). Medications known to increase the risk of QT prolongation include anti-arrhythmics, certain antidepressants, some antiemetics (e.g., ondansetron), atypical antipsychotics, azole antifungals, fluoroquinolones, and methadone (Nachimuthu et al., 2012). For vemurafenib, the risk of QT prolongation is known to be concentration dependent (Genentech, 2017). Accordingly, concomitant medications that increase vemurafenib plasma concentrations should be avoided in patients at risk for QT prolongation. By promoting open communication concerning concomitant therapies, nurses can help alert various members of the oncology team to the possible need for DDI-related dose modifications or alternative therapies. In addition, patients should be encouraged, whenever possible, to have all of their medications filled by one pharmacy to ensure familiarity with the full medication list and to avoid polypharmacy issues.
Implications for Nursing and Conclusion
Targeted therapy with BRAF/MEK inhibitor combinations improves overall survival in patients with BRAF V600E- or V600K-mutated metastatic melanoma but is also associated with AEs that need concerted management if patients are to stay on therapy. By educating patients and ensuring prompt AE recognition and management, nurses can minimize the impact of AEs on patients’ lives and increase the likelihood of therapy continuation with limited interruptions. Nursing awareness and a proactive stance to DDIs across the entire multidisciplinary team can also improve therapeutic outcomes. Finally, nurses can improve AE management by establishing connections with referral clinicians to care for more challenging or serious AEs and to ensure optimal AE management and prevention of more serious sequelae.
The authors gratefully acknowledge Jill Maria Weberding, MPH, BSN, RN, OCN®, for reviewing the manuscript from the community oncology nursing perspective and Lois J. Loescher, PhD, RN, FAAN, for reviewing the manuscript from a nursing education perspective.
About the Author(s)
Maria Czupryn, ARNP, AOCNP®, is a nurse practitioner and was, at the time of this writing, in the Department of Cutaneous Oncology and the Comprehensive Melanoma Research Center at the Moffitt Cancer Center in Tampa, FL, and Jennifer Cisneros, PharmD, is a clinical pharmacist in the Department of Cutaneous Oncology and the Comprehensive Melanoma Research Center at the Moffitt Cancer Center. The authors take full responsibility for this content. This supplement was funded by the AIM at Melanoma Foundation, with support via unrestricted grants from Amgen, Array Biopharma, Bristol-Myers Squibb, Incyte Corporation, Merck and Co., and Novartis Pharmaceuticals. Writing and editorial support was provided by Sharon L. Cross, PhD; Lisa A. Tushla, PhD, H(ASCP), of Terranova Medica; and Marjorie Joyce, BA. Czupryn has previously consulted for and has served on speakers bureaus for Merck and Co. and Novartis. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Czupryn can be reached at maria.czupryn@moffitt.org, with copy to CJONEditor@ons.org. (Submitted April 2017. Accepted June 4, 2017.)
References
Anforth, R.M., Blumetti, T.C., Kefford, R.F., Sharma, R., Scolyer, R.A., Kossard, S., . . . Fernandez-Peñas, P. (2012). Cutaneous manifestations of dabrafenib (GSK2118436): A selective inhibitor of mutant BRAF in patients with metastatic melanoma. British Journal of Dermatology, 167, 1153–1160. doi:10.1111/j.1365-2133.2012.11155.x
Atkinson, V., Long, G.V., Menzies, A.M., McArthur, G., Carlino, M.S., Millward, M., . . . Cebon, J. (2016). Optimizing combination dabrafenib and trametinib therapy in BRAF mutation-positive advanced melanoma patients: Guidelines from Australian melanoma medical oncologists. Asia-Pacific Journal of Clinical Oncology, 12(Suppl. 7), 5–12. doi:10.1111/ajco.12656
Bristol-Myers Squibb. (2017). Eliquis® (apixaban) [Package insert]. Retrieved from http://packageinserts.bms.com/pi/pi_eliquis.pdf
Bryce, J., & Boers-Doets, C.B. (2014). Non-rash dermatologic adverse events related to targeted therapies. Seminars in Oncology Nursing, 30, 155–168. doi:10.1016/j.soncn.2014.05.003
Davis, M.E. (2016). Ocular toxicity of tyrosine kinase inhibitors. Oncology Nursing Forum, 43, 235–243. doi:10.1188/16.ONF.235-243
de Golian, E., Kwong, B.Y., Swetter, S.M., & Pugliese, S.B. (2016). Cutaneous complications of targeted melanoma therapy. Current Treatment Options in Oncology, 17(11), 57. doi:10.1007/s11864-016-0434-0
Dummer, R., Ascierto, P.A., Gogas, H.J., Arance, A., Mandala, M., Liszkay, G., . . . Flaherty, K.T. (2016, November). Results of COLUMBUS Part 1: A phase 3 trial of encorafenib (ENCO) plus binimetinib (BINI) versus vemurafenib (VEM) or ENCO in BRAF-mutant melanoma. Paper presented at the Society for Melanoma Research 2016 Congress, Boston, MA. Retrieved from http://www.arraybiopharma.com/files/6314/7865/9329/COLUMBUSprimary_SMR2…
Dy, G.K., & Adjei, A.A. (2013). Understanding, recognizing, and managing toxicities of targeted anticancer therapies. CA: A Cancer Journal for Clinicians, 63, 249–279. doi:10.3322/caac.21184
Eroglu, Z., & Ribas, A. (2016). Combination therapy with BRAF and MEK inhibitors for melanoma: Latest evidence and place in therapy. Therapeutic Advances in Medical Oncology, 8, 48–56. doi:10.1177/1758834015616934
Flaherty, K.T., Robert, C., Hersey, P., Nathan, P., Garbe, C., Milhem, M., . . . Schadendorf, D. (2012). Improved survival with MEK inhibition in BRAF-mutated melanoma. New England Journal of Medicine, 367, 107–114. doi:10.1056/NEJMoa1203421
Flockhart, D.A. (2007). P450 drug interaction table. Retrieved from http://bit.ly/2uf6AcA
Genentech. (2016). Cotellic® (cobimetinib) [Package insert]. Retrieved from https://www.gene.com/download/pdf/cotellic_prescribing.pdf
Genentech. (2017). Zelboraf® (vemurafenib) [Package insert]. Retrieved from https://www.gene.com/download/pdf/zelboraf_prescribing.pdf
Huang, W., Yang, A.H., Matsumoto, D., Collette, W., Marroquin, L., Ko, M., . . . Younis, H.S. (2009). PD0325901, a mitogen-activated protein kinase kinase inhibitor, produces ocular toxicity in a rabbit animal model of retinal vein occlusion. Journal of Ocular Pharmacology and Therapeutics, 25, 519–530. doi:10.1089/jop.2009.0060
Lacouture, M.E., Duvic, M., Hauschild, A., Prieto, V.G., Robert, C., Schadendorf, D., . . . Joe, A.K. (2013). Analysis of dermatologic events in vemurafenib-treated patients with melanoma. Oncologist, 18, 314–322. doi:10.1634/theoncologist.2012-0333
Larkin, J., Ascierto, P.A., Dréno, B., Atkinson, V., Liszkay, G., Maio, M., . . . Ribas, A. (2014). Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. New England Journal of Medicine, 371, 1867–1876. doi:10.1056/NEJMoa1408868
Lee, C.I., Menzies, A.M., Haydu, L.E., Azer, M., Clements, A., Kefford, R.F., & Long, G.V. (2014). Features and management of pyrexia with combined dabrafenib and trametinib in metastatic melanoma. Melanoma Research, 24, 468–474. doi:10.1097/CMR.0000000000000110
Livingstone, E., Zimmer, L., Vaubel, J., & Schadendorf, D. (2014). BRAF, MEK and KIT inhibitors for melanoma: Adverse events and their management. Chinese Clinical Oncology, 3(3), 29. doi:10.3978/j.issn.2304-3865.2014.03.03
Long, G.V., Stroyakovskiy, D., Gogas, H., Levchenko, E., de Braud, F., Larkin, J., . . . Flaherty, K. (2014). Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. New England Journal of Medicine, 371, 1877–1888. doi:10.1056/NEJMoa1406037
Ma, W., Agersborg, S., Brodie, S., De Dios, I., Chen, W., Yang, S., . . . Albitar, M. (2016). Companion testing using next-generation sequencing as compared with FDA-cleared kits [Abstract e23146]. Journal of Clinical Oncology, 34(Suppl.), e23146. doi: 10.1200/JCO.2016.34.15_suppl.e23146
Macdonald, J.B., Macdonald, B., Golitz, L.E., LoRusso, P., & Sekulic, A. (2015). Cutaneous adverse effects of targeted therapies: Part II: Inhibitors of intracellular molecular signaling pathways. Journal of the American Academy of Dermatology, 72, 221–236. doi:10.1016/j.jaad.2014.07.033
Mandalà, M., Massi, D., & De Giorgi, V. (2013). Cutaneous toxicities of BRAF inhibitors: Clinical and pathological challenges and call to action. Critical Reviews in Oncology/Hematology, 88, 318–337. doi:10.1016/j.critrevonc.2013.06.002
Mavropoulos, J.C., & Wang, T.S. (2014). Managing the skin toxicities from new melanoma drugs. Current Treatment Options in Oncology, 15, 281–301. doi:10.1007/s11864-014-0284-6
Medina, T.M., & Lewis, K.D. (2016). The evolution of combined molecular targeted therapies to advance the therapeutic efficacy in melanoma: A highlight of vemurafenib and cobimetinib. OncoTargets and Therapy, 9, 3739–3752. doi:10.2147/OTT.S86774
Menzies, A.M., Ashworth, M.T., Swann, S., Kefford, R.F., Flaherty, K., Weber, J., . . . Daud, A. (2015). Characteristics of pyrexia in BRAFV600E/K metastatic melanoma patients treated with combined dabrafenib and trametinib in a phase I/II clinical trial. Annals of Oncology, 26, 415–421. doi:10.1093/annonc/mdu529
Mutual Pharmaceutical Company. (2009). ColcrysTM (colchicine) [Package insert]. Retrieved from http://www.accessdata.fda.gov/drugsatfda_docs/label/2009/022351lbl.pdf
Nachimuthu, S., Assar, M.D., & Schussler, J.M. (2012). Drug-induced QT interval prolongation: Mechanisms and clinical management. Therapeutic Advances in Drug Safety, 3(5), 241–253. doi:10.1177/2042098612454283
National Cancer Institute. (2010). Common Terminology Criteria for Adverse Events (CTCAE) [v.4.03]. Retrieved from http://bit.ly/2ujuDrN
Novartis. (2016). Tafinlar® (dabrafenib) [Package insert]. Retrieved from https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/fil…
Novartis. (2017a). Mekinist® (trametinib) [Package insert]. Retrieved from https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/fil…
Novartis. (2017b, March 16). Storage, stability, and handling of mekinist tablets [Standard response letter to Valerie Guild, AIM at Melanoma Foundation].
Robert, C., Karaszewska, B., Schachter, J., Rutkowski, P., Mackiewicz, A., Stroiakovski, D. . . . Schadendorf, D. (2015). Improved overall survival in melanoma with combined dabrafenib and trametinib. New England Journal of Medicine, 372, 30–39. doi:10.1056/NEJMoa1412690
Rubin, K.M. (in press). Care and management of unique toxicities associated with MAPK pathway-targeted therapies in patients with advanced melanoma. Clinical Journal of Oncology Nursing.
Schoenberger, S.D., & Kim, S.J. (2013). Bilateral multifocal central serous-like chorioretinopathy due to MEK inhibition for metastatic cutaneous melanoma. Case Reports in Ophthalmological Medicine, 2013, 673796. doi:10.1155/2013/673796
Segaert, S. (2008). Management of skin toxicity of epidermal growth factor receptor inhibitors. Targeted Oncology, 3, 245–251. doi:10.1007/s11523-008-0092-7
Segaert, S., & Van Cutsem, E. (2005). Clinical signs, pathophysiology and management of skin toxicity during therapy with epidermal growth factor receptor inhibitors. Annals of Oncology, 16, 1425–1433. doi:10.1093/annonc/mdi279
Sinha, R., Edmonds, K., Newton-Bishop, J.A., Gore, M.E., Larkin, J., & Fearfield, L. (2012). Cutaneous adverse events associated with vemurafenib in patients with metastatic melanoma: Practical advice on diagnosis, prevention and management of the main treatment-related skin toxicities. British Journal of Dermatology, 167, 987–994. doi:10.1111/bjd.12010
U.S. Food & Drug Administration. (2017). List of cleared or approved companion diagnostic devices (in vitro and imaging tools). Retrieved from http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/InVitroD…
Valentine, J., Belum, V.R., Duran, J., Ciccolini, K., Schindler, K., Wu, S., & Lacouture, M.E. (2015). Incidence and risk of xerosis with targeted anticancer therapies. Journal of the American Academy of Dermatology, 72, 656–667. doi:10.1016/j.jaad.2014.12.010
Welsh, S.J., & Corrie, P.G. (2015). Management of BRAF and MEK inhibitor toxicities in patients with metastatic melanoma. Therapeutic Advances in Medical Oncology, 7, 122–136. doi:10.1177/1758834014566428




