Efficacy and Safety of Treating Glioblastoma With Tumor-Treating Fields Therapy
Background: Glioblastoma (GBM) is a highly aggressive astrocytoma with a dismal prognosis. Since 1976, only three chemotherapeutic agents have been approved for the treatment of GBM. Tumor-treating fields (TTFields) therapy, delivered via a noninvasive device, is a new therapy approved for use in patients with recurrent GBM and in combination with temozolomide for the treatment of newly diagnosed GBM.
Objectives: This article reviews the mechanism of action and findings from preclinical and clinical studies supporting the use of TTFields for patients with newly diagnosed and recurrent GBM.
Methods: This article provides an overview of published literature on the efficacy and safety of treating GBM with TTFields.
Findings: For the first time in more than a decade, patients with GBM have a noninvasive treatment option that has been shown to increase progression-free survival and overall survival with minimal adverse events.
Jump to a section
Glioblastoma (GBM) is a World Health Organization grade 4 astrocytoma, the highest grade. Astrocytomas are the most common form of diffuse gliomas, a group of primary central nervous system tumors that most frequently originate in the central hemisphere of adult brains (Appin & Brat, 2015; Crespo et al., 2015). GBM accounts for 15% of all primary brain tumors and 46% of primary malignant brain tumors (Ostrom et al., 2015). The majority of GBMs arise as a highly invasive and aggressive disease (de novo) without any associated clinical, histopathologic, or radiologic evidence of prior lower-grade lesions (Crespo et al., 2015). About two-thirds of patients with de novo GBM are older aged and present with less than a three-month clinical history, characterized by accelerated development of symptoms. In contrast, patients who develop a secondary GBM, which are most often derived from the transformation or progression of lower-grade astrocytomas (Crespo et al., 2015) are 10–15 years younger and have a prolonged course of treatment that eventually reaches a state where symptom burden is high and most therapeutic options have been exhausted (Hassler et al., 2015).
Since 1976, when lomustine (Ceenu®) was approved as a single agent for intracranial tumors, only three new drugs have been approved for treating brain cancers. In 1996, the carmustine wafer (Gliadel®) was approved for recurrent GBM and is now approved for newly diagnosed GBM as an adjunct to surgery. In 1999, temozolomide (TMZ) (Temodar®) was approved for patients with grade 3 anaplastic astrocytoma. This indication was subsequently expanded to newly diagnosed GBM with concomitant radiation followed by TMZ maintenance therapy (Stupp et al., 2005). In 2009, bevacizumab (Avastin®) was granted accelerated approval for patients with progressive GBM after prior therapy.
Optune®, a portable, noninvasive device that delivers tumor-treating fields (TTFields), was initially approved by the U.S. Food and Drug Administration (FDA) in 2011 for the treatment of adults with histologically confirmed GBM that recurred or progressed after receiving chemotherapy. In 2015, the FDA expanded Optune’s indication for use in combination with TMZ for patients with newly diagnosed GBM following surgery, chemotherapy, and radiation therapy.
Prior to the approval of TTFields, standard treatment for GBM consisted of maximal safe resection with or without carmustine wafer, or stereotactic or open biopsy or subtotal resection if maximal safe resection is not feasible, followed by standard or hypofractionated focal brain radiation therapy and concurrent adjuvant TMZ (Nabors et al., 2013). For disease recurrence, standard of care included systemic chemotherapy, surgery, re-irradiation, or alternating electric field therapy (Nabors et al., 2013). However, despite treatment, most patients will die within the second year following initial diagnosis. GBM has a dismal prognosis, with median progression-free survival (PFS) reported at 6.2–7.5 months and overall survival (OS) at 14.6–16.7 months (Stupp et al., 2015).
In 2015, TTFields therapy was added to the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology for central nervous system cancers, recommending that providers consider the therapy for patients with GBM who progress or recur after initial treatment. Establishing a national consensus among providers led to the upgrading of TTFields from a category 3 to a category 2B treatment (National Comprehensive Cancer Center, 2015).
Tumor-Treating Fields Mechanism of Action
Compared with historic cancer treatment modalities, TTFields have an innovative mechanism of action. Nonbiochemical and nonablative, TTFields use frequency-specific, low-intensity, alternating electric fields to disrupt structures within the cancer cell during mitosis, leading to apoptosis. Specifically, TTFields technology takes advantage of the special characteristics, geometric shape, and the rate of dividing cancer cells, which make them susceptible to the effects of TTFields. TTFields alter tumor cell polarity at an intermediate frequency (100–300 kHz). The frequency used for TTFields is specific to the target cell type (e.g., 200 kHz for gliomas). TTFields have been shown to disrupt the normal assembly of the microtubule spindle by exerting directional forces on polar intracellular elements, such as macromolecules and organelles. These processes lead to physical disruption of the cell membrane and to programmed cell death (Giladi et al., 2015) (see Figure 1). 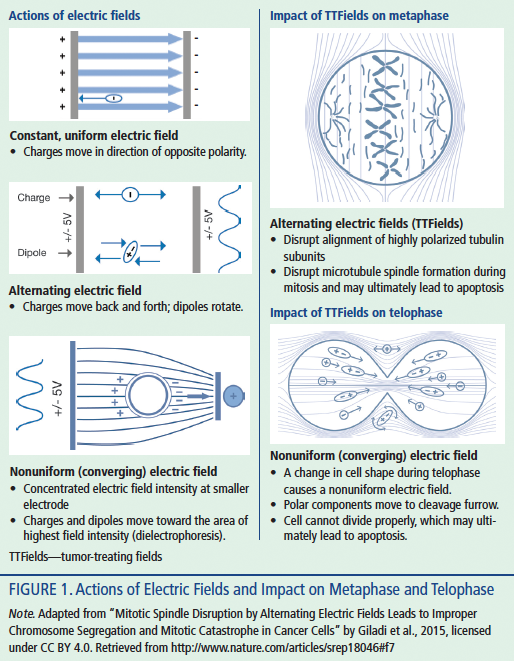
TTFields at a frequency of 200 kHz do not stimulate nerves and muscles and will have minimal or no effect on normal quiescent cells. Because TTFields are a local therapy and are delivered by transducer arrays to the brain of patients with GBM, they do not affect rapidly proliferating cells in the rest of the body. In addition, most normal adult brain cells proliferate very slowly, if at all; therefore, they are hypothesized to be minimally affected by TTFields (Kirson et al., 2007).
No differences were detected between TTFields-treated and untreated control animals upon histologic examination of major internal organs, and cardiac rhythm, body temperature, and animal behavior were also found to be unaffected (Kirson et al., 2007). The intensities of the electric fields within the tissues are small and do not result in any clinically meaningful increase in tissue temperature (Kirson et al., 2007).
TTFields are delivered using noninvasive, insulated transducer arrays that are placed directly on the skin in the region surrounding the tumor. The complete delivery system includes a portable electric field generator, transducer arrays, rechargeable batteries, a power supply, and a carrying case. Because TTFields are physical antimitotic modalities with no half-life, their application should be continuous (Gutin & Wong, 2012). Therefore, the device is designed to be worn continuously. The portable delivery system allows patients to perform their daily activities while receiving treatment.
Preclinical Studies
A series of publications and conference presentations (2004–2010) explored the applicability range of TTFields in a variety of in vitro and in vivo cancer models, either alone or in combination with standard chemotherapy (Gutin & Wong, 2012; Kirson et al., 2009). These studies showed that TTFields effectively inhibited cancer cell growth in various cell lines in vitro. In addition, the combination of TTFields with different chemotherapeutic agents revealed additive, if not synergistic, effects (Kirson et al., 2009). Tumor growth inhibition was also reported in in vivo animal models. TTFields were delivered using a noninvasive single electrically insulated transducer array placed on the head or torso surrounding the region of the tumor. The additive effect of TTFields with chemotherapy seen in vitro was recapitulated in different in vivo models (Gutin & Wong, 2012).
Pilot Clinical Study in Glioblastoma
TTFields were first studied in patients in a feasibility trial (n = 6) in 2003 (Salzberg, Kirson, Palti, & Rochlitz, 2008). Although the number of participants tested was small, the lack of adverse events and the efficacy observed indicated the potential of TTFields to serve as a new treatment modality for solid tumors (Salzberg et al., 2008). Efficacy and safety were evaluated in a clinical trial involving 10 participants with recurrent GBM in a single-center pilot study in 2004 (Kirson et al., 2007). In this open-label, prospective, single-arm study, patients were treated with TTFields as monotherapy with multiple four-week treatment courses using TTFields continuously for 24 hours per day and were followed for six months after disease progression. TTFields monotherapy led to a significant increase in time to disease progression (from 13–26 weeks; p = 0.013), six-month PFS (from 15%–50%), and OS (from 6–14.7 months; p = 0.002) compared to matched concomitant and historic controls. With more than 70 months of cumulative treatment, the only device-related adverse event seen in the trial was a mild to moderate skin irritation beneath the device electrodes (Kirson et al., 2007). Efficacy is increased when TTFields are applied continuously for more than 16 hours (Kirson et al., 2007); therefore, patients in this pilot study were treated for an average of 18 hours daily until disease progression.
In another pilot study, the efficacy and safety of TTFields were evaluated for patients with newly diagnosed GBM (Kirson et al., 2009). The study design was similar to the pilot study for recurrent GBM, and all patients received TMZ concurrently as maintenance therapy. The therapy was well tolerated, with the exception of mild to moderate skin irritation in 9 of 10 patients. Compared with historic controls, this study demonstrated a significant increase in PFS with TTFields, with 155 versus 31 weeks. In addition, median OS was greater, with 40 versus 14.7 months.
Pivotal Clinical Study in Recurrent Glioblastoma
Based on the results of the pilot trial for recurrent GBM, a pivotal phase III multicenter clinical study was initiated in patients with recurrent GBM (Stupp et al., 2012). The study was a prospective, randomized, open-label, active parallel control trial to compare the effectiveness and safety outcomes of recurrent GBM in participants treated with TTFields only (n = 120) to those treated with effective, standard-of-care chemotherapy (including bevacizumab) (n = 17). Median survival for the TTFields-treated group was 6.6 versus 6 months (hazard ratio [HR] = 0.86; 95% confidence interval [0.66, 1.12]; p = 0.27), and the one-year survival rate was 20% for both arms. Rate of PFS at six months was 21% for TTFields compared to 15% for the control arm (p = 0.13). Similar results showing comparability of TTFields to standard-of-care chemotherapy were seen in all secondary endpoints (PFS, six-month PFS, one-year survival rate, radiologic response rate, quality of life [QOL], and safety) (Stupp et al., 2012).
In general, patients who received TTFields experienced fewer adverse events, significantly fewer treatment-related adverse events, and significantly lower gastrointestinal, hematologic, and infectious adverse events compared to the controls. Patients assigned to the active control group received chemotherapy at the local investigators’ discretion. The best available chemotherapy was prescribed according to local practice and depending on prior treatment exposure. The only device-related adverse events observed were mild to moderate skin irritation beneath the device electrodes that were easily treated with topical ointments. Nursing management of manageable and reversible dermatologic adverse events includes prophylactic interventions (proper shaving, cleansing of wound, and array relocation) that diminish the risk of these side effects and treatment interventions (Lacouture et al., 2014).
Specific QOL measures were better for patients in the TTFields-treated group compared to patients receiving standard-of-care chemotherapy. No significant differences were found in global health and social functioning. Cognitive, emotional, and role functioning were better with TTFields treatment. Although physical functioning was reported to be slightly worse in the TTFields arm, chemotherapy-related symptoms (e.g., decreased appetite, constipation, diarrhea, nausea, vomiting) were found to be directly associated with standard-of-care chemotherapy, as were pain and fatigue. These longitudinal QOL measures were analyzed in 27% (n = 63) of the patients for whom QOL data was available and who remained in the study for more than three months (Stupp et al., 2012). The results of the study were presented at the 2010 American Society of Clinical Oncology Annual Meeting and were updated at the 2011 Society for Neuro-Oncology Annual Meeting (Novocure, 2016; Stupp et al., 2010, 2011). This study led to the 2011 FDA approval of the use of TTFields for treatment of adult patients with histologically confirmed GBM in the supratentorial region of the brain following recurrence after receiving chemotherapy.
Expanded Indication in Newly Diagnosed Glioblastoma
The results of a large, multinational, open-label, randomized phase III trial (Stupp et al., 2015) comparing TTFields in combination with TMZ (n = 466) to TMZ alone (n = 229) in patients with newly diagnosed GBM led to FDA approval of the expanded indication in 2015. Interim analysis demonstrated that two-year survival among patients treated with TTFields in combination with TMZ was 48%, compared to 32% in patients treated with TMZ alone. In addition, those in the treatment arm demonstrated a statistically significant increase in median PFS compared to the standard-of-care arm in the intent-to-treat group (7.2 versus 4 months; HR = 0.62, p = 0.001) and a statistically significant increase in median OS (20.5 versus 15.6 months; HR = 0.66, p = 0.004). Similar to the study in recurrent GBM, with the exception of easily manageable and reversible mild to moderate skin irritation, no significant toxicities were reported in patients in the treatment arm (Stupp et al., 2015). TTFields therapy is the first FDA-approved therapy in more than a decade to demonstrate a statistically significant survival advantage in patients with newly diagnosed GBM.
Implications for Nursing
TTFields therapy is a targeted, regional treatment modality for GBM based on the delivery of alternating electric fields to the tumor cells. The dividing malignant cells are disrupted in their growth, leading to cell death. In contrast to chemotherapy, TTFields therapy involves a physical, nonchemical mechanism of action; therefore, TTFields do not have a half-life. Antitumor activity is interrupted as soon as the therapy is stopped, which justifies the need for continuous application. The optimal daily duration of TTFields treatment is greater than 18 hours per day (Rehman, Elmore, & Mattei, 2015).
In clinical studies, TTFields proved to be effective, safe, and tolerable, with improvements in median PFS and OS and no significant increase in toxicity when used concurrently with standard of care. In the era of precision medicine initiatives, TTFields have changed the face of GBM management by contributing a modality that improves survival, minimizes toxicities, and increases QOL.
Oncology nurses play a critical role in educating patients and caregivers about the benefits of proper adherence to TTFields. In addition to discussing the clinical evidence and training on electrode application, nurses need to emphasize the correlation between therapy compliance and efficacy. In the postmarketing registry data of GBM patients on TTFields (from 2011–2013), a correlation was reported between adherence and patient outcome (median OS) (Mrugala et al., 2014). This correlation was also seen in the EF-11 clinical trial patients (Stupp et al., 2012). 
Conclusion
The pivotal, randomized phase III study leading to the FDA approval of Optune for recurrent GBM and the subsequent prospective, multicenter, open-label, randomized, controlled trial leading to the approval of Optune for patients with newly diagnosed GBM demonstrate that the addition of TTFields to maintenance TMZ chemotherapy significantly improves survival without increasing toxicities in patients with glioblastoma. The introduction of the second-generation device, which weighs about 50% less than the original device, is expected to improve GBM patient compliance. Trials are evaluating the safety and efficacy of TTFields in low-grade gliomas, solid tumor brain metastases, non-small cell lung cancer, pancreatic cancer, and other solid malignancies.
References
Appin, C.L., & Brat, D.J. (2015). Biomarker-driven diagnosis of diffuse gliomas. Molecular Aspects of Medicine, 45, 87–96. doi:10.1016/j.mam.2015.05.002
Crespo, I., Vital, A.L., Gonzalez-Tablas, M., Patino Mdel, C., Otero, A., Lopes, M.C., . . . Tabernero, M.D. (2015). Molecular and genomic alterations in glioblastoma multiforme. American Journal of Pathology, 185, 1820–1833. doi:10.1016/j.ajpath.2015.02.023
Giladi, M., Schneiderman, R.S., Voloshin, T., Porat, Y., Munster, M., Blat, R., . . . Palti, Y. (2015). Miotic spindle disruptions by alternating electric fields leads to improper chromosome segregation and miotic catastrophe in cancer cells. Scientific Reports, 5, 18046. doi:10.1038/srep18046
Gutin, P.H., & Wong, E.T. (2012). Noninvasive application of alternating electric fields in glioblastoma: A fourth cancer treatment modality. American Society of Clinical Oncology Educational Book, 126–131. doi:10.14694/EdBook_AM.2012.32.126
Hassler, M.R., Sax, C., Flechl, B., Ackerl, M., Preusser, M., Hainfellner, J.A., . . . Marosi, C. (2015). Thalidomide as palliative treatment in patients with advanced secondary glioblastoma. Oncology, 88, 173–179. doi:10.1159/000368903
Kirson, E.D., Dbalý, V., Tovarys, F., Vymazal, J., Soustiel, J.F., Itzhaki, A., . . . Palti, Y. (2007). Alternating electric fields arrest cell proliferation in animal tumor models and human brain tumors. Proceedings of the National Academy of Sciences of the United States of America, 104, 10152–10157. doi:10.1073/pnas.0702916104
Kirson, E.D., Schneiderman, R.S., Dbalý, V., Tovarys, F., Vymazal, J., Itzhaki, A., . . . Palti, Y. (2009). Chemotherapeutic treatment efficacy and sensitivity are increased by adjuvant alternating electric fields (TTFields). BMC Medical Physics, 9, 1. doi:10.1186/1756-6649-9-1
Lacouture, M.E., Davis, M.E., Elzinga, G., Butowski, N., Tran, D., Villano, J.L., . . . Wong, E.T. (2014). Characterization and management of dermatologic adverse events with the NovoTTF-100A System, a novel anti-mitotic electric field device for the treatment of recurrent glioblastoma. Seminars in Oncology, 41(Suppl. 4), S1–S14.
Mrugala, M.M., Engelhard, H.H., Tran, D.D., Kew, Y., Cavaliere, R., Villano, J.L., . . . Butowski, N. (2014). Clinical practice experience with NovoTTF-100A™ system for glioblastoma: The Patient Registry Dataset (PRiDe). Seminars in Oncology, 41(Suppl. 6), S4–S13
Nabors, L.B., Ammirati, M., Bierman, P.J., Brem, H., Butowski, N., Chamberlain, M.C., . . . Ho, M. (2013). Central nervous system cancers. Journal of the National Comprehensive Cancer Network, 11, 1114–1151.
National Comprehensive Cancer Network. (2015). Clinical Practice Guidelines in Oncology: Central nervous system cancers [v.1.2015]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/cns.pdf
Novocure. (2016). Updated survival data from study comparing TTF™ (tumor treating fields) therapy to best standard chemotherapy for recurrent glioblastoma reported at the 16th annual scientific meeting of the Society for NeuroOncology [Press release]. Retrieved from http://www.novocure.com/~/media/Files/N/Novocure/press-release/2011/201…
Ostrom, Q.T., Gittleman, H., Fulop, J., Liu, M., Blanda, R., Kromer, C., . . . Barnholtz-Sloan, J.S. (2015). CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2008–2012. Neuro-Oncology, 17(Suppl. 4), iv1–iv62. doi:10.1093/neuonc/nov189
Rehman, A.A., Elmore, K.B., & Mattei, T.A. (2015). The effects of alternating electric fields in glioblastoma: Current evidence on therapeutic mechanisms and clinical outcomes. Neurosurgical Focus, 38(3), E14. doi:10.3171/2015.1.FOCUS14742
Salzberg, M., Kirson, E., Palti, Y., & Rochlitz, C. (2008). A pilot study with very low-intensity, intermediate-frequency electric fields in patients with locally advanced and/or metastatic solid tumors. Onkologie, 31, 362–365. doi:10.1159/000137713
Stupp, R., Kanner, A.A., Engelhard, H., Heidecke, S., Tallibert, F.S., Lieberman, V., . . . Gutin, P.H. (2010). A prospective, randomized, open-label, phase III clinical trial of NovoTTF-100A versus best standard of care chemotherapy in patients with recurrent glioblastoma [Abstract LBA2007]. Retrieved from http://meetinglibrary.asco.org/content/40532
Stupp, R., Mason, W.P., van den Bent, M.J., Weller, M., Fisher, B., Taphoorn, M.J.B., . . . Mirimanoff, R.O. (2005). Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. New England Journal of Medicine, 352, 987–996.
Stupp, R., Taillibert, S., Kanner, A.A., Kesari, S., Steinberg, D.M., Toms, S.A., . . . Ram, Z. (2015). Maintenance therapy with tumor-treating fields plus temozolomide vs. temozolomide alone for glioblastoma: A randomized clinical trial. JAMA, 314, 2535–2543. doi:10.1001/jama.2015.16669
Stupp, R., Wong, E.T., Kanner, A.A., Steinberg, D., Engelhard, H., Heidecke, V., . . . Gutin, P.H. (2012). NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: A randomised phase III trial of a novel treatment modality. European Journal of Cancer, 48, 2192–2202. doi:10.1016/j.ejca.2012.04.011
About the Author(s)
Marlon Garzo Saria, PhD, RN, AOCNS®, FAAN, is an advanced practice RN and the director of the Center for Quality and Outcomes Research in the Department of Translational Neuro-Oncology and Neurotherapeutics; and Santosh Kesari, MD, PhD, FANA, FAAN, is the chair of the Department of Translational Neuro-Oncology and Neurotherapeutics, a professor of neurosciences at John Wayne Cancer Institute, and director of Neuro-Oncology, both at the John Wayne Cancer Institute at Providence Saint John’s Health Center in Santa Monica, CA. The authors take full responsibility for the content of the article. Kesari has previously consulted for and received honorarium from Novocure, Inc. Editorial support was provided by Stephen Swyberius, BA, copy supervisor at PharmaHealthLabs, through support from Novocure, Inc. The content of this article has been reviewed by independent peer reviewers to ensure that it is balanced, objective, and free from commercial bias. No financial relationships relevant to the content of this article have been disclosed by the independent peer reviewers or editorial staff. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Clinical Journal of Oncology Nursing or the Oncology Nursing Society. Saria can be reached at sariam@jwci.org, with copy to editor at CJONEditor@ons.org. (Submitted May 2016. Revision submitted July 2016. Accepted for publication July 19, 2016.)




